- 1. Introduction: 2026 — The Reset Year for the Global Biopharmaceutical Supply Chain at Bio Convention 2026
- 2. Technological Frontiers and Supply Logic: Business Realities of ADCs, GLP-1, and Nucleic Acid Therapeutics at Bio Convention 2026
- 3. Geopolitics and Risk Hedging: Procurement Philosophy from a U.S. Domestic Perspective at Bio Convention 2026
- 4. DCAT On-Site Tactics: New York Elite's "Secret Path" to Success at Bio Convention 2026
- 5. Value Conversion: Transform a 4-day Bio Convention 2026 into a 365-day Project Milestone
- 6. Conclusion: Seeking Reliable Allies in an Era of Uncertainty at Bio Convention 2026
1. Introduction: 2026 — The Reset Year for the Global Biopharmaceutical Supply Chain at Bio Convention 2026
If you attended any closed-door dinner gatherings for North American biopharmaceutical executives in the final quarter of 2025, you would have noticed that the conversations never revolved around breakthrough clinical data for a particular molecule or the latest FDA approval updates—as Bio Convention 2026 approaches, everyone was asking the same question: Is your supply chain ready?
This wasn’t casual small talk, but a sword of Damocles hanging over every CBO, supply chain leader, and even CEO at North American pharmaceutical companies. Over the past three years, we’ve witnessed the complete restructuring of geopolitical landscapes, disruptive shifts in regulatory frameworks, and the rapid disintegration of the global division of labor within the biopharmaceutical sector.And 2026 is neither the beginning nor the end of this transformation—it is the final critical window for all industry players to achieve a “soft landing” for their supply chains. It marks the reset year when the entire sector shifts decisively from “cost-driven globalization” to “resilience-driven regionalization.”
In this year of profound uncertainty, no industry platform can anchor certainty like New York DCAT Week.This note is not a generic online conference guide nor a rehash of the official agenda. It is a practical roadmap for all industry professionals, written by me—a CBO of a publicly listed company with 15 years of deep experience in North American pharmaceuticals, having attended 12 DCAT Weeks and secured over $500 million in business contracts through this platform.I will break down and explain in detail how to leverage DCAT Week’s momentum during the 2026 industry shakeup to secure supply chain opportunities in the three golden sectors—ADC, GLP-1, and nucleic acid therapeutics—and execute a full-chain strategy from business matching and risk hedging to project implementation.
1.1 The Paradigm Shift: From “Globalization” to “Regional Resilience”
The formal enactment of the Biosecure Act in 2025 isn’t the final straw breaking the back of global supply chains—it’s the watershed moment that fundamentally rewrites the industry’s rules of engagement.Prior to this, all supply chain discussions remained in the buffer phase of “risk warnings” and “alternative plans.” Post-implementation, every conversation has shifted to “hard compliance,” “hard deadlines,” and “hard switches”—leaving no room for ambiguity or luck.
First, let’s establish the most critical timeline for all industry players: The Act explicitly mandates that by 2027, all pharmaceutical companies receiving U.S. federal funding must completely sever production links for critical active pharmaceutical ingredients (APIs), highly potent active pharmaceutical ingredients (HPAPIs), and key excipients from “foreign entities of concern.”Even for companies not directly receiving federal funds, supply chain compliance will become a core prerequisite for FDA inspections, business mergers and acquisitions, and healthcare insurance access.This means 2026 represents the final full buffer year to complete supplier transitions, secure production capacity, and establish compliance frameworks. Failure to implement core supply chain alternatives by the end of 2026 will result in far more than compliance penalties in 2027—it could trigger product supply disruptions in the U.S. market, suspension of federal funding, or even project termination.
According to industry research released by the Pharma Supply Chain Association (PSCA) in January 2026: – 87% of North America’s Top 50 pharmaceutical companies completed structural adjustments to their supply chain budgets by 2025, significantly increasing the investment share for “regional resilience building” from 12% in 2023 to 38% in 2026;62% of companies have formally terminated at least one long-term supply contract originating from high-risk regions. More notably, 79% explicitly stated that their 2026 core business team KPIs include securing 2-3 compliant core alternative suppliers and completing critical material supply transitions.
Many practitioners ask: How should companies of different scales, facing varying compliance pressures and paces of action, align with the critical 2026 window? Drawing from legislative requirements, industry research data, and practical experience serving diverse-sized enterprises over the past year, I’ve compiled compliance milestones and action priorities for different company types. This helps you pinpoint core objectives and avoid blindly following trends:
| Business Type | Statutory Compliance Hard Line | 2026 Core Action Priorities | Core Attendance Goals for DCAT Week 2026 |
| Global Top 50 Big Pharma | 100% completion of high-risk regional supply chain divestment for federally funded projects by 2027; Full pipeline supply chain compliance integrated into executive KPIs and board audit items | 1. Secure second and third suppliers for core commercial products<br>2. Establish nearshore/friendly-shore capacity reserve systems<br>3. Complete full-pipeline supply chain compliance audits and risk ratings | 1. Sign long-term capacity reservation agreements with leading CDMO/API suppliers<br>2. Engage local HPAPI and excipient suppliers to complete commercial substitution<br>3. Participate in industry closed-door meetings to standardize supply chain compliance |
| Mid-sized Commercial Pharmaceutical Companies (Annual Revenue: $500M–$5B) | Complete high-risk supply chain transitions for core marketed products by 2027; FDA on-site inspection compliance is a deal-breaker | 1. Complete alternative supplier validation and transfer for core marketed products<br>2. Optimize supply chain layout for clinical pipelines to preemptively mitigate compliance risks<br>3. Establish supply chain compliance management systems | 1. Partner with 2-3 compliant CDMOs to secure clinical + commercial manufacturing capacity<br>2. Identify dedicated suppliers for core materials in specialized segments<br>3. Learn practical experience in building compliance systems from leading enterprises |
| Clinical-Stage Biotech | Supply chain compliance has become a core prerequisite for FDA IND/BLA approvals and a key due diligence threshold in M&A transactions | 1. Audit supplier compliance credentials for core materials in clinical pipelines<br>2. Pre-secure CDMOs meeting FDA compliance for clinical samples<br>3. Mitigate future supply chain compliance risks during commercialization | 1. Partner with compliance-focused CDMOs specializing in clinical stages to secure clinical sample production capacity<br>2. Collaborate with patent holders and core excipient suppliers to preemptively establish compliant supply chain systems<br>3. Engage with capital partners to optimize supply chain strategies and enhance pipeline valuation |
This trend never targets only Big Pharma—even small and medium-sized biotech companies cannot remain unaffected. Many biotech founders tell me, “We don’t receive federal funding, so the legislation doesn’t apply to us. There’s no need to fuss over the supply chain.” But my consistent reminder is: The rules of this industry are always set by the leading players.If your CDMO partner also serves Big Pharma companies receiving federal funding, their compliant production capacity will inevitably be prioritized for compliant projects. Your project could face the risk of capacity being squeezed out at any time.If you plan to commercialize your product in the U.S., FDA inspections will rigorously scrutinize supply chain compliance as a core audit item. Should you seek acquisition by a Big Pharma company, supply chain compliance has become the top priority in due diligence—by late 2025, three biotech M&A deals in North America had already collapsed due to supply chain compliance risks, with the largest transaction valued at $870 million. No acquirer will pay for your supply chain risks.
For the past 30 years, the foundational logic of the global pharmaceutical supply chain has been “cost-driven global division of labor.” We placed API manufacturing in lower-cost Asia-Pacific regions, formulation filling in areas with significant labor advantages, and clinical sample production in markets with flexible regulations. The sole objective was to maximize cost reduction and boost commercial profits.Back then, our core criteria for evaluating suppliers were price, delivery cycle, and production scale. Geopolitical risk was merely an inconspicuous addendum in contracts. Today, this paradigm has collapsed entirely. The industry’s foundational logic has shifted to “resilience-first regionalized Layout”—we no longer ask “Can this supplier offer me a lower price?” but first inquire “Can this supplier help me mitigate compliance risks?”We no longer concentrate all production capacity in a single region, instead adhering to the principle of “never putting all eggs in one basket.” We no longer pursue extreme cost optimization, but are willing to pay a reasonable premium for supply chain certainty.
To clarify the core shifts in this paradigm shift, I’ve compiled a comparison of the core logic guiding pharmaceutical supply chains across the old and new eras. This comparison also serves as the foundational starting point for all our business decisions:
| Core Evaluation Dimensions | Globalization Paradigm (1990–2022) | Regional Resilience Paradigm (2023–Present) |
| Core Decision Metrics | Production Costs, Economies of Scale, Delivery Efficiency | Compliance Security, Geopolitical Risk Controllability, Supply Redundancy |
| Capacity Deployment Logic | Single-region centralized layout to maximize cost advantages | Multi-regional decentralized layout with nearshoring/friendshoring outsourcing as core |
| Supplier Selection Priorities | 1. Price 2. Production capacity scale 3. Delivery cycle | 1. Compliance credentials 2. Geographic footprint 3. Technical alignment 4. Cost |
| Budget allocation focus | Production and procurement cost optimization, accounting for over 70% | Resilience building, compliance audits, and alternative supplier development, accounting for over 40% |
| Risk Management Model | Post-incident emergency response, focusing on delivery delay risks | Preemptive end-to-end forecasting, prioritizing geopolitical and systemic compliance risks |
| Business cooperation cycle | Short-term framework agreements with annual renegotiation for flexible switching | Long-term strategic partnerships with capacity commitments and deep alignment of interests |
Many ask me: Is this paradigm shift merely short-term policy volatility or a permanent industry reality? My answer is unequivocal: It is an irreversible long-term trend.Even if geopolitical tensions ease or regulatory rules undergo minor adjustments in the future, the industry will never return to the era of pure globalization. Every company has paid a steep price through this pandemic, geopolitical conflicts, and regulatory transformations, learning a fundamental truth: without supply chain certainty, even the most promising molecules, clinical data, or market prospects remain castles in the air.
The year 2026 represents the critical window for adapting to this new normal and completing supply chain restructuring. Missing this window will result in a complete loss of competitive initiative during the upcoming industry shakeout.
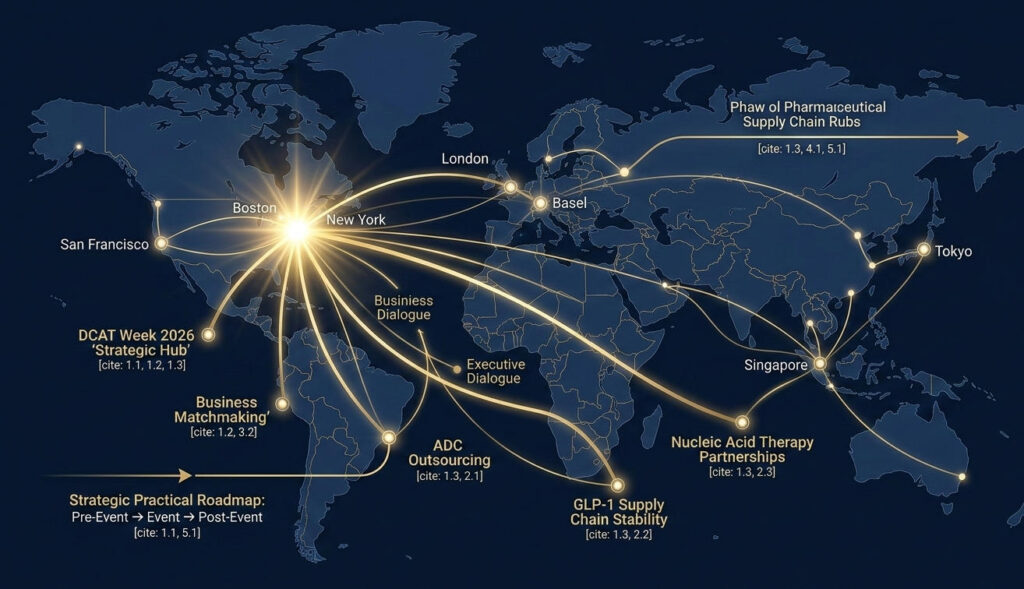
1.2 DCAT Week: The “Informal Davos” for Pharmaceutical Executives
If your understanding of DCAT Week remains limited to “a pharmaceutical industry exhibition in New York,” I can confidently say that over 80% of the opportunities at every previous event you attended went wasted.Having attended 12 editions of DCAT Week—from a BD specialist swiping business cards across floors to a CBO leading teams to secure annual business targets—my deepest insight is this: DCAT Week’s core value never resides in the main Javits Center venue, the official lecture schedule, or the bustling exhibition booths.Its true vitality lies in the hotel suites of Midtown Manhattan, the closed-door dinners at Lotte Palace, the private clubs on Fifth Avenue, and those one-on-one, off-the-record meetings that never appear on any official agenda.
This is precisely why executives in North America’s pharmaceutical circles refer to DCAT Week as the “informal Davos” of the pharmaceutical industry.The core value of the World Economic Forum in Davos has never been its official declarations, but rather the consensus and collaborations forged behind closed doors by global political and business leaders. DCAT Week operates similarly—it is the only platform where all core decision-makers in the global pharmaceutical supply chain converge in one city for four days, serving as the “invisible engine” driving business connections across the entire industry.
Many ask: With so many global pharmaceutical exhibitions, why has DCAT Week emerged as this pivotal hub? I’ve created a table to clearly break down the core differences between DCAT Week and other leading pharmaceutical exhibitions. You’ll instantly grasp its irreplaceable value:
| Exhibition Name | Core Positioning | Percentage of Core Decision-Makers Attending | Core Business Scenario | 2026 Industry Core Demand Alignment | Effective Business Matching Efficiency (Conversion Rate Within 3 Months Post-Event) |
| JPM Healthcare Conference | Investment, Financing, and Pipeline Value Unlocking | 35% (primarily CEOs, BD leads, and investors) | Pipeline Roadshows, Financing Matchmaking, M&A Negotiations | Moderate (Focused on capital side, low supply chain demand matching) | 28% |
| CPhI North America | Full-industry-chain product and technology exhibition | 22% (Primarily procurement and production managers; minimal executive participation) | Booth matchmaking, public lectures, product displays | Moderate to low (covers all categories but lacks precise decision-maker matching) | 22% |
| Biotech Showcase | Innovation pipelines and biotech financing | 27% (Primarily biotech founders and investors) | Pipeline pitches, early-stage funding connections | Low (Focused on R&D, minimal supply chain coverage) | 19% |
| 2026 DCAT Week | Pharmaceutical Supply Chain Full-Link Business Decision Matching | 81% (Chief Business Officers, Supply Chain VPs, Procurement Heads, CDMO CEOs/Commercial Presidents) | One-on-one private meetings, executive private dinners, dedicated business matchmaking sessions | Exceptionally High (Precise Matching for Core Needs: Supply Chain Restructuring, Capacity Lock-in, Compliance Substitution) | 82% (DCAT official post-2025 conference survey data) |
Official data shows that DCAT Week 2025 attracted over 11,000 attendees from 47 countries, spanning the entire industry chain from APIs, excipients, CDMO, and formulation production to distribution and capital. But here’s an industry truth: among these 11,000 attendees, fewer than 1,000 hold actual business decision-making authority—those who can finalize contracts and secure production capacity.These individuals include: – Global Supply Chain VPs, Chief Commercial Officers, and Procurement Heads at Top 50 Pharma companies – CEOs and Commercial Presidents of leading global CDMO/CMO firms – Controlling shareholders of core material patent holders (HPAPI, LNP, etc.) – Core partners at Wall Street-focused PE/VC firms specializing in pharmaceuticalsThey won’t linger at main venue booths for more than an hour, nor attend generic public lectures. Their four days in New York are meticulously scheduled in 15-minute increments, entirely filled with one-on-one closed-door meetings.
Many ask me: Beyond data differences, what truly makes DCAT Week irreplaceable? Three core reasons underpin its status as the “informal Davos”:
1.2.1 New York’s status as a global business hub sets the efficiency ceiling for DCAT Week.
Midtown Manhattan is the epicenter of global capital and the core node for worldwide business connections. Here, you can accomplish in a single day what would normally require flying across three countries and five cities.Let me share a real example from my 2025 participation: At 8 a.m., I finalized a long-term supply framework for ADC toxin intermediates with a Swiss HPAPI manufacturer at Lotte New York Palace; By noon, I secured production capacity for Phase III clinical samples during a lunch meeting with an Irish CDMO executive at Grand Hyatt New York;At 3 PM, I finalized commercial supply negotiations with a U.S.-based excipient supplier at the Hilton Midtown; By 7 PM, I attended a closed-door dinner hosted by a Wall Street healthcare fund, where I secured strategic partnerships for our next-generation pipeline with investors focused on the ADC field. This level of cross-regional, end-to-end supply chain efficiency is unattainable at any other global industry exhibition.
1.2.2 DCAT Week’s industry positioning precisely addresses core pain points in the pharmaceutical supply chain.
Unlike other events focused on R&D, clinical trials, or investment, DCAT Week was conceived from the outset to facilitate business connections within the pharmaceutical supply chain. Its attendees aren’t lab researchers or clinical practitioners, but decision-makers with final say—business executives, supply chain managers, and procurement leaders.Attendees arrive with crystal-clear objectives: sourcing suppliers, securing production capacity, negotiating partnerships, and expanding distribution channels—all without any fluff. According to DCAT’s official 2025 post-event survey, over 82% of participants reported that all business cooperation agreements reached during the conference originated from closed-door meetings outside the official agenda. Less than 10% of cooperation intentions stemmed from main venue lectures or booth networking.My own experience aligns perfectly with this. Over the past five years, every single one of the over $500 million in supply contracts our company signed at DCAT Week originated from one-on-one meetings in hotels or discussions at private dinner tables—not a single deal was secured during the main conference sessions.
1.2.3. DCAT Week 2026 has hit the industry’s most critical demand point, with strategic value far exceeding previous years.
In previous years, the primary objective at DCAT Week was “cost reduction”—finding cheaper suppliers, negotiating lower procurement prices, and optimizing supply chain cost structures. But in 2026, every attendee’s core goal is “seeking certainty”—identifying compliant alternative suppliers, securing scarce production capacity, hedging geopolitical risks, and achieving a soft landing for supply chains.This industry-wide convergence of demand has transformed the 2026 DCAT Week into the central marketplace for supply chain restructuring across the entire sector. I can confirm that as of February 2026, 85% of the DCAT meeting schedules for supply chain leaders at the top 20 North American pharmaceutical companies I know are already fully booked. For senior commercial executives at leading CDMOs, their calendars were completely filled as early as January.In previous years, you could still secure meetings with key players upon arriving in New York. But in 2026, if you haven’t started scheduling appointments now, you’ll likely miss this pivotal opportunity for industry transformation.
I often tell my team: DCAT Week isn’t a conference for “learning”—it’s a battlefield for “battling.” You won’t find extensive industry knowledge lectures or cutting-edge trend presentations here. Instead, you’ll encounter direct business matchmaking, authentic industry demands, and the most critical decision-makers.Whether you can secure the partners you need, lock in the production capacity you require, and finalize your core business objectives for the entire year within these four days depends entirely on your understanding of this platform and the preparation you undertake beforehand.
1.3 Core Value and Guide to This Article
Before writing this guide, I scoured every online resource about DCAT Week. Ninety percent of the content was formulaic “attendance guides”: telling you when to register, how to book hotels, which lectures are at the main venue, and how booths are laid out. You can find all this on the DCAT website—it doesn’t require my time to write, nor is it worth yours to read.
The remaining 10% consists of broad industry trend analyses—discussing how hot ADCs are, the massive GLP-1 market, or the promising prospects of nucleic acid drugs. It’s all correct but useless chatter, offering no actionable value. After reading it, you’d still be clueless about who to approach at DCAT Week, what to discuss, or how to convert conference connections into actual contracts.
1.3.1 That’s why I set the tone for this guide from the start.
It is not a conference guide, nor an industry trend report. Instead, it is a practical, actionable roadmap for all pharmaceutical industry professionals—one that is replicable and implementable.Every piece of content here stems from my 15 years of hands-on industry experience, from the pitfalls I’ve navigated and deals I’ve closed across 12 DCAT Weeks, from the methodologies I’ve refined, and most crucially, from the “unspoken rules” of the industry—those unpublished insights gained through exchanges with hundreds of North American pharmaceutical executives, CDMO leaders, and supply chain specialists.
My core objective in writing this guide is to empower you to achieve business goals in just four days at DCAT Week—goals that might take you an entire year otherwise—during this pivotal 2026 supply chain reset. Amidst an uncertain industry landscape, I’ll help you firmly secure opportunities in three golden sectors: ADCs, GLP-1, and nucleic acid therapeutics.
Next, I will break down the entire practical logic across six chapters. Each chapter has clear actionable objectives—no empty rhetoric:
1.3.2 In Chapter Two, I’ll take you deep into the supply chain realities of these three golden sectors.
Many claim ADC capacity is scarce, GLP-1 expansion is difficult, and nucleic acid drugs have high compliance requirements. But no one tells you: Which specific production stage is truly scarce? Where exactly are the bottlenecks in scaling up? What are the compliance pitfalls?I’ll reveal the true scarcity of “high-toxicity payload production lines” in ADCs and how to pinpoint CDMO partners at DCAT Week with compliant capacity to execute your projects. I’ll deconstruct the GLP-1 landscape—the interplay between peptide synthesis and small-molecule conversion, the intersection of raw material shortages and green chemistry—and show you how to identify cross-industry collaboration opportunities at conferences.I’ll also integrate the latest 2026 regulatory developments to reveal how nucleic acid therapeutics can leverage the DCAT platform to secure upstream suppliers like LNP patent holders and build globally compliant downstream distribution networks. Every topic centers on “business implementation”—no meaningless technical lectures.
1.3.3 In Chapter Three, I’ll explain the core logic of supply chain risk hedging from the perspective of U.S.-based pharmaceutical executives.
Many struggle to connect with North American companies during collaborations because they fail to grasp their procurement philosophy. I’ll clarify what “Supply Chain Diversity” truly means to them and how to evaluate suppliers’ geographic footprints to meet their risk management requirements.I’ll dissect the underlying logic of “Nearshore Outsourcing” versus “Friendly-shore Outsourcing,” revealing which deployment model suits enterprises of different scales and stages. I’ll also demystify real-world AI-powered supply chain applications, demonstrating how digital tracking, predictive maintenance, and inventory transparency aren’t industry buzzwords—they’re tangible tools that genuinely reduce risk and boost efficiency, serving as your key competitive edge when engaging North American firms.
1.3.4 In Chapter Four, I’ll equip you with a comprehensive DCAT field methodology—the “secret path” of New York’s elite.
Many attendees wander aimlessly through the main venue like headless flies, collecting stacks of business cards only to return home without securing a single partnership. The core reason? They simply don’t understand the DCAT game rules.I’ll provide precise navigation for the Manhattan venue, showing you how to plan your route to meet your target contacts most efficiently. I’ll clearly explain why one-on-one meetings are more crucial than any large lecture, and guide you on how to schedule, prepare, and negotiate to turn these meetings into collaborations.I’ll reveal the core value of closed-door dinners and supplier private showcases, showing you how to secure invitations and navigate essential unspoken rules. I’ll even provide elevator pitch templates tailored for senior tech executives in ADC, GLP-1, and nucleic acid therapeutics—teaching you how to capture their attention in 30 seconds and secure deeper engagement opportunities.
1.3.5 In Chapter Five, I’ll teach you how to transform four days of conference outcomes into 365-day project milestones.
The biggest pitfall for many attendees is having lively discussions at the conference only to send a single email afterward that goes unanswered, wasting all those connections. I’ll provide a replicable post-conference CRM follow-up system, detailing the timing, focus, and approach for different types of partners. I’ll also give you ready-to-use follow-up email templates to ensure every connection made in New York translates into tangible business projects.Simultaneously, I’ll show you how to transform the industry insights gained during meetings into strategic decisions for your company. This approach will not only help you secure major deals but also elevate your strategic value and irreplaceability within the organization.
1.3.6. In the concluding Chapter 6, I will help you redefine the core value of DCAT Week and provide clear actionable guidance.
I’ll explain what DCAT Week truly means for you in 2026—a pivotal year for industry reset. Discover the preparatory steps you must take now to secure your desired outcomes at the conference. Ultimately, I aim to walk into New York’s conference halls with you, armed with clear objectives, and emerge with tangible major contracts to kickstart your 2026 growth trajectory.
If you’re ready to seize this DCAT Week opportunity and avoid being left behind in the industry’s supply chain shakeup, follow my rhythm step by step. Next, we’ll start by dissecting the supply chain business realities across three golden sectors—uncovering exactly what you need to seek and discuss at DCAT Week 2026.
2. Technological Frontiers and Supply Logic: Business Realities of ADCs, GLP-1, and Nucleic Acid Therapeutics at Bio Convention 2026

After 15 years navigating North America’s pharmaceutical landscape, I’ve witnessed countless “star pipelines” crumble—not due to lackluster molecular design or underwhelming clinical data, but because they faltered in the supply chain transition from clinical development to commercialization.By 2026, industry competition will no longer be a race for molecular innovation in the lab, but a battle for certainty at the supply chain end. Even if your ADC target is novel, your GLP-1 molecule highly active, or your nucleic acid drug delivery system cutting-edge, without a stable, compliant, and scalable supply chain to support it, everything remains mere theory.
This is precisely why the core value of DCAT Week 2026 for professionals in the three golden sectors far surpasses any R&D summit or academic conference. Here, you can identify key suppliers to resolve ADC capacity bottlenecks, connect with green chemistry technologies that address GLP-1 scaling anxieties, and build the globally compliant supply chain networks essential for nucleic acid drug commercialization.Next, drawing on frontline practical experience, I will break down and thoroughly explain the underlying supply chain logic of each track, the pitfalls the industry won’t disclose, and the core opportunities you must seize at DCAT Week.
2.1 The ADC “Delivery Race”: Securing Scarce Capacity
If you’ve tracked any ADC pipeline’s commercialization progress in 2025, you’ll deeply resonate with this insight: IND approval is merely the starting point. The true test lies in producing the drug on time and in compliance.
I’ve witnessed too many biotech founders—armed with impressive Phase I clinical data and oversubscribed funding from JP Morgan conferences—get stuck at the manufacturing stage: scouring North America for compliant high-potency production lines, only to face repeated delays in delivering Phase II samples. Their once-leading pipelines were overtaken by competitors.Worse still, some commercial-stage ADC drugs, hampered by insufficient production capacity, failed to meet market demand post-launch, watching helplessly as competitors eroded their market share.
By 2026, this ADC “delivery race” has reached a fever pitch. The core conflict lies in the imbalance between the explosive growth of clinical trials and commercial demands versus the extreme scarcity of compliant high-potency production capacity.
First, consider these industry figures released by the American Association for Cancer Research (AACR) in January 2026: By the end of 2025, over 1,400 ADC pipelines were in active development globally, with North America accounting for 42%—firmly ranking first worldwide.Throughout 2026, North America is projected to file over 320 new ADC IND applications, advance more than 55 pipelines into Phase III clinical trials, and see 8-10 new ADC drugs approved for market launch. This implies that in 2026, clinical sample production demand for ADC drugs in North America will surge by 270% compared to 2023, while commercial production demand will skyrocket by 390%.
This explosive demand growth stands in stark contrast to the near-stagnant supply of compliant production capacity. Here, we must correct a common industry misconception: the “ADC capacity” advertised by many CDMOs is entirely distinct from the high-potency toxin-loading (HPAPI) capacity that meets FDA compliance requirements and is truly needed.
The core barrier for ADC drugs lies in the conjugation technology of “monoclonal antibody + linker + toxin.” The highly potent active toxins (HPAPIs) involved are predominantly highly cytotoxic substances, with occupational exposure limits (OEL) typically requiring ≤10ng/m³. For certain second- and third-generation ADC toxins, OEL requirements can even reach ≤1ng/m³.This means production lines handling such substances must meet extreme standards for containment, isolation, safety protection, and waste treatment. They cannot be simply retrofitted from ordinary GMP monoclonal antibody facilities.
Let me break down the costs: A high-potency ADC production line compliant with FDA cGMP standards, meeting OEL ≤ 1 ng/m³, and capable of supporting commercial scale-up requires at least 3-5 years from design, construction, and validation through FDA inspection, with fixed asset investments exceeding $120 million. Even a pilot-scale line supporting only Phase I-II clinical trials demands over $20 million and a construction period of no less than 18 months.More critically, following the 2025 implementation of the Biosecurity Act, North American pharmaceutical companies’ geographic requirements for production capacity have shifted from “globally available” to “domestic U.S. + friendly offshore regions.” This has further widened the gap in already scarce compliant production capacity.
According to a February 2026 special survey by the Pharmaceutical Supply Chain Association (PSCA): By 2026, North America’s total available capacity for high-potency production lines meeting FDA cGMP standards with OEL ≤ 1 ng/m³ and suitable for ADC commercialization will only satisfy 35% of market demand, creating a 65% supply gap. Even for pilot-scale capacity supporting clinical stages, the supply-demand gap reaches 48%.The harsher reality is that as of February 2026, leading North American CDMOs have booked their high-potency production lines through the fourth quarter of 2027. Some top firms have even closed new order booking channels for 2027.
This is the reality of the 2026 ADC landscape: while barriers to molecular innovation are lowering, supply chain barriers are rising exponentially. Your ability to secure reliable CDMO partners and snag scarce production slots at DCAT Week directly determines whether your pipeline progresses on schedule and survives fierce competition.
But I must warn you: 90% of CDMOs claiming ADC capacity at DCAT Week are not your optimal choice—they could even be major pitfalls that derail your project.Over the past decade, I’ve engaged with over 100 CDMOs at DCAT Week claiming ADC production capabilities. Ultimately, fewer than 10 resulted in long-term collaborations without pitfalls. Next, I’ll share a battle-tested methodology I’ve refined over years to help you precisely identify reliable CDMO partners and secure genuinely available capacity at DCAT Week.
2.1.1 Step One: Screen for “Hard Credentials,” Reject “Paper Capacity”
Many attendees grab CDMO brochures, see phrases like “high-potency ADC production lines,” and immediately jump into discussions—this is the biggest mistake. During DCAT Week one-on-one meetings, don’t start with pricing or capacity. Instead, ask these three core questions. If the partner can’t answer one clearly or gives vague responses, end the meeting immediately—don’t waste your time:
- Has your high-potency production line undergone an FDA Pre-Approval Inspection (PAI) within the last 24 months? Do you possess comprehensive experience in manufacturing and delivering commercial ADC drugs?
- What is the OEL level your production line operates stably at? Do you have validation reports from third-party authoritative institutions? Can you provide environmental monitoring data for production conditions corresponding to that OEL level over a continuous period of 6 months or longer?
- Does your ADC production capability cover the entire process chain, or is it limited to a single step? Do you have independent project experience completing the full workflow encompassing “linker + toxin synthesis, conjugation reaction, purification, and formulation filling”?
Let me explain the core logic behind these three questions:
- The first question immediately eliminates 90% of “paper capacity.”Many CDMOs claim ADC capacity but only possess lab-scale pilot equipment. They’ve never passed FDA inspections and lack commercial delivery experience. ADC production involves exponentially increasing process complexity from pilot to scale-up to commercial manufacturing. CDMOs without commercial scale-up experience will likely face process failures or delivery delays when your project reaches Phase III clinical trials or commercialization.I fell into this trap in 2023: I selected a CDMO that advertised ADC capacity. While Phase I clinical samples delivered smoothly, scaling up to 500L for Phase II resulted in three consecutive batches failing purity standards. The project was delayed by seven months, incurring over $12 million in losses.
- The second issue is avoiding the pitfall of “false compliance.”Many CDMOs claim OEL ≤ 1 ng/m³ production capability, yet often only meet design standards without consistent operational compliance—let alone third-party validation data. FDA inspections for high-potency drugs focus critically on sustained occupational exposure risk control. Failure here risks immediate project suspension or inspection failure.
- Third, assess a CDMO’s true capabilities while mitigating supply chain fragmentation risks. The ADC production chain is exceptionally long—spanning monoclonal antibody expression, linker synthesis, toxin synthesis, conjugation reactions, purification, and formulation filling. A failure at any stage can derail the entire project timeline.If a CDMO only handles the conjugation step while you must source toxins and linkers from separate suppliers, you’ll not only juggle multiple partners and increase communication overhead but also face audit risks from multiple suppliers and supply chain disruption risks under Biosafety Act compliance requirements. By 2026, CDMOs offering full-chain ADC manufacturing services should be your primary target for collaboration.
To help you more intuitively assess a CDMO’s reliability, I’ve compiled a ready-to-use scoring sheet for evaluating core ADC CDMO capabilities. During DCAT Week meetings, you can directly score your CDMO partners using this sheet: exclude those scoring below 60 points, and only engage in deeper discussions and partnership consideration with those scoring 80 points or higher:
| Evaluation Dimensions | Core Evaluation Metrics | Weight | Scoring Criteria (Maximum 100) |
| Compliance Qualifications | FDA cGMP Certification and PAI Inspection Compliance | 25% | 25 points: Passed PAI inspection for commercial ADC projects within the last 24 months; 15 points: Passed GMP inspection only at the clinical stage; 5 points: No FDA inspection record; 0 points: Received FDA warning letter |
| Production Capacity & Delivery | High-Activity Production Line Scale, Available Slots, Historical On-Time Delivery Rate | 20% | 20 points: Available commercial production slots in 2026-2027 with 100% on-time delivery rate over past 2 years; 12 points: Only clinical-stage capacity available with ≥90% on-time delivery rate; 3 points: Capacity fully booked with <90% on-time delivery rate |
| Full-chain technical capabilities | Technical capabilities and project experience covering the entire ADC production process | 20% | 20 points: Commercial-scale experience across the entire chain including linker/toxin synthesis, conjugation, purification, and formulation; 10 points: Covers only 2-3 stages with clinical-stage experience; 0 points: Capable of completing only a single stage |
| Safety Control Capability | Production line OEL classification, validation reports, environmental monitoring data | 15% | 15 points: Consistently achieves OEL ≤ 1 ng/m³ with comprehensive third-party validation and ongoing monitoring data; 8 points: Achieves OEL ≤ 10 ng/m³ with foundational validation data; 0 points: No defined OEL level or validation reports |
| Geographic Layout | Does the production site comply with Biosafety Act requirements? | 10% | 10 points: 100% of production capacity located in the U.S. mainland/friendly offshore regions; 5 points: Partial production capacity located in compliant regions; 0 points: Core production capacity located in high-risk regions |
| Project Team | Core team’s ADC project experience and stability | 10% | 10 points: Core team possesses experience in 5+ ADC commercialization projects with stable personnel; 5 points: Team has clinical-stage project experience; 0 points: No dedicated ADC team identified |
2.1.2 Step Two: Secure Production Capacity, Not Just a Letter of Intent
A common misconception among attendees is that a pleasant discussion with a CDMO and obtaining a Letter of Intent (LOI) signifies mission accomplished. However, I must emphasize that in the 2026 production capacity crunch, an LOI without secured timelines or a signed capacity reservation agreement is worthless.
I’ve witnessed too many practitioners sign LOIs with CDMOs at DCAT Week, only to discover after internal approvals and audits—three months later—that the reserved production slots had been allocated to other clients, pushing their projects back over a year. Post-2025 DCAT Week, this scenario occurs over 60% of the time.
Therefore, your core objective during DCAT Week discussions isn’t finalizing a comprehensive cooperation agreement, but securing specific production line schedules and signing binding capacity reservation agreements. Here’s a practical negotiation script you can directly apply:
“Our ADC pipeline is currently in [Phase X clinical trials]. We anticipate requiring a high-potency production line with [X L capacity] in [QX of 202X] for [clinical sample production/commercial batch manufacturing].We highly value your company’s technical capabilities and regulatory compliance. We aim to confirm your available time slots for the corresponding period during this meeting and sign a capacity reservation agreement. We are prepared to pay [10%-15% of the batch production cost] as a reservation deposit to secure exclusive access to this time slot, while also locking in the production quote and delivery timeline for the corresponding batch.”
The core of this negotiation tactic is using a deposit to secure time slots and leveraging confirmed demand to obtain guaranteed capacity. In the 2026 seller’s market, securing capacity with empty promises is nearly impossible. However, if you’re willing to pay a reasonable deposit, you’ll likely secure your desired time slots. After all, for CDMOs, locking in a deposit means locking in guaranteed revenue, making them willing to reserve capacity for you.
Additionally, here’s a negotiation tip exclusive to DCAT Week: Don’t fixate solely on top-tier CDMOs like Catalent, Lonza, or Samsung Biologics. Their capacity was locked up by the Top 20 Pharma companies by late 2025. Your chances of securing it are extremely low, and you’ll face sky-high quotes with extremely stringent payment terms.Your real focus should be mid-tier CDMOs with compliant high-potency capacity, commercial project experience, yet not top-tier market visibility—like U.S.-based AAIPharma and Alcami, Ireland’s Aenova, or Patheon’s Puerto Rico facility.These companies’ capacity hasn’t been fully locked up by top pharmaceutical firms, their quotes are more reasonable, and they place greater emphasis on biotech projects. They represent the “hidden gem partners” at DCAT Week 2026.
2.1.3 Step Three: Proactively Positioning for Next-Generation ADC Technology Supply Chains
Many companies engaging with ADC CDMOs focus solely on current pipeline needs while overlooking supply chain positioning for next-generation ADC technologies. By 2026, next-generation conjugates like bispecific ADCs, isotope-labeled ADCs, and peptide-drug conjugates (PDCs) will enter a clinical explosion phase. Manufacturing these drugs demands higher-activity production lines and more advanced conjugation technologies than traditional ADCs, with even scarcer capacity.
DCAT Week is the world’s only platform dedicated to connecting next-generation conjugated drug CDMOs. While securing capacity for existing pipelines, you can simultaneously negotiate strategic collaborations with CDMOs for next-generation technologies, preemptively securing dedicated R&D and manufacturing resources.For instance, you can sign a framework agreement with a CDMO to reserve dedicated R&D teams and production line slots for your next-generation ADC pipeline. This enables rapid advancement into manufacturing and clinical trials immediately upon molecular development completion, securing a competitive market advantage.
At DCAT Week 2024, I employed this approach to establish a strategic collaboration framework for a bispecific antibody-drug conjugate (ADC) with a U.S.-based CDMO. They reserved a dedicated R&D team and pilot production line for us. After completing the bispecific ADC molecule design, we achieved preclinical sample production in just four months—six months faster than the industry average. This directly positioned our pipeline as the first to enter clinical development among targets with the same target.
Ultimately, the 2026 ADC race boils down to a supply chain competition. Securing scarce compliant capacity ahead of rivals at DCAT Week and strategically positioning next-generation technology supply chains early grants absolute control in this delivery race.
2.2 GLP-1’s “Scalability Anxiety”: The Peptide vs. Small Molecule Dilemma
If the core challenge in the ADC space is “capacity scarcity,” then the central issue in the GLP-1 arena is “scalability anxiety.”
Across North America’s pharmaceutical industry, no one doubts GLP-1’s market potential. According to IQVIA’s January 2026 report, North America’s GLP-1 drug market surpassed $90 billion in 2025, is projected to exceed $120 billion in 2026, and will surpass $500 billion globally by 2030.Yet this explosive market demand stands in stark contrast to an industry-wide, persistent anxiety: Can we produce sufficient quantities of compliant GLP-1 active pharmaceutical ingredients (APIs) at scale and at an affordable cost?
I’ve spoken with CEOs and Chief Business Officers from dozens of North American pharmaceutical companies developing GLP-1 pipelines. Whether they’re generic drug manufacturers or biotech firms pursuing innovative pipelines, their anxieties are identical:
- For GLP-1 biosimilar manufacturers: – Solid-phase synthesis keeps API production costs high – Medicare negotiations relentlessly drive down prices, squeezing profit margins – Expanding capacity faces stringent EPA environmental regulations – Investing hundreds of millions in factories risks earning no profit
- Companies developing innovative GLP-1 peptides face challenges: Long-acting, multi-target peptide molecules are harder to synthesize and scale up. Clinical sample production requires six-month waitlists, let alone subsequent commercial-scale manufacturing.
- Companies developing small-molecule GLP-1 face this dilemma: while the molecules exhibit excellent activity and high oral bioavailability, chiral synthesis processes are extremely complex. Experience in scaling up production is virtually nonexistent, making it difficult to find partners capable of reliably achieving commercial-scale manufacturing.
This is the reality of the GLP-1 landscape in 2026: market demand has no ceiling, but your supply chain’s scaling capacity defines your ceiling. DCAT Week is your sole platform to overcome this scaling anxiety—here, you’ll discover core technologies to resolve raw material shortages, connect with partners to break through expansion bottlenecks, and find the optimal solution in the peptide vs. small molecule competition.
2.2.1 The Peptide vs. Small Molecule Game: Core Differences from a Supply Chain Perspective
First, I must explain the core differences between peptide GLP-1 and small-molecule GLP-1 from a supply chain perspective, along with their respective scaling bottlenecks. Many discussions about GLP-1 focus solely on molecular activity and clinical efficacy, overlooking the foundational logic of supply chains. Ultimately, even the most promising R&D efforts can falter during large-scale production.
Consider the currently dominant peptide GLP-1 drugs, such as semaglutide, tirzepatide, and Lilly’s tirzepatide. Their core advantage lies in their well-established clinical efficacy and high market acceptance. With patents expiring, the market potential for generics is enormous. However, their mass production bottlenecks are widely recognized across the industry:
Current mainstream peptide GLP-1 API production relies on solid-phase synthesis. Taking semaglutide as an example, the entire synthesis process involves over 30 reaction steps. Each step generates impurities, necessitating repeated purification steps to achieve the FDA-required purity of ≥99.5%. This process introduces three unavoidable bottlenecks for scaling up:
- Persistently high production costs: Solid-phase synthesis consumes substantial quantities of expensive solid-phase carriers and organic solvents. Batch yields remain extremely low, with current industry-wide yields for semaglutide API rarely exceeding 35%. This directly prevents significant reductions in API production costs.For generic drug manufacturers, future Medicare negotiation prices will continue to decline. Without reducing API costs, companies will ultimately face the dilemma of “the more you sell, the more you lose.”
- Environmental pressures are immense, and expansion is extremely challenging: Solid-phase synthesis processes generate substantial volumes of organic waste liquids and solid waste. The cost of treating these three wastes is prohibitively high. Moreover, the U.S. Environmental Protection Agency (EPA) is imposing increasingly stringent environmental requirements on the pharmaceutical industry. In 2025, the EPA updated pollutant emission standards for the pharmaceutical sector, tightening organic solvent emission limits by 70%.This means establishing a new peptide API plant in the U.S. requires 2-3 years just for environmental approvals, with fixed asset investments exceeding $500 million—creating extremely high barriers to expansion.
- Process scaling presents significant challenges: Transitioning peptide synthesis from laboratory pilot tests to ton-scale commercial production requires immense stability control. Many companies achieve high-purity samples in labs, but encounter purity failures and drastic yield drops during scale-up, directly hindering commercialization.
Next, consider next-generation small-molecule GLP-1 drugs like Lilly’s Orforglipron and Pfizer’s Danuglipron, which have already entered Phase III clinical trials, with some even submitting NDA applications. The core advantage of these drugs lies in their high oral bioavailability, leading to better patient compliance. They also involve far fewer synthesis steps than peptides, theoretically resulting in lower production costs and easier scaling up.However, from a supply chain perspective, they face their own core bottlenecks:
- Patent landscapes are highly congested, posing significant freedom-to-operate (FTO) risks: Core patents for small-molecule GLP-1s have been densely layered by industry leaders like Eli Lilly and Pfizer. From compound structures and crystal forms to synthesis processes and indications, virtually no gaps remain.Many small-molecule GLP-1 candidates from biotech companies appear highly active, but in-depth FTO analysis reveals patent pitfalls at every turn. This makes them prone to patent litigation, effectively blocking commercialization.
- Extreme difficulty in scaling chiral synthesis processes: Most small-molecule GLP-1 compounds feature multiple chiral centers, demanding exceptionally high stereoselectivity during synthesis. While purification via column chromatography is feasible at the laboratory scale, achieving stable, high-selectivity, high-yield synthesis at commercial ton-scale production requires highly sophisticated process development. Currently, only a handful of CDMOs and API manufacturers possess relevant large-scale experience in this field.
- Commercial-scale production validation remains virtually nonexistent: Currently, no oral small-molecule GLP-1 drug has been approved for market launch globally. All pipeline candidates remain in clinical stages, and the corresponding large-scale manufacturing processes have not undergone commercial validation. This means that even if you find a partner capable of pilot-scale production, there is no guarantee they can consistently achieve stable commercial-scale manufacturing—the uncertainty is extremely high.
To help everyone clearly see the supply chain differences between the two and the corresponding DCAT Week matchmaking priorities, I’ve compiled a core comparison table:
| Core Dimensions | Peptide-based GLP-1 | Small-Molecule GLP-1 | Key DCAT Week Matchmaking Focus for 2026 |
| Core Scaling Bottlenecks | Solid-phase synthesis involves multiple steps, low yields, significant environmental pressure, and challenges in scaling up | High scaling difficulty for chiral synthesis, significant patent barriers, and unvalidated commercial processes | Peptides: Green chemistry service providers, continuous flow synthesis CDMOs; Small molecules: Chiral synthesis CDMOs, patent law firms, crystal form technology service providers |
| Production Costs | High (scalable production costs approx. $1,800–$2,500/kg) | Theoretically low (estimated large-scale production cost: ~$800–1,200/kg), but high early-stage R&D costs | Peptides: Process technology partners capable of reducing production costs; Small Molecules: CDMOs capable of lowering process development costs |
| Compliance and Environmental Risks | High (Challenging EPA environmental approvals, stringent waste treatment requirements) | Moderate (Environmental pressure significantly lower than peptides; FDA imposes extremely stringent requirements for chiral impurity control) | Peptides: Green process technology partners meeting EPA environmental standards; Small Molecules: CDMOs with FDA submission experience for chiral drugs |
| Supply Chain Maturity | High (established commercial production systems; leading API manufacturers possess stable, scaled experience) | Low (No commercialization cases, immature process systems) | Peptides: API manufacturers with scalable capacity and production capacity reservation partnerships; Small Molecules: CDMOs with experience producing chiral drugs in clinical stages |
| Core Requirements for 2026 | Cost reduction, capacity expansion, resolving environmental bottlenecks | Process scale-up, patent risk mitigation, commercialization validation | Peptides: Cross-industry collaborations in green chemistry technologies; Small Molecules: Full-chain partnerships spanning process development and manufacturing |
This table clearly illustrates: Whether for peptides or small molecule GLP-1, overcoming scaling challenges hinges on technological innovation and cross-sector collaboration—precisely where the greatest opportunities lie at DCAT Week 2026.
2.2.2 The Intersection of Raw Material Shortages and Green Chemical Synthesis: The Breakthrough Core for GLP-1
In 2026, the sole breakthrough to resolve GLP-1 raw material shortages and scale-up anxieties lies in green chemical synthesis technology. This is not an industry buzzword, but a proven core technology that tangibly solves problems—and will be the fiercely sought-after collaboration hotspot at DCAT Week 2026.
Consider this real-world example: In 2024, a U.S. GLP-1 biosimilar manufacturer partnered with a company specializing in continuous flow chemistry. They transformed the solid-phase synthesis process for semaglutide into a combined continuous flow + liquid-phase synthesis workflow. This transformation directly reduced the number of reaction steps from 31 to 7, cut organic solvent consumption by 82%,waste generation decreased by 87%, production cycles shortened from 28 days to 36 hours, API production costs plummeted from $2,200/kg to $900/kg, and yield improved from 32% to 68%.
This case exemplifies the perfect convergence of raw material scarcity and green chemistry technology. For the GLP-1 industry, green chemistry isn’t merely a “cherry on top”—it’s a “lifeline in times of need.” It not only addresses the environmental challenges of solid-phase synthesis, enabling companies to secure EPA approvals and scale up production, but also drastically lowers costs, boosts efficiency, and fundamentally resolves scaling anxieties.
Currently, three primary green chemistry technologies are truly scalable for GLP-1 production. These represent the core technology partners you must prioritize at DCAT Week 2026:
- Continuous Flow Chemical Synthesis Technology: The most mature and widely implemented green chemistry technology today. Unlike traditional batch reactions, continuous flow chemistry enables uninterrupted chemical reactions within microchannel reactors. It allows precise control over reaction temperature, pressure, and material ratios, substantially improving selectivity and yield while drastically reducing solvent usage and waste generation.For GLP-1 peptides, continuous flow technology compresses reactions that previously took days into mere minutes while substantially lowering purification complexity. It stands as the most critical technology for cost reduction and efficiency gains today. At the 2025 DCAT Week, over 60% of peptide API manufacturers engaged with continuous flow technology providers, and this proportion will only increase in 2026.
- Enzyme-Catalyzed Synthesis Technology: Leveraging the high selectivity of biological enzymes, this technology enables directed peptide synthesis. It effectively resolves the challenges of high chiral impurity levels and difficult purification inherent in solid-phase synthesis processes.For instance, using enzyme-catalyzed technology to synthesize key peptide segments of semaglutide achieves nearly 100% stereoselectivity, virtually eliminating chiral impurities while significantly boosting yields. Additionally, the mild reaction conditions require minimal organic solvents, resulting in minimal environmental impact.Currently, multiple companies have achieved large-scale production of GLP-1 peptide fragments using enzyme-catalyzed technology. Within the next 2-3 years, this approach is poised to become one of the mainstream technologies for GLP-1 peptide production.
- Novel Liquid-Phase Synthesis Technology: While traditional liquid-phase synthesis is suitable for large-scale production, it poses significant purification challenges and is difficult to apply to long-chain peptide synthesis.The novel liquid-phase synthesis technology combines the advantages of solid-phase and liquid-phase synthesis. By employing new carriers and purification methods, it enables large-scale liquid-phase synthesis of long-chain peptides. Production costs are reduced by over 50% compared to solid-phase synthesis, while waste generation is decreased by more than 70%. This makes it highly suitable for ton-scale GLP-1 peptide production.
Here, I must caution everyone: At DCAT Week, many companies claim to possess green chemistry technologies. However, most of these remain at the laboratory stage, never having been implemented in large-scale peptide production, let alone validated for FDA compliance. Therefore, when engaging with technology service providers, you must ask three critical questions:
- Has your technology been implemented in the large-scale production of peptide drugs? Are there corresponding commercial project cases?
- Has the API produced using your technology passed FDA DMF filing? Do you have corresponding regulatory submission experience?
- Can you provide technical validation data at the pilot scale? Can you complete process transfer and scale-up within the agreed timeframe?
Only technical service providers that can provide clear answers to all three questions are truly worthy partners for collaboration. Otherwise, no matter how advanced their technical concepts or impressive their laboratory data may be, they cannot solve the practical challenges of large-scale production.
2.2.3 Cross-Industry Collaboration Opportunities at DCAT Week: The Core Path to Overcoming Scale-Up Bottlenecks
Many attendees seeking GLP-1 collaborations focus solely on API manufacturers and CDMOs, overlooking DCAT Week’s greatest opportunity: cross-industry partnerships. By 2026, GLP-1 production bottlenecks will stem not from isolated links but from supply chain coordination. Only cross-industry collaboration can fundamentally resolve these challenges.
Drawing on my years of conference experience and 2026 industry trends, I’ve identified three core cross-industry collaboration points at DCAT Week that you must prioritize:
2.2.3.1 Deep Integration Between Peptide API Manufacturers and Green Chemistry Technology Providers
This represents the most certain cross-industry collaboration opportunity at DCAT Week 2026. Currently, the vast majority of North American peptide API manufacturers possess mature GMP production systems, FDA filing experience, and large-scale production capabilities. However, their core weakness lies in insufficient R&D capacity for green chemistry technologies, hindering rapid process upgrades and iterations. This creates bottlenecks in cost reduction and capacity expansion.
Meanwhile, many service providers specializing in continuous flow and enzyme-catalyzed technologies possess advanced capabilities that could significantly reduce costs and boost capacity for API manufacturers. However, lacking GMP production licenses and FDA submission experience, they cannot independently commercialize their technologies.
The needs of both parties perfectly complement each other, and DCAT Week serves as the optimal platform for connecting these two types of companies. At the 2025 DCAT Week, I facilitated a strategic partnership between an Irish peptide API manufacturer and a U.S.-based continuous flow technology company:The API manufacturer contributed production facilities, GMP systems, and commercialization expertise, while the technology company provided continuous flow processes and technical support. Together, they upgraded the production process for semaglutide. Within just 10 months, they completed process validation and scale-up, tripling API production capacity while reducing manufacturing costs by 58%. The project also successfully passed EPA environmental approvals.
For the API manufacturer, this partnership enabled capacity doubling and cost reduction without massive new plant investment. For the technology provider, it achieved commercial deployment of its technology and secured ongoing revenue sharing. For biotech companies, DCAT Week offers a platform to connect with such pre-integrated partners, pre-book upgraded API capacity at lower procurement prices, and seize first-mover advantages in the generic drug market.
2.2.3.2 Early Strategic Collaboration Between Small-Molecule GLP-1 Developers and High-End Chiral Synthesis CDMOs
The core bottleneck for small-molecule GLP-1 development is process scale-up. Many biotechs fall into the trap of waiting until preclinical studies are complete—or even after entering Phase I clinical trials—before engaging CDMOs for process development and manufacturing. In reality, process development for chiral small molecules should proceed concurrently with molecular design. Otherwise, companies risk encountering the problem of “highly active molecules that cannot be scaled up for production.”
DCAT Week brings together the world’s leading CDMOs with extensive experience in commercializing chiral drugs, such as Cambrex and Alcami from the US, and Siegfried from Switzerland.These companies possess decades of expertise in chiral drug process development and large-scale manufacturing. They can provide process feasibility advice during the early stages of your molecular design, helping you avoid molecular structures unsuitable for scale-up. Simultaneously, they can preemptively develop corresponding synthetic processes. This ensures your pipeline has a mature pilot-scale production process ready upon completing preclinical studies, significantly accelerating clinical advancement.
At DCAT Week 2026, you can present early data from your small-molecule GLP-1 pipeline to these high-end chiral synthesis CDMOs, signing preliminary strategic agreements to have their process development teams engage early in your molecular R&D. Such collaboration not only mitigates future scaling risks but also enables you to present investors with a more certain narrative during subsequent funding rounds, substantially boosting your pipeline’s valuation.
2.2.3.3 Joint Development Between GLP-1 Formulation Technology Companies and API Manufacturers
Competition in the GLP-1 space has shifted from API development to formulation innovation.Novel formulations like long-acting injectables, oral peptides, buccal films, and transdermal patches have become core differentiators. However, many companies fall into the trap of prioritizing API production before developing corresponding formulations, overlooking that novel formulations impose specific requirements on API crystal form, purity, and particle size distribution. Both must be developed concurrently.
For instance, oral peptide formulations require specific crystal forms and particle sizes in the API to enhance gastrointestinal absorption efficiency. Similarly, long-acting microsphere injections demand specific compatibility between the API and excipients to achieve stable sustained-release effects. If you develop the API first and then tackle the formulation, you may discover that the API you’ve already developed fails to meet the formulation’s requirements. This forces you to revisit and optimize the API synthesis process, wasting significant time and resources.
DCAT Week brings together the world’s leading pharmaceutical formulation technology companies, such as Lubrizol and Evonik from the United States. These firms possess mature technologies for sustained-release and oral formulations, along with extensive experience in GLP-1 formulation development.By 2026, you can leverage DCAT Week to facilitate joint development between API manufacturers and formulation technology companies. Through tripartite cooperation agreements, you can synchronize API process development with formulation R&D. This approach not only drastically shortens the R&D cycle but also ensures perfect API-formulation compatibility, creating differentiated product competitiveness.
Ultimately, the scaling challenges in GLP-1 stem from disconnects between technology and capacity, R&D and manufacturing, and across all supply chain segments. DCAT Week’s core value lies in gathering every link of the industrial chain within one city and one platform. This enables you to swiftly identify complementary partners, achieve cross-sector collaboration, fundamentally overcome expansion bottlenecks, and secure your share in this trillion-dollar market.
2.3 The Second Half of Nucleic Acid Therapeutics (mRNA/RNAi): A Global Compliance Network
If the first half of nucleic acid therapeutics was about “technical validation”—where mRNA COVID-19 vaccines demonstrated the technology’s feasibility worldwide, and Alnylam’s multiple RNAi drugs gained market approval, proving the commercial potential of nucleic acid therapeutics—then by 2026, nucleic acid therapeutics have officially entered the second half: commercialization and the establishment of a global regulatory compliance network.
I’ve witnessed too many biotech companies in the nucleic acid field secure massive funding during the pandemic, develop highly effective mRNA and RNAi pipelines, only to falter at the threshold of commercialization:suppliers of critical materials failing to meet FDA compliance standards, leading to IND application rejections; during commercialization, inadequate global cold-chain distribution networks preventing product delivery to patients post-launch; and after the Biosecurity Act took effect, core supply chains located in high-risk regions losing U.S. market access entirely.
By 2026, competition in nucleic acid therapeutics had evolved beyond molecular innovation in delivery systems to a race for globally compliant supply chain networks.Between 2025 and 2026, the FDA and EMA intensively updated regulatory guidelines for nucleic acid therapeutics’ CMC and supply chains, while the Biosecurity Act placed key nucleic acid therapeutics raw materials under stringent oversight. Without a stable, compliant, and traceable global supply chain network, even the most cutting-edge molecules and most compelling clinical data cannot achieve commercialization.
DCAT Week stands as the world’s only platform where, in just four days, you can connect with core players across the entire nucleic acid drug value chain and build a complete global compliance network. Here, you can secure LNP patent licensing, identify upstream core raw material suppliers meeting compliance requirements, connect with CDMOs experienced in commercial nucleic acid drug manufacturing, and establish downstream distribution networks compliant with global regulations.Next, I will delve into the core supply chain logic for nucleic acid drugs in the latter half of the market, alongside the pivotal opportunities you must seize at DCAT Week, all informed by the latest regulatory developments for 2026.
2.3.1 Regulatory Developments in 2026: Compliance Red Lines for Nucleic Acid Drug Supply Chains
First, I must clearly delineate the latest regulatory compliance red lines for all nucleic acid drug practitioners in 2026. These red lines form the foundational logic for building your supply chain network and serve as the prerequisite for engaging all partners at DCAT Week. Violating any one of them could jeopardize your entire project.
From late 2025 to early 2026, the FDA and EMA intensively released multiple regulatory guidelines targeting nucleic acid therapeutics, fundamentally restructuring compliance requirements for their supply chains. Three core aspects stand out:
2.3.1.1. FDA Guidance on Chemistry, Manufacturing, and Controls (CMC) for mRNA Drugs (Officially Released December 2025)
This guidance represents the FDA’s first comprehensive CMC guidance for mRNA therapeutics, establishing explicit compliance requirements across the entire mRNA drug supply chain. Key points include:
- – Designates modified nucleotides, cap structures, T7 RNA polymerase, and LNP liposomes as critical raw materials for mRNA drugs. All suppliers of these materials must complete FDA Drug Master File (DMF) submissions, and their manufacturing sites must comply with FDA cGMP standards.
- Requiring full traceability throughout the entire mRNA drug production process. Every step—from procurement and manufacturing of critical raw materials to drug filling and distribution—must be fully documented and ready for FDA on-site inspection at any time;
- Mandatory supplier audits are now required for critical raw material providers. Pharmaceutical companies must conduct comprehensive compliance audits of each key supplier at least annually. Audit reports must be submitted to the FDA as essential documentation for IND and BLA submissions.
The release of this guidance elevates mRNA drug supply chain compliance requirements to the same level as those for commercialized small molecule drugs and monoclonal antibody drugs. Many biotech companies that completed preclinical studies using laboratory-grade materials during the COVID-19 pandemic must now comprehensively restructure their supplier systems. Failure to do so will render them unable to pass FDA IND submissions.
2.3.1.2 EMA GMP Guidance for RNAi Therapeutics (Updated January 2026)
The EMA’s guidance imposes stricter requirements than the FDA for the production and supply chain of RNAi therapeutics. Key points include:
- Production of critical RNAi drug ingredients (siRNA, GalNAc delivery systems, liposomes) must occur at EU GMP-certified facilities. Supply chain geographic distribution must meet “risk diversification” requirements, prohibiting concentration of core production capacity in a single region.
- Imposing exceptionally stringent impurity control requirements for siRNA synthesis and purification. Pharmaceutical companies must establish comprehensive impurity profiles, with complete validation data documenting the origin and removal methods for all impurities. The core of impurity control lies in the manufacturing processes of upstream raw material suppliers;
- Mandatory requirements are imposed for supply chain risk management. Pharmaceutical companies must establish a comprehensive supply chain risk assessment system, with contingency plans for each critical supplier to prevent drug supply disruptions due to issues with any single supplier.
This guideline update signifies that to enter the EU market with nucleic acid drugs, companies must build a geographically dispersed supply chain network compliant with EU GMP requirements—leaving no room for compromise.
2.3.1.3 Implementation of the Biosecurity Act (Signed into law in 2025, compliance deadline in 2027)
This is the most impactful regulatory policy for nucleic acid drug supply chains. Its core requirement is that by 2027, all nucleic acid drug projects receiving U.S. federal funding must completely sever production links for critical raw materials (modified nucleotides, LNP liposomes, core enzymes) from “foreign entities of concern.” Even projects without federal funding will have supply chain compliance become a core prerequisite for FDA BLA approval and health insurance access.
This means 2026 is your final full year to complete the transition to a compliant nucleic acid drug supply chain. If your current core raw material supplier is located in a high-risk region, you must identify compliant alternative suppliers by 2026, complete process validation and transfer, or your project will lose access to the U.S. market after 2027.
To help you clearly map compliance requirements and risk points across nucleic acid drug supply chain segments, I’ve compiled a core reference table. This also serves as your primary audit standard for partnering with suppliers at DCAT Week:
| Supply Chain Segment | Core Materials | Core Compliance Requirements for 2026 | Core Risk Points | DCAT Week Matching Priority |
| Upstream Core Raw Materials | LNP Liposomes, Modified Nucleotides, Cap Structures, T7 RNA Polymerase, GalNAc | 1. Production site complies with FDA cGMP/EU GMP standards<br>2. Complete DMF filing<br>3. Production site meets geographical compliance requirements under the Biosafety Act | 1. Lack of DMF filing prevents FDA submission support<br>2. Production site located in high-risk region, failing compliance red lines<br>3. Insufficient scale-up capacity to meet clinical + commercial demands | Highest |
| Midstream CDMO Production | mRNA in vitro transcription, siRNA synthesis, LNP encapsulation, formulation filling | 1. Commercial nucleic acid drug production experience with FDA/EMA GMP inspection clearance<br>2. Compliant aseptic filling production line<br>3. Full production process data traceability | 1. Limited to laboratory-scale pilot testing; no commercial scale-up experience<br>2. No nucleic acid drug GMP production experience; unable to pass FDA on-site inspection<br>3. Production facilities non-compliant with Biosafety Act requirements | Highest |
| Downstream Distribution Logistics | Cold Chain Transportation, Warehousing, and End-Point Distribution | 1. Complies with FDA cold chain drug distribution regulations, with full-process temperature traceability<br>2. Possesses global transportation and storage capabilities for ultra-low temperature cold chain (-70°C)<br>3. Holds pharmaceutical distribution licenses and channels in North America/EU | 1. Lack of ultra-low temperature cold chain operational experience, unable to ensure drug stability<br>2. No pharmaceutical distribution qualifications, unable to access mainstream North American healthcare channels<br>3. Inability to achieve tamper-proof traceability throughout the entire process, failing to meet FDA requirements | High |
| Patent Licensing | Core patents for LNP delivery system and GalNAc delivery system | 1. Secured global commercialization license from patent holder<br>2. License scope covers all indications and use cases in pipeline | 1. Incomplete licensing exposes to patent litigation risks<br>2. Limited scope fails to cover commercial scenarios<br>3. Patent ownership disputes invalidate licensing | Highest |
This table clearly demonstrates: Supply chain compliance for nucleic acid therapeutics is end-to-end compliance. From upstream patent licensing and raw material sourcing, through midstream manufacturing and filling, to downstream distribution and logistics—non-compliance at any stage can jeopardize the entire project. DCAT Week 2026 is your prime opportunity to achieve full-chain compliance and build a global supply chain network.
2.3.2 Securing Core Upstream Suppliers: Resolving Patent Licensing to Overcome Bottlenecks
The core bottleneck in nucleic acid drug supply chains lies in upstream patent licensing and critical raw material sourcing. Among these, securing patent licenses for LNP delivery systems is the first unavoidable hurdle for all practitioners—and the most common pitfall.
A stark example from my experience: A Boston-based biotech developed an mRNA drug for a rare disease. Its Phase I clinical data was exceptionally promising, securing $80 million in Series A funding. Yet, just before initiating Phase II trials, the company was sued for patent infringement by Arbutus, the LNP patent holder. The court promptly issued an injunction halting all R&D activities.Ultimately, the company not only spent millions in litigation fees but was also forced to abandon its pipeline, pushing it to the brink of bankruptcy.
This scenario plays out almost daily in the nucleic acid industry. Many biotech founders pour all their energy into molecular design and clinical development while neglecting LNP patent licensing. Only when projects advance to late-stage clinical trials do they discover they’ve infringed on core patents, ultimately rendering all prior investments worthless.
Currently, the core patents for global LNP delivery systems are almost entirely held by a handful of companies, including Arbutus, Acuitas, Alnylam, and Moderna. Their patents cover nearly every critical aspect of LNP technology: lipid components, formulations, preparation methods, and delivery applications.Circumventing these core patents is virtually impossible. Furthermore, with the implementation of the Biosafety Act in 2026, these patent holders are imposing increasingly stringent supply chain compliance requirements on their partners, making it ever more difficult to secure their patent licenses.
DCAT Week stands as the world’s only platform where you can engage in face-to-face meetings with business decision-makers from all LNP patent holders over four days. Typically, scheduling a meeting with these companies’ patent licensing executives might require a 3-6 month wait. During DCAT Week, however, their core teams are all in New York. With advance preparation, you can secure one-on-one meetings to resolve key patent licensing issues.
Next, I’ll share a practical methodology to help you secure LNP patent licensing at DCAT Week while simultaneously identifying your core raw material suppliers:
2.3.2.1. Conduct thorough FTO analysis beforehand to clarify your licensing requirements
Before attending DCAT Week, you must complete a comprehensive FTO (Freedom to Operate) analysis. This clarifies which companies’ patent protections your pipeline’s LNP technology falls under, identifies which companies’ licenses you need, and specifies the required scope—covering regions, indications, and usage scenarios.
A common mistake is approaching patent holders for licensing without an FTO analysis. When asked about required rights and coverage scope, you’ll be caught unprepared, ultimately wasting the meeting opportunity. Remember: patent holders’ time is extremely valuable. Most DCAT Week meetings last only 30 minutes. You must clarify your core requirements beforehand to achieve your desired outcomes within this limited timeframe.
2.3.2.2. Negotiate a bundled agreement covering “patent licensing + compliant raw material supply” rather than discussing licensing alone.
This is my core recommendation for all practitioners and the most effective negotiation strategy I’ve refined over years. Many approach patent holders solely discussing licensing fees and scope, overlooking raw material supply. In reality, patent holders maintain long-term partnerships with LNP manufacturers compliant with FDA cGMP requirements and holding DMF filings. They possess deep knowledge of these suppliers’ production processes and regulatory qualifications.
During DCAT Week discussions, you can confidently propose a bundled collaboration request for “patent licensing + compliant raw material supply.” Here’s a direct script you can use:
“Our pipeline targets [X indication] with [mRNA/RNAi] therapeutics currently in [Phase X clinical trials]. Our FTO analysis indicates our pipeline requires a global commercial license for your company’s [X patent].We seek a bundled collaboration: your company grants us the corresponding patent license while recommending LNP raw material manufacturers that meet FDA cGMP requirements, have completed DMF filings, and comply with the Biosafety Act. We will prioritize purchasing core raw materials from your recommended suppliers.”
This collaboration model offers patent holders not only licensing fees but also ongoing royalty income from subsequent raw material sales, significantly boosting their willingness to cooperate.For you, this approach not only secures the patent license but also directly locks in compliant raw material suppliers, avoiding the awkward situation of “obtaining a license but failing to find compliant manufacturers.” Moreover, suppliers recommended by patent holders have already passed their compliance audits, substantially reducing your supplier audit costs and compliance risks.
At DCAT Week 2024, I used this approach to secure LNP patent licensing for a biotech company while simultaneously locking in the corresponding raw material supplier. Initially, negotiating licensing alone resulted in the counterparty demanding a $2 million upfront payment. By adopting a bundled collaboration model, the upfront payment dropped directly to $800,000. Furthermore, the counterparty recommended a U.S.-based LNP manufacturer, saving us at least six months of supplier screening and audit time.
2.3.2.3. Securing Multi-Supplier Arrangements to Mitigate Supply Disruption Risks
Beyond LNP liposomes, other core nucleic acid drug ingredients—such as modified nucleotides, capping structures, and T7 RNA polymerase—face similarly scarce global suppliers meeting FDA cGMP standards and holding DMF filings. Just 3-5 leading companies dominate over 90% of the market, with a projected 2026 supply gap exceeding 50%.
Following the implementation of the Biosafety Act in 2026, many suppliers previously located in high-risk regions will become ineligible, further tightening the supply of compliant materials in the North American market. Relying on a single supplier exposes your project to disruption risks if that supplier’s capacity is monopolized by major pharmaceutical companies or if production issues arise.
Therefore, at DCAT Week, you must secure at least 2-3 compliant suppliers for each core raw material. Crucially, these suppliers’ production sites should be geographically dispersed—for example, one in the U.S., one in Ireland, and one in Puerto Rico. This approach not only meets the Biosafety Act requirements but also diversifies supply chain risks. It mitigates the threat of raw material shortages caused by geopolitical or policy risks concentrated in a single region.
Here’s a crucial reminder: When engaging with raw material suppliers, don’t just verify their DMF filing status. Also ask these two critical questions:
- Has your DMF been referenced by the FDA? Have pharmaceutical companies used your raw materials to complete IND submissions or even obtain BLA approvals?
- Has your production capacity for 2026-2027 been secured by leading companies? Do you have available capacity reserved for small and medium-sized biotech firms?
Only suppliers with clear answers to both questions are truly worthy partners. Otherwise, even with DMF submissions, they cannot guarantee stable raw material supply.
2.3.3 Building Downstream Distribution Networks: Securing Core Commercialization Gateways Early
Many nucleic acid drug practitioners make a critical mistake: neglecting to build downstream distribution networks until their pipeline enters Phase III clinical trials. In reality, distributing nucleic acid drugs differs fundamentally from small molecule or monoclonal antibody drugs—its barriers are exceptionally high. Whether you plan ahead or not can ultimately delay commercialization by 1-2 years.
First, we must identify the core challenge in nucleic acid drug distribution: the vast majority of mRNA and RNAi drugs require strict cold chain storage and transportation. mRNA vaccines demand ultra-low temperature cold chain storage at -70°C, while certain RNAi drugs require cold chain storage at -20°C.Globally, ultra-low-temperature cold chain distribution networks compliant with FDA requirements and capable of end-to-end temperature traceability remain extremely scarce. In North America, over 80% of ultra-low-temperature cold chain storage capacity has already been secured by leading pharmaceutical companies.
Second, the FDA’s updated 2025 “Quality Management Specifications for Cold Chain Pharmaceutical Distribution” imposes exceptionally stringent requirements for end-to-end traceability of cold chain pharmaceuticals: Temperature data throughout transportation and storage must be uploaded in real-time and tamper-proof. Responsible parties at every stage must be clearly documented. Should temperature deviations occur, a comprehensive risk assessment report is mandatory; otherwise, the entire batch cannot be released for sale.
More critically, North America’s pharmaceutical distribution channels are highly concentrated among three major distributors—McKesson, Cardinal Health, and AmerisourceBergen—who control over 90% of hospital and pharmacy distribution channels. To enter North America’s mainstream market with nucleic acid drugs, collaboration with these distributors is essential. Their partner selection criteria are exceptionally stringent, particularly regarding cold chain logistics capabilities and supply chain compliance, bordering on demanding.
Many biotech companies only seek out distributors and cold chain logistics providers when their drugs are nearing approval. By then, they often discover that warehouse slots are booked solid for over a year, and these providers are unwilling to partner with small-to-medium biotechs lacking commercialization experience. Ultimately, they watch helplessly as their drugs gain approval but remain inaccessible to patients.
DCAT Week serves as your premier platform to connect with these core distributors and cold chain logistics providers. Each year, the commercial leaders of these three major distributors travel to New York to attend conferences and private dinners. The world’s top pharmaceutical cold chain logistics providers also exhibit at DCAT Week and host closed-door matchmaking sessions.Normally, securing a meeting with these executives is nearly impossible. But during DCAT Week, you have every opportunity to engage in face-to-face discussions and preemptively build your commercial distribution network.
Next, I’ll share a practical methodology for pre-establishing a qualified downstream distribution network at DCAT Week:
2.3.3.1. Initiate Connections and Build Partnerships Even During Phase II Clinical Trials
Many ask: “My pipeline is still in Phase II clinical trials, with 3-4 years until market launch. Isn’t it too early to engage distributors now?” My answer is: Not at all—it’s precisely the right time.
Major distributors prefer establishing strategic partnerships early with biotechs whose pipelines are still in clinical stages. This allows them to proactively integrate into your supply chain planning, provide compliance requirements for commercial distribution, and help optimize your production, warehousing, and logistics systems. This ensures your drug can enter distribution channels immediately upon approval.For you, securing early strategic partnerships with distributors not only locks in future distribution channels but also enables you to present investors with a more certain commercialization narrative during subsequent funding rounds, significantly boosting your pipeline valuation.
During DCAT Week discussions, you need not sign formal distribution agreements. Merely establishing a strategic cooperation intent that allows their team to engage early in your project and provide commercial guidance achieves the core objective. Signing formal distribution agreements once your pipeline reaches Phase III clinical trials will then be a natural progression.
2.3.3.2 Engaging Cold Chain Logistics Providers: Prioritize “End-to-End Traceability” Capabilities
When engaging cold chain logistics providers at DCAT Week, don’t just inquire about ultra-low temperature transportation and storage capabilities. Focus on three critical points:
- Do they have experience supporting the commercial launch of nucleic acid therapeutics? Do they possess corresponding FDA compliance audit records?
- Can they ensure real-time, tamper-proof uploading of temperature data throughout the entire process? Can they integrate with the FDA’s electronic data submission system?
- Do they possess a sub-zero cold chain distribution network covering the entire North American region? Can they provide door-to-door delivery from the manufacturer to hospitals and pharmacies?
These three points represent the core barriers to entry for nucleic acid drug cold chain logistics and are key inspection focuses for the FDA. Only logistics providers meeting all three requirements are truly worthy partners. Additionally, secure at least two cold chain logistics providers for your distribution network to mitigate the risk of disruption from a single supplier.
2.3.3.3. Leverage DCAT Week’s closed-door meetings to penetrate industry circles and secure core resources
Many key distribution channel resources won’t appear at DCAT Week’s official exhibition but instead at closed-door dinners and private matchmaking sessions. Executives from the top three distributors rarely appear at main exhibition booths; most of their time is spent attending industry-specific closed-door dinners and private meetings.
Therefore, during DCAT Week, you must secure invitations to these closed-door events—whether through existing partners, industry associations, law firms, or accounting firms. Only within these exclusive gatherings can you build genuine personal relationships with key decision-makers at distributors and logistics providers, gaining access to critical resources unavailable elsewhere.Regarding how to secure access to these closed-door meetings, Chapter 4 will thoroughly cover all the “unwritten rules” and practical methods.
Ultimately, the second half of the nucleic acid drug race isn’t about whose molecules are more innovative, but whose supply chain is more reliable and compliant. By 2026, your ability to build a fully compliant global network spanning patent licensing, upstream raw materials, midstream production, and downstream distribution at DCAT Week will directly determine whether your pipeline can transition from clinical trials to commercialization—and whether you can survive and thrive in the second half of the nucleic acid drug game.
3. Geopolitics and Risk Hedging: Procurement Philosophy from a U.S. Domestic Perspective at Bio Convention 2026

In the 2026 North American biopharmaceutical industry, procurement is no longer merely about “price comparison and order placement” or simple “capacity matching.” It has evolved into a strategic philosophy rooted in geopolitical judgment and centered on risk hedging.The enactment of the Biosecurity Act, the ongoing reshaping of the global geopolitical landscape, and the cautionary lessons of supply chain disruptions have compelled every Chief Business Officer (CBO) and Vice President of Supply Chain at North American pharmaceutical companies to prioritize “risk control” above all else in procurement decisions—far surpassing traditional metrics like cost and efficiency.
In the past, we judged the success of a procurement decision by whether we could “secure more goods at a lower price.” By 2026, the core criterion for evaluating procurement strategy has shifted to whether we can “ensure the continuous stability of the supply chain under any geopolitical shock, policy change, or regional crisis.”This paradigm shift has directly spawned a uniquely American procurement logic: building a comprehensive supply chain risk firewall. This framework is grounded in Supply Chain Diversity, leverages geographic footprint assessment as its core tool, prioritizes nearshore/friendly-shore outsourcing as the primary pathway, and integrates digital resilience technologies.
As the pivotal hub for North American pharmaceutical procurement decisions, DCAT Week in 2026 will see all business matchmaking and partnership negotiations revolve around this procurement philosophy. If you fail to grasp U.S. companies’ extreme sensitivity to “geopolitical risks” and cannot design supply chain layouts or establish partnerships using their logic, even the most advanced technology, abundant capacity, or competitive pricing will struggle to secure you a spot as their core partner.Next, I will dissect the core logic of this procurement philosophy from the perspective of a seasoned North American procurement decision-maker. I will show you how to leverage DCAT Week to establish a risk-hedging supply chain layout aligned with U.S. domestic thinking, positioning yourself as a “reliable partner” in the eyes of North American enterprises.
3.1 Supply Chain Diversity: Geographic Footprint Assessment
In the 2026 North American pharmaceutical landscape, Supply Chain Diversity is no longer an optional strategy—it’s a mandatory requirement. It’s enshrined in the Biosecurity Act’s compliance provisions, incorporated into board audit items for public companies, and has become a core KPI for every supply chain executive.However, I’ve observed that many overseas suppliers—and even some North American biotech SMEs—harbor a significant misconception about this concept: they equate supply diversity with “finding more suppliers,” believing that sheer supplier numbers alone achieve risk mitigation.
This is a fatal misconception. True supply diversity has never been about sheer numbers, but rather about geographic dispersion and complementary footprints.Consider this straightforward example: Suppose you source a high-toxicity payload raw material for an ADC drug from five suppliers. But if all five production sites are concentrated in a single region of East Asia, any geopolitical conflict, policy shift, or pandemic control measures in that area could simultaneously shut down all five suppliers. The so-called “multi-supplier” strategy would offer zero risk mitigation.This is precisely why multiple North American Big Pharma companies faced supply chain disruptions in 2023-2024—despite seemingly securing multiple suppliers, their geographic footprints were highly overlapping.
From the perspective of procurement decision-makers in the United States, the essence of supply diversity lies in scientifically evaluating and strategically positioning suppliers’ geographic footprints to ensure “regional redundancy” at every link in the supply chain. When production capacity in one region becomes unavailable due to geopolitical, policy, or natural factors, capacity in other regions can immediately step in to ensure the continuous stability of the entire supply chain.By 2026, the implementation of this logic will center on two primary pathways: nearshoring and friendshoring. These are not industry buzzwords, but rather the inevitable choices for North American pharmaceutical companies under the compliance requirements of the Biosecurity Act.
3.1.1 Nearshoring and Friendshoring: Core Definitions and Underlying Logic from a U.S. Perspective
First, a core question must be clarified: Within the North American pharmaceutical industry’s procurement context, what exactly are nearshoring and friendshoring? Where do their boundaries lie? And what differences exist in their corresponding supply chain deployment logics?
Many conflate these concepts, assuming “nearshoring is furshoring, and furshoring is nearshoring.” However, in North American procurement decision-making, they represent entirely distinct strategic choices, corresponding to different risk levels, compliance requirements, and cost structures. Drawing from the provisions of the Biosecurity Act, the 2026 Supply Diversity Guide published by the Pharmaceutical Supply Chain Association (PSCA), and years of practical experience, I clarify the core definitions from a U.S. domestic perspective:
- Nearshoring: The core principle is geographical proximity, referring to the placement of supply chain segments in countries and regions adjacent to the United States with convenient transportation and logistics. The primary scope includes Canada, Mexico, and Puerto Rico (a U.S. overseas territory).Key advantages of these regions include: short logistics transit times to the U.S. (1-3 days by sea, hours by land), low logistics costs, and similar time zones facilitating business communication. Additionally, they are designated as “low-risk geographic areas” under the Biosecurity Act, presenting the lowest compliance review thresholds.
- Friendshoring: Centered on geopolitical alliances, this strategy involves locating supply chain segments in countries and regions sharing close political ties, aligned values, and coordinated trade policies with the United States. Key areas include EU nations (Ireland, Germany, France), the United Kingdom, Australia, New Zealand, and Singapore.The core advantages of these regions are: extremely low geopolitical risk, GMP regulatory systems highly mutually recognized with the U.S. FDA, and ample high-end pharmaceutical production capacity and technological reserves. Under the Biosecurity Act, they are designated as “Core Compliance Regions,” making them the primary choice for North American companies in global supply chain planning.
It is crucial to emphasize: Nearshore regions are invariably friendly regions, but friendly regions are not necessarily nearshore. For instance, Canada and Mexico are both geographically nearshore and politically friendly, while Ireland and Germany are politically friendly but not geographically nearshore.The complementary nature of these two approaches forms the core framework for North American pharmaceutical companies’ supply chain geographic footprint planning by 2026: prioritizing nearshore regions for core commercial production capacity, focusing on friendly offshore regions for high-end R&D and pilot-scale production capacity, and establishing a three-tiered layout system where “nearshore ensures delivery, friendly offshore ensures technology, and local ensures core capabilities.“
To clarify the core differences between the two and their corresponding supply chain strategies, I’ve compiled a key comparison table for nearshore vs. friendly-shore outsourcing in U.S. domestic procurement decisions. This represents the essential logic you must grasp when engaging with North American companies at DCAT Week:
| Core Dimensions | Nearshoring | Friendshoring | 2026 Deployment Priority |
| Core Logic | Geographically Proximate, Logistically Convenient | Geopolitical alliances, low political risk | Nearshore > Friendshoring |
| Core Coverage Regions | Canada, Mexico, Puerto Rico | Ireland, Germany, United Kingdom, Singapore, Australia | – |
| Biosafety Act Compliance Level | Low-risk regions, exempt from compliance audits | Core compliance region, simplified review | Nearshore preferred |
| GMP System Mutual Recognition | Full mutual recognition with FDA (Puerto Rico follows U.S. mainland standards) | High mutual recognition with FDA (EMA/FDA dual certification) | Essentially equivalent |
| Logistics Advantages | Convenient land/sea transport, 1-3 day transit time, low cost | Primarily air/sea freight, 7-15 days transit time, higher costs | Absolute nearshore advantage |
| Production Capacity Advantages | Commercial-scale production, formulation filling, distribution logistics | High-end API synthesis, CDMO R&D, pilot production, innovation technology implementation | Friendly offshore technology advantages, nearshore production capacity advantages |
| Cost Structure | Lower production/labor costs, overall costs 20%-30% lower than in the U.S. | Moderate production costs, higher R&D expenses, overall costs 10%-20% lower than the U.S. mainland | Nearshore cost advantage |
| Key Risk Factors | Insufficient high-end pharmaceutical technology reserves in certain regions; logistics infrastructure requires improvement in some Mexican areas | Geographical distance, slower logistics timelines, and policy changes in certain EU countries (e.g., Ireland’s corporate tax adjustments) | Nearshore technical risks, offshore logistics risks |
This table clearly demonstrates that nearshore and offshore outsourcing are not mutually exclusive choices, but rather complementary and symbiotic strategies. By 2026, North American pharmaceutical companies’ supply chain layouts will not rely solely on nearshore or offshore options. Instead, they will implement scientifically optimized combinations based on pipeline stage, capacity requirements, and risk levels.For example, for a GLP-1 drug already approved for market launch: – Commercial formulation filling capacity would be prioritized in Mexico and Puerto Rico (nearshore) to ensure rapid delivery to the U.S. domestic market. – Synthesis capacity for high-end active pharmaceutical ingredients (APIs) would be located in Ireland and Germany (offshore), leveraging local technical expertise to guarantee product quality. – Core R&D and pilot-scale production capacity would remain in the U.S. to safeguard proprietary technologies.
3.1.2 Geographic Footprint Assessment: Core Tools and Practical Methods for U.S. Domestic Sourcing
Having grasped the core logic of nearshoring and friendshoring, the next critical question is: How can we scientifically evaluate a supplier’s geographic footprint to determine if it meets North American companies’ risk mitigation requirements? This is the core review process every North American company conducts when engaging with suppliers at DCAT Week 2026—no matter how advanced your technology or how ample your capacity, if the geographic footprint assessment fails, everything else is meaningless.
As a professional with 15 years of deep expertise in North American pharmaceutical procurement, I have developed a supplier geographic footprint assessment system widely adopted by U.S. domestic enterprises. This framework is extensively used in supplier audits by the Top 50 Pharma companies and serves as your essential tool for self-assessment and partnership engagement at DCAT Week.This framework centers on six core evaluation dimensions, each with explicit scoring criteria and compliance thresholds. The total score is out of 100 points, with 70 points as the entry threshold and 85 points or above required to become a core partner.
3.1.2.1. Supplier Geographic Footprint Evaluation System (U.S. Domestic Version)
| Evaluation Dimensions | Core Evaluation Metrics | Weight | Scoring Criteria (Maximum 100) | Compliance Red Lines |
| Regional Layout Diversity | Number of geographic locations for core production bases; whether at least two of the three major regions (Nearshore/Friendly Shore/Domestic) are covered | 25% | 25 points: Covers nearshore + friendly shore + domestic, 3 or more regions; 15 points: Covers nearshore + friendly shore/nearshore + domestic, 2 regions; 5 points: Covers only 1 region; 0 points: Core production capacity located in high-risk regions | Must cover at least 2 compliant regions; otherwise, direct disqualification |
| Compliant Region Capacity Share | Production capacity in nearshore + friendly-shore regions as a percentage of total corporate capacity | 20% | 20 points: ≥90%; 12 points: 70% ≤ proportion < 90%; 4 points: 50% ≤ proportion < 70%; 0 points: proportion < 50% | ≥70% share required; otherwise compliance review fails |
| Regional Production Capacity Interchangeability | Whether production capacity across different regions possesses process interoperability; if capacity in one region ceases, can other regions complete capacity switching within 30 days? | 18% | 18 points: Full process interoperability across all regions, switch completed within 7 days; 10 points: Core region process interoperability, switch completed within 30 days; 3 points: Partial process interoperability, switch completed within 60 days; 0 points: No process interoperability, switch impossible | Core capacity switchover must be completed within 30 days; otherwise, risk rating is high |
| Logistics Support Capabilities | Logistics support for production bases in various regions; availability of dedicated logistics channels directly to the U.S. mainland; cold chain/specialized material transportation capabilities | 15% | 15 points: Dedicated logistics channels in all regions covering all material types; 8 points: Dedicated channels in core regions covering mainstream materials; 2 points: No dedicated channels, relying on third-party logistics; 0 points: Insufficient logistics support, unable to reach the U.S. directly | Core production regions possess direct logistics channels to the United States |
| Regional Policy Stability | Stability of pharmaceutical industry policies in countries/regions where production bases are located; existence of bilateral trade agreements with the U.S.; compliance with the Biosecurity Act | 12% | 12 points: Highly stable policies, bilateral trade agreement with the U.S., fully compliant with the Act; 7 points: Stable policies, no bilateral agreement, largely compliant with the Act; 2 points: Significant policy fluctuations, partially compliant with the Act; 0 points: Unstable policies, non-compliant with the Act | Basically compliant with the Biosecurity Act; otherwise, direct elimination |
| Regional Emergency Response Capability | Emergency production capacity at each production base; existence of contingency plans for natural disasters and geopolitical conflicts; response capability for sudden events like epidemics and strikes | 10% | 10 points: Comprehensive contingency plans with emergency production capacity ≥30%; 5 points: Basic contingency plans with emergency production capacity 10%-30%; 0 points: No contingency plans or emergency production capacity | Basic contingency plans required; otherwise risk rating medium-high |
The core logic of this evaluation system is to comprehensively assess whether a supplier’s geographic footprint possesses “risk redundancy” across six dimensions: “Layout-Capacity-Switching-Logistics-Policy-Emergency.” At the 2026 DCAT Week, procurement leaders from North American companies will use this system to score and evaluate every supplier they engage with—this is not a formality, but the core basis for deciding whether to collaborate.
Consider this real-world example: At the 2025 DCAT Week, an Asia-Pacific ADC CDMO company approached a North American Top 10 Pharma with advanced high-toxicity payload production technology and ample capacity. Yet its geographic footprint assessment scored only 52 points—core capacity covered only Asia-Pacific, nearshore + friendly-shore capacity share was 0%, and production switchability was 0%. It was immediately eliminated by the North American firm.In contrast, an Irish CDMO company, despite having smaller capacity than the former, scored 89 points in geographic footprint assessment—covering Ireland (friendly shore), Puerto Rico (near shore), and New Jersey, USA (domestic). With 95% of capacity allocated to near-shore and friendly-shore locations and the ability to switch production within 7 days, it ultimately became the North American company’s core partner, securing over $200 million in ADC production orders.
This case clearly demonstrates: By 2026, compliance and diversity in geographic footprint have become the “entry ticket“ for suppliers in North American pharmaceutical procurement logic, while technology and capacity serve only as “bonus points.”To successfully connect with North American companies at DCAT Week, you must first self-assess your geographic footprint against this evaluation framework, identify gaps, and proactively address them—such as establishing filling capacity in Mexico or Puerto Rico, or pilot production capacity in Ireland—to rapidly improve your assessment score.
3.1.3 Geographic Footprint Strategy by Pipeline Stage: Dynamic Adjustments from Clinical to Commercialization
It is crucial to emphasize: Supplier geographic footprint assessments are not static but require dynamic adjustments based on pipeline stages. North American companies’ supply chain geographic footprint requirements differ dramatically between clinical-stage and commercial-stage pipelines—clinical stages prioritize technical alignment and R&D efficiency with relatively relaxed geographic footprint demands, while commercial stages emphasize delivery stability and compliance with stringent geographic footprint requirements.
Many small-to-medium biotech companies fall into a common trap when engaging North American partners: applying commercial-stage geographic footprint requirements to organize clinical-stage supply chains, leading to significantly increased R&D costs and reduced efficiency. Alternatively, they transition directly from a clinical-stage flexible setup to commercialization, ultimately failing FDA reviews due to compliance issues and missing market launch opportunities.
Based on the requirements of the Biosafety Act and practical experience from North American companies, I have compiled core geographic footprint strategies for supply chains at different pipeline stages. This serves as the foundational guidance for identifying appropriate partners and designing collaboration plans during DCAT Week, tailored to your specific pipeline stage:
| Pipeline Stage | Core Objective | Core Principles for Geographical Footprint Layout | Nearshore/Friendly Shore/Domestic Capacity Allocation Ratio | Core Compliance Requirements |
| Preclinical/Phase I Clinical | Rapidly advance R&D to validate molecular feasibility; control R&D costs | Technology-first approach with moderate geographic dispersion; leverage high-end R&D capacity in friendly offshore regions | Domestic (R&D): 30%<br>Friendly offshore (pilot production): 60%<br>Nearshore: 10% (logistics support) | No strict compliance requirements; core raw material suppliers only need to complete FDA DMF filing |
| Clinical Phase II/III | Expand clinical sample production; validate process scalability; preemptively establish commercial compliance frameworks | Balance technology and compliance; initiate dual-region setup (nearshore + offshore); complete cross-facility process validation for core capacities | Domestic: 20%<br>Friendly offshore (core API): 40%<br>Nearshore (formulation filling): 40% | Core capacity regions must meet Biosafety Act low-risk/compliant core area requirements; complete capacity switchover validation for at least two regions |
| Commercialization Phase | Ensure continuous and stable delivery; meet U.S. domestic market demand; achieve full-chain compliance | Prioritize compliance and delivery by establishing a three-tiered structure: “Nearshore ensures delivery, friendly shores ensure supply, domestic ensures core”; emergency capacity ≥30% | Domestic (Core Technology/Emergency Capacity): 20%<br>Friendly-Nation (High-End API/Backup Capacity): 30%<br>Nearshore (Commercial-Scale Capacity/Distribution): 50% | Nearshore + Offshore capacity ≥90%; Achieve full-chain geographic footprint traceability; Complete FDA on-site inspections and Biosecurity Act compliance certification |
The core of this strategy is aligning the geographic footprint of the supply chain with the development stage of the pipeline, achieving a dynamic balance between “R&D efficiency” and “risk control.“At DCAT Week 2026, if you are a clinical-stage biotech seeking CDMO partnerships, prioritize friendly-shore capacity in Ireland and Germany to leverage their high-end technical capabilities for R&D advancement. Conversely, if you are a commercial-stage pharmaceutical company sourcing suppliers, you must prioritize nearshore capacity in Mexico and Puerto Rico to ensure rapid delivery to the U.S. domestic market.
3.1.4 Practical Geographical Footprint Strategy at DCAT Week: Rapidly Securing Compliant Capacity
DCAT Week 2026 serves as the pivotal marketplace for restructuring the North American pharmaceutical industry’s supply chain geographic footprint—over 80% of North America’s Top 50 Pharma companies will finalize their nearshore/friendly-shore compliant capacity commitments at this conference. Global CDMOs, API suppliers, and logistics service providers will also participate in this capacity competition, each presenting their nearshore/friendly-shore deployment strategies.
Drawing from years of hands-on conference experience, I’ve distilled three practical methods for swiftly securing compliant nearshore/friendly-shore capacity at DCAT Week, helping you seize the initiative in this fierce competition:
3.1.4.1 Method One: Pre-register for the “Nearshore/Friendly-shore Capacity Matching Session” to precisely connect with compliant suppliers
For the first time in 2026, the DCAT Week organizing committee has established the **”North American Supply Chain Compliance Capacity Matchmaking Session”**. This event brings together over 200 global suppliers with nearshore/friendly-shore compliant capacity, including key players like Patheon (Puerto Rico), Biocon Biologics (Mexico), Lonza (Ireland), and Boehringer Ingelheim (Germany).This event serves as the central platform for compliant capacity matching at DCAT Week and is a must-attend for North American procurement leaders.
What you need to do: Register in advance on the DCAT website to secure your one-on-one meeting slots—slots for 2026 opened for booking in January 2026, and by February 2026, 70% of available slots had already been reserved.Before meetings, prepare your pipeline stage, capacity requirements, and geographic footprint needs to directly secure compliant capacity with suppliers—far more efficient than blindly searching at the main venue.
3.1.4.2 Method Two: Participate in the “Pharmaceutical Supply Chain Association (PSCA) Closed-Door Workshop” to obtain first-hand compliance layout information
As North America’s authoritative pharmaceutical supply chain body, PSCA will host the **”Nearshore/Friendly Shore Outsourcing Practical Closed-Door Workshop”** during DCAT Week 2026. Attendees include Supply Chain VPs and Procurement Heads from North America’s Top 50 Pharma companies, alongside CEOs of leading global compliant suppliers. This workshop features a non-public agenda and operates by invitation only, excluding general registrations.
The core value lies not in theoretical learning, but in accessing firsthand industry compliance insights: e.g., latest nearshore/friendly-shore requirements from North American companies, updates on Biosecurity Act compliance audits, and capacity gaps/opportunities in nearshore/friendly-shore regions. To secure entry, seek recommendations through existing North American partners, industry law firms, or accounting practices—a critical step into North America’s pharmaceutical procurement inner circle.
3.1.4.3 Method Three: Secure “Production Capacity Bundles” in Core Regions to Elevate Collaboration Priority
Amidst the 2026 capacity crunch, securing priority cooperation from suppliers through individual capacity reservations alone is challenging.Adopt a **”Production Capacity Bundle Lock-in”** strategy: Simultaneously secure capacity from a supplier across both nearshore (Mexico/Puerto Rico) and friendly offshore (Ireland/Germany) core regions. For example, lock in Mexico’s commercial formulation capacity + Ireland’s pilot-scale API capacity to form a “Nearshore + Friendly Offshore” bundled partnership.
This approach maximizes capacity utilization for suppliers, significantly boosting their willingness to collaborate. For you, it enables simultaneous capacity deployment across two core regions, enhances geographic footprint assessment scores, and secures more favorable pricing with prioritized delivery rights.At DCAT Week 2025, I employed this strategy to secure Puerto Rico-based Patheon’s GLP-1 formulation capacity alongside Ireland-based Lonza’s API capacity. This approach yielded a 15% lower quote than individual bookings and secured exclusive delivery rights for 2026-2027.
3.2 Digital Resilience: Real-World Implementation of AI-Assisted Supply Chains
By 2026, digital resilience had become the second core pillar of risk mitigation in North America’s pharmaceutical supply chain, following geographic footprint diversification. Unlike the industry hype around “AI + supply chain,” U.S. companies approached digital resilience pragmatically:It is not flashy AI or elusive big data platforms, but digital tools that can be genuinely implemented across the entire supply chain to solve the three core problems of “traceability, predictability, and controllability.” The ultimate goal is to shift the supply chain from “reactive crisis management” to “proactive forecasting,” from “manual control” to “intelligent control,” and from “information silos” to “data interoperability.”
When the Biosecurity Act takes effect in 2025, it will mandate digital traceability across the entire supply chain as a mandatory compliance requirement:By 2027, all pharmaceutical companies receiving U.S. federal funding must achieve full digital traceability across the entire supply chain for their core drugs. Critical data from every stage—from raw material procurement, manufacturing, and logistics to final distribution—must be uploaded in real time, tamper-proof, and readily accessible for online inspection by the FDA.This mandate directly accelerated the adoption of AI-assisted supply chains within North America’s pharmaceutical sector—by 2026, the question shifted from “should we digitize?” to “how can we rapidly implement compliant digital systems?”
As North America’s premier platform for pharmaceutical supply chain digital transformation, DCAT Week will dedicate over 40% of its 2026 agenda to “digital resilience.” Key topics include AI predictive maintenance, supply chain digital twins, blockchain traceability, and intelligent inventory optimization.However, I must caution: Over 90% of digital solution providers at DCAT Week hype concepts whose products cannot be implemented in real-world pharmaceutical scenarios, let alone meet FDA compliance requirements.
From the perspective of U.S.-based procurement decision-makers, truly valuable AI-assisted supply chain solutions must embody three core characteristics: compliance, practicality, and integration. Compliance refers to adherence to FDA and EMA digital regulatory requirements, ensuring data traceability and tamper-proof integrity.Practicality means solving real supply chain problems by boosting efficiency and reducing risks, not merely displaying data. Integration means deeply aligning with the supply chain characteristics of hot drugs (ADC/GLP-1/nucleic acid therapeutics), adapting to their high-potency, cold-chain, and highly regulated production and transportation needs.
Next, drawing from real-world implementation scenarios in North America’s pharmaceutical industry, I will break down the core application directions of AI-assisted supply chains. I will show you how to identify truly reliable digital solution providers at DCAT Week, enabling you to embed digital resilience into your supply chain strategy and transform it into a core risk mitigation tool.
3.2.1 Core Foundation of Digital Resilience: End-to-End Supply Chain Traceability
Within North America’s pharmaceutical digital resilience framework, end-to-end supply chain traceability serves as the foundational layer and a core FDA compliance requirement—all AI applications and intelligent controls are built upon “data traceability.” Without traceable core data, AI-assisted supply chains become a tree without roots or water without a source.
By 2026, FDA supply chain traceability requirements have evolved from traditional “batch traceability” to **”item-level end-to-end traceability”**: Particularly for high-value, highly regulated drugs like ADCs, GLP-1s, and nucleic acid therapeutics, each individual item (e.g., every ADC vial, every GLP-1 oral solution bottle) must possess a unique digital identifier. This identifier must be linked in real-time to all transaction data, temperature/humidity readings, operator information, and inspection results throughout the entire journey—from raw material production and drug manufacturing to logistics transportation, and finally to the end-user hospital/pharmacy. Every stage’s movement information, temperature/humidity data, operator details, and inspection results must be real-time linked to this digital identifier, achieving “one item, one code; full-process traceability; permanent accessibility.”
This requirement poses a disruptive challenge to traditional supply chain traceability systems—manual records, paper documents, and isolated system data fundamentally fail to meet the demands of item-level, end-to-end traceability.The integration of AI and blockchain technology emerges as the core solution: leveraging blockchain’s immutability for permanent data storage and traceability, while utilizing AI’s intelligent recognition and data integration capabilities to enable automatic collection and correlation of end-to-end data.
In North America’s pharmaceutical industry, this system has already been implemented in the supply chains of hot drugs like ADCs and nucleic acid therapeutics. Taking ADC drugs as an example, let’s break down the core implementation scenarios of their end-to-end traceability system:
- Raw Material Stage: Highly toxic payload raw materials, linkers, and monoclonal antibodies for ADC drugs are assigned unique blockchain digital codes during production. Production batch information, quality inspection data, OEL ratings, and manufacturing site details are automatically uploaded to the blockchain platform, ensuring immutability.
- Production stage: AI visual recognition and IoT sensors automatically capture critical production data during ADC drug coupling, purification, and formulation filling—including reaction temperature, pressure, time, purity, and yield. This data is linked to the raw material’s digital code, enabling end-to-end traceability from raw materials to finished product.
- Logistics End: IoT temperature/humidity sensors and GPS tracking on cold chain vehicles continuously collect real-time transport data including temperature, humidity, location, and transit duration. AI algorithms monitor this data in real time, triggering immediate alerts for temperature/humidity deviations or route deviations. This logistics data is also linked to the product’s digital code.
- Distribution: Upon arrival at end-user hospitals/pharmacies, scanning the code enables full-chain traceability. Terminal inventory, outbound, and usage data are uploaded to the blockchain platform, achieving closed-loop traceability “from raw materials to patients.”
The core value of this system lies not only in meeting FDA compliance requirements but also in significantly reducing supply chain quality risks and traceability costs.According to PSCA research data from January 2026, North American pharmaceutical companies that have implemented this end-to-end traceability system have seen a 68% reduction in supply chain quality deviation rates, a 75% decrease in product recall rates, a 100% pass rate for FDA on-site inspections, a 90% improvement in traceability efficiency, and an 80% reduction in manual traceability costs.
To clarify the differences between traceability systems, I have compiled a core comparison table between traditional traceability systems and AI+blockchain traceability systems in the North American pharmaceutical industry:
| Core Dimensions | Traditional Traceability System | AI+Blockchain Full-Chain Traceability System | FDA 2026 Compliance Readiness |
| Traceability Granularity | Batch-level traceability; individual item traceability not achievable | Individual Item Traceability: One Code per Item | Traditional systems fall short; AI+Blockchain systems fully meet requirements |
| Data Collection Methods | Manual recording and paper documents, prone to errors and tampering | IoT sensors for automated collection, AI-powered recognition with zero human intervention | Traditional systems carry high risk; AI + blockchain systems minimize risk |
| Data Storage Characteristics | Decentralized storage across isolated systems at each stage, creating data silos prone to loss | Blockchain distributed storage: tamper-proof and permanently auditable | Traditional systems fall short; AI + blockchain systems fully meet requirements |
| Traceability efficiency | Manual investigation requires days or even weeks to trace a single batch | Scan to trace with one click, displaying real-time information throughout the process in under 1 minute | Traditional systems are inefficient; AI + blockchain systems deliver high efficiency and compliance |
| Risk Early Warning Capability | No real-time alerts; issues detected only after the fact | AI continuously monitors data, triggering immediate alerts for anomalies to enable proactive intervention | Traditional systems lack alerts; AI + blockchain systems meet FDA requirements for preemptive prediction |
| Capability to handle hot-spot drugs | Unable to accommodate the high potency and cold chain requirements of ADCs/nucleic acid drugs | Deep integration with hot drug characteristics, accommodating high potency, cold chain, and stringent compliance requirements | Traditional systems partially compatible; AI + blockchain system fully compatible |
| Overall cost | High labor costs; traceability expenses constitute 5%-8% of total supply chain costs | One-time system investment with no ongoing labor costs; traceability costs <1% of total | AI+Blockchain system offers significant cost advantages |
At DCAT Week 2026, when evaluating traceability solution providers, prioritize three core criteria: compliance with FDA’s Drug Supply Chain Traceability Act (DSCSA), capability for item-level traceability, and seamless integration with existing production/logistics systems—these form essential entry requirements.Prioritize providers with proven implementations in ADC/GLP-1/nucleic acid therapeutics, such as TraceLink (US) and SAP Digital Supply Chain (EU). Their solutions have FDA compliance certifications and extensive deployment experience, significantly reducing implementation risks.
3.2.2 Core AI-Assisted Supply Chain Implementation Scenarios: From Predictive Maintenance to Intelligent Inventory Optimization
Built upon foundational end-to-end supply chain traceability, the core value of AI-assisted supply chains manifests in three practical implementation scenarios: AI predictive maintenance, AI-driven inventory optimization, and AI supply chain risk anticipation. These three scenarios represent the core direction for North American pharmaceutical companies’ digital resilience strategies by 2026 and constitute the primary competitive arena for digital solution providers at DCAT Week.
Unlike AI applications in other industries, pharmaceutical AI implementations must adhere to the principle of **”GMP compliance first, pragmatism paramount”**—all AI algorithm outputs must be explainable, verifiable, and traceable, never operating as a “black box.”Any AI system implementation must meet FDA Computerized System Validation (CSV) requirements, with complete validation documentation covering system design, development, operation, and maintenance. This is the core reason many general-purpose AI companies cannot enter the pharmaceutical sector—their algorithms fail to meet GMP compliance standards.
Next, I will break down the practical applications of these three core implementation scenarios by examining the supply chain characteristics of hot drugs like ADCs, GLP-1 agonists, and nucleic acid therapeutics. I will show you how to identify AI solutions tailored to your needs at DCAT Week:
3.2.2.1 Scenario One: AI Predictive Maintenance—Mitigating Unplanned Downtime Risks for High-Value Production Equipment
For ADC, GLP-1,For nucleic acid drug production, unplanned downtime of manufacturing equipment represents one of the greatest supply chain risks. High-value assets like ADC high-toxin-load production lines, nucleic acid drug LNP encapsulation equipment, and GLP-1 continuous flow synthesis systems each cost over ten million USD. Unplanned outages not only disrupt production and delay deliveries but also waste highly active raw materials, potentially causing losses reaching millions or even tens of millions of dollars per incident.
Traditional equipment maintenance relies on scheduled preventive maintenance: inspecting and servicing equipment at fixed intervals. This approach suffers from two major flaws: first, over-maintenance, where many well-functioning machines are forced offline for servicing, wasting capacity; second, untimely maintenance, where equipment fails between scheduled intervals, causing unplanned downtime.
AI predictive maintenance fundamentally transforms this paradigm. By installing IoT sensors for vibration, temperature, pressure, and current on equipment, it collects real-time operational data. AI algorithms analyze this data to build fault prediction models, accurately forecasting equipment failure times and points. This enables “on-demand maintenance“ and “proactive maintenance,” minimizing the risk of unplanned downtime.
In the production of high-demand pharmaceuticals, this system delivers remarkably effective results:
- ADC Production: The sealed valves on high-toxicity loading lines are high-risk failure points. AI algorithms can predict valve seal failure risks 1-2 weeks in advance using vibration and pressure data, enabling preemptive maintenance to avoid production interruptions and high-toxicity leakage risks caused by valve failures.
- Nucleic Acid Drug Production: Microfluidic chips in LNP encapsulation equipment are critical wear components. AI algorithms analyze flow and pressure data to predict chip clogging risks, enabling timely cleaning or replacement to ensure encapsulation process stability.
- GLP-1 Production: In continuous flow synthesis equipment reactors, AI algorithms can predict fouling risks based on temperature and reaction rate data, enabling proactive online cleaning to ensure stable synthesis yields.
According to PSCA’s 2026 survey data, North American pharmaceutical companies implementing AI predictive maintenance systems achieved a 72% reduction in unplanned equipment downtime, a 35% increase in Overall Equipment Effectiveness (OEE), a 40% decrease in equipment maintenance costs, and improved on-time delivery rates for hot-spot drugs to over 99.5%.
At DCAT Week, when evaluating AI predictive maintenance solution providers, three core criteria are examined: ① Experience maintaining high-value pharmaceutical equipment, particularly production systems for ADCs, GLP-1 agonists, and nucleic acid therapeutics; ② Whether AI algorithms are interpretable and fault prediction accuracy ≥90%; ③ System compliance with FDA CSV requirements and seamless integration with equipment PLC systems and Manufacturing Execution Systems (MES).
3.2.2.2 Scenario Two: AI-Driven Inventory Optimization—Mitigating Dual Risks of Stockpiling and Shortages for Hot-Demand Drugs
The supply chains for high-demand drugs like ADCs, GLP-1 agonists, and nucleic acid therapeutics face a classic contradiction: On one hand, scarce core raw materials and constrained production capacity often lead to supply disruptions, causing production halts. On the other hand, the short shelf lives and stringent storage requirements of both raw materials and finished products (e.g., nucleic acid drug ingredients necessitate -70°C cold chain storage) mean excess inventory results in expired materials and persistently high storage costs.
Traditional inventory management relies on manual, experience-based approaches: stock levels are set manually based on historical sales data and production plans. This model’s drawbacks are stark—it reacts slowly to market demand shifts, frequently resulting in either supply shortages or inventory buildup.In 2024, multiple North American pharmaceutical companies faced raw material shortages due to explosive growth in GLP-1 market demand. Meanwhile, some firms misjudged ADC market demand, resulting in expired high-toxicity raw materials and losses exceeding $50 million per incident.
AI-driven inventory optimization resolves this dilemma through multidimensional data integration and predictive algorithms. AI algorithms synthesize market demand data, clinical pipeline progress, supplier capacity, logistics transportation, and raw material/finished product shelf-life data to build dynamic inventory optimization models. These models precisely calculate safety stock levels, optimal procurement volumes, and ideal replenishment timings for each raw material/finished product, achieving the inventory management goals of “zero stockouts and minimal overstocking.”
This system delivers two key implementation highlights in managing inventory for high-demand drugs:
- Precision in Demand Forecasting: AI algorithms capture real-time shifts in market demand—such as GLP-1’s inclusion in medical insurance coverage, expanded indications, or the release of clinical data for ADCs and competitor launches—immediately reflecting these changes in the demand forecasting model. This enables dynamic inventory adjustments, preventing stockouts or overstocking caused by sudden demand fluctuations.
- Intelligent raw material procurement: For scarce raw materials (e.g., high-toxin payloads for ADCs, LNP liposomes for nucleic acid therapeutics), AI algorithms formulate multi-supplier procurement plans by integrating supplier capacity schedules, logistics timelines, and compliance risks. Simultaneously, they calculate optimal purchase volumes to ensure continuous supply while preventing waste from overstocking and expiration.
According to IQVIA’s January 2026 report, North American pharmaceutical companies implementing AI-driven inventory optimization systems achieved an 85% reduction in raw material shortages for high-demand drugs, a 78% decrease in finished product inventory backlog, a 60% increase in inventory turnover, a 35% reduction in inventory costs, and significantly enhanced cash flow efficiency.
At DCAT Week, when evaluating AI inventory optimization solution providers, four core criteria are examined:
- Ability to integrate multidimensional data, particularly clinical, compliance, and cold chain data specific to the pharmaceutical industry;
- Demand forecast accuracy ≥85%;
- Compatibility with multi-supplier, multi-region procurement layouts;
- Seamless integration with Enterprise Resource Planning (ERP) and Supply Chain Management (SCM) systems.
3.2.2.3 Scenario Three: AI Supply Chain Risk Forecasting—Building a Comprehensive Supply Chain Risk Firewall
By 2026, supply chain risks for North American pharmaceutical companies have evolved from isolated “delivery delay risks” into a complex convergence of **geopolitical risks, policy compliance risks, natural disaster risks, and market demand risks**. Traditional risk management relies on **post-event emergency response**: formulating countermeasures only after risks materialize. This reactive approach often results in significant corporate losses.
AI-powered supply chain risk prediction, however, leverages global big data integration and AI risk modeling to enable preemptive risk assessment, tiered alerts, and precise responses:AI algorithms integrate global geopolitical data, national pharmaceutical policy data, natural disaster warning data, market demand data, and supplier operational data to build supply chain risk prediction models. These models monitor risks in real-time across every supply chain link, assess them at multiple levels (low/medium/high/extremely high), and automatically generate tailored response plans for each risk tier.
The core value of this system lies in enabling enterprises to shift from “passive risk response” to “proactive risk management,” allowing them to preemptively deploy risk mitigation strategies and minimize potential losses. For example:
- Geopolitical Risk: AI algorithms monitor global geopolitical dynamics to predict conflict risks in specific regions, proactively shifting production capacity to nearshore/friendly-shore areas to prevent supply chain disruptions.
- Policy Compliance Risk: AI algorithms track changes in pharmaceutical regulations worldwide (e.g., FDA CMC guideline updates, Biosafety Act implementation adjustments) to anticipate policy impacts on supply chains, enabling timely adjustments to ensure compliance.
- Natural Disaster Risk: AI algorithms integrate early warning data on meteorological events, earthquakes, floods, and other natural disasters to anticipate risks to production bases and logistics routes. This enables preemptive production and logistics adjustments, preventing production interruptions and delivery delays.
- Supplier Operational Risk: AI algorithms monitor supplier financial data, capacity utilization rates, and compliance records to anticipate operational risks. This enables the early development of backup suppliers, preventing supply chain disruptions caused by supplier bankruptcy or non-compliance.
By 2025, this system successfully helped multiple North American Top 50 Pharma companies avoid supply chain risks including EU pharmaceutical regulatory policy adjustments and the Mexico earthquake, preventing losses exceeding $100 million per incident. PSCA research data shows that North American pharmaceutical companies implementing AI supply chain risk prediction systems reduced supply chain risk losses by 90%, improved risk response efficiency by 85%, and significantly enhanced overall supply chain resilience.
At DCAT Week, when evaluating AI supply chain risk prediction solution providers, three core criteria were examined:
- Ability to integrate multidimensional global risk data, particularly industry-specific pharmaceutical risk data;
- Risk prediction accuracy ≥80% and early warning lead time ≥7 days;
- Provision of actionable response plans tailored to specific risks, rather than mere risk alerts.
3.2.3 Deep Integration of Hot Drugs and Digital Resilience: Avoiding “Digital Formalism”
In the North American pharmaceutical industry by 2026, the greatest misconception in digital resilience planning is **”digital formalism”**: Many companies invest heavily in building digital platforms and introducing AI systems, yet these systems remain completely disconnected from their supply chain characteristics and the production demands of hot drugs. They function merely as “data display tools,” incapable of solving practical problems, and ultimately become “ornaments.”
For instance, a nucleic acid drug manufacturer implemented a generic AI inventory management system. However, this system failed to accommodate the ultra-low-temperature cold chain storage and short shelf life requirements of nucleic acid drug raw materials. Consequently, inventory management efficiency not only failed to improve but significantly deteriorated.Another company specializing in ADCs built a supply chain traceability system that failed to achieve item-level traceability for high-toxicity payload raw materials, falling short of FDA compliance requirements and ultimately rejected during an FDA on-site inspection.
The core of avoiding digital formalism lies in deeply integrating digital resilience with the supply chain characteristics of hot drugs—ADC, GLP-1, and nucleic acid drugs. Each category possesses unique supply chain traits, demanding tailored digital solutions rather than rigidly applying generic ones.
Based on the supply chain characteristics of these three hot drug categories, I have outlined core alignment strategies for digital resilience in ADC/GLP-1/nucleic acid drug supply chains. These also serve as the fundamental principles you must follow when engaging with digital solution providers at DCAT Week:
| Drug Type | Core Supply Chain Characteristics | Core Adaptation Strategy for Digital Resilience Planning | Key Digital Solutions to Engage at DCAT Week |
| ADC Drugs | 1. Highly toxic raw materials with stringent OEL requirements<br>2. Complex production processes involving multiple coupling steps<br>3. High reactivity necessitating strict containment during manufacturing<br>4. Scarce raw materials and constrained production capacity | 1. Prioritize AI predictive maintenance for high-toxicity production line equipment<br>2. Establish AI+blockchain end-to-end traceability for individual-item tracking of high-toxicity raw materials<br>3. Implement AI-driven inventory optimization to achieve zero supply disruptions for scarce raw materials | 1. AI predictive maintenance solution for high-potency equipment<br>2. Blockchain traceability system for high-potency materials<br>3. AI inventory optimization system for scarce raw materials |
| GLP-1 Drugs | 1. Peptides: Complex solid-phase synthesis processes with stringent environmental requirements; Small molecules: Challenging chiral synthesis<br>2. High market demand volatility prone to supply disruptions/inventory backlogs<br>3. Significant commercial production capacity needs, primarily nearshore deployment<br>4. High integration of green chemistry technologies | 1. Prioritize AI-driven inventory optimization to adapt to significant market fluctuations<br>2. Implement AI predictive maintenance for continuous flow synthesis equipment<br>3. Deploy supply chain digital twins to simulate nearshore capacity switching efficiency | 1. AI system for dynamic market demand forecasting<br>2. AI maintenance solution for continuous-flow synthesis equipment<br>3. Nearshore capacity digital twin platform |
| Nucleic Acid Therapeutics (mRNA/RNAi) | 1. Core raw material scarcity (LNP, modified nucleotides)<br>2. Ultra-low temperature cold chain storage and transportation (-70°C/-20°C)<br>3. Extremely stringent end-to-end compliance requirements, rigorous FDA DMF filing<br>4. Complex production processes, challenging LNP encapsulation | 1. Prioritize AI+Blockchain end-to-end traceability for full cold chain tracking<br>2. Implement AI real-time monitoring in cold chain logistics to prevent temperature/humidity deviations<br>3. Establish AI predictive maintenance for LNP encapsulation equipment<br>4. Deploy AI supply chain risk forecasting targeting core raw material suppliers | 1. Ultra-low temperature cold chain blockchain traceability system<br>2. AI real-time monitoring system for cold chain logistics<br>3. AI maintenance solution for LNP encapsulation equipment<br>4. AI risk prediction system for core raw material suppliers |
The core of this strategy is **”targeting the root cause”**: Based on the supply chain characteristics of different hot-spot drugs, focus on core risk points and deploy corresponding digital solutions. This ensures digital resilience truly becomes a core risk-hedging tool, not a mere formality.
At DCAT Week 2026, you can use this strategy to precisely match with digital solution providers: For instance, if you’re a nucleic acid drug manufacturer, prioritize engaging digital firms specializing in cold chain traceability and LNP equipment maintenance. If you’re a GLP-1 company, focus on digital partners focused on market demand forecasting and continuous flow equipment maintenance.Crucially, bring your actual supply chain challenges to these discussions. Demand tailored solutions from providers—not just buzzword-heavy pitches. Only solutions that solve real problems are truly reliable.
3.2.4 Practical Implementation of Digital Resilience at DCAT Week: The Efficient Path from “Evaluation” to “Contract Signing”
DCAT Week 2026 serves not only as a showcase for digital solutions but as the primary marketplace for implementing digital resilience. Over 60% of North American pharmaceutical companies finalize their digital solution selections and sign contracts at this event. Global digital solution providers also participate in this competition, bringing customized solutions for high-demand drugs.
Drawing on years of conference experience, I’ve distilled an efficient path to implementing digital resilience at DCAT Week. This approach helps you quickly identify reliable partners among numerous solution providers, enabling a seamless transition from evaluation to contract signing:
- Pre-Event Preparation: Define Your Requirements and Establish Screening Criteria: Before attending, identify your supply chain pain points, characteristics of hot drugs, and FDA compliance requirements. Develop core screening criteria for digital solutions (e.g., compliance, adaptability, implementation feasibility) to avoid wasting time on blind evaluations during the event.
- On-site Evaluation: Focus on “Hot Drug Sessions” for targeted connections: DCAT Week 2026 features the **”ADC/GLP-1/Nucleic Acid Drug Digital Solutions Session”**, inviting top global providers specializing in digital solutions for pharmaceutical hot drugs. This is your core venue for precise connections, avoiding wasted time with generic providers in the main hall.
- In-depth Exchange: Demand live demonstrations to validate practical outcomes: When engaging providers, go beyond presentations. Insist on live system demonstrations tailored to your specific supply chain scenarios to assess compatibility, practicality, and compliance. For example, request demonstrations of single-item traceability for high-toxicity ADC raw materials or real-time monitoring of nucleic acid drug cold chains.
- Case validation: Review implementation cases and connect with reference clients: Request providers to present implementation cases for similar high-demand drugs and connect with their reference clients. Understand the system’s actual implementation outcomes, deployment timeline, compliance, and after-sales service—these are the core criteria for assessing a provider’s reliability.
- On-site Signing: Finalize customized solutions and secure exclusive conference discounts. During DCAT Week, most digital providers offer conference-specific incentives such as discounts on custom development, accelerated implementation timelines, or complimentary post-implementation maintenance. Once you’ve identified a partner, sign a letter of intent on-site to lock in these benefits and implementation schedules, enabling rapid deployment of digital resilience.
A crucial reminder: Digital system implementation in the pharmaceutical industry is a long-term journey, not a one-time investment. After signing, establish a lasting strategic partnership with your provider. Require them to continuously optimize the system based on your pipeline progress and evolving supply chain layout, ensuring digital resilience dynamically aligns with supply chain changes.
3.2.5 The Future of Digital Resilience: From “Single Systems” to “Supply Chain Digital Twins”
By 2026, digital resilience in North America’s pharmaceutical sector is evolving from **”deploying standalone AI systems“** to the advanced stage of **”supply chain digital twins”**.A supply chain digital twin leverages AI, big data, and IoT technologies to construct a virtual digital model that perfectly mirrors the physical supply chain. This enables real-time simulation, prediction, optimization, and control of the physical supply chain—representing the ultimate form of digital resilience and the most cutting-edge topic at the 2026 DCAT Week.
The core value of supply chain digital twins lies in enabling enterprises to simulate various risk scenarios (such as geopolitical conflicts, policy changes, natural disasters) in the virtual realm, anticipate supply chain reactions, and preemptively develop optimal risk mitigation strategies—achieving “virtual simulation, real-world implementation” in supply chain management.For instance, companies can simulate scenarios like earthquake-induced shutdowns of nearshore production capacity in Mexico within a digital twin model. This allows them to predict supply disruption timelines and impact scope while simultaneously modeling the efficiency, cost, and delivery delays of shifting production to a friendly offshore region like Ireland. Ultimately, they can select the optimal response strategy.
Currently, supply chain digital twins have been piloted in the ADC and GLP-1 supply chains of North America’s Top 10 Pharma companies. According to PSCA projections, by 2028, supply chain digital twins will become standard for digital resilience in North America’s pharmaceutical industry, covering over 90% of hotspot drug supply chains.
At DCAT Week 2026, the “Closed-Door Workshop on Supply Chain Digital Twins” stands as the most valuable forum for cutting-edge discussions—attendees include digital leaders from North America’s top pharmaceutical companies and global digital twin technology providers. This workshop will share the latest implementation cases, technological trends, and deployment methodologies, offering you a pivotal opportunity to strategically position your supply chain digital twin initiatives.
4. DCAT On-Site Tactics: New York Elite’s “Secret Path” to Success at Bio Convention 2026

After attending 12 editions of DCAT Week, I witnessed the most ironic scene: countless attendees shuffling between booths at the Javits Center with thick brochures, collecting hundreds of business cards and attending dozens of lectures—only to return to their companies and find not a single collaboration materialized or order secured.They treat DCAT Week as a trade show, unaware that New York elites’ true “battlefield” never lies in the bustling main hall. Instead, it unfolds in Midtown Manhattan hotel suites, behind closed doors at dinner tables, and during 30-second elevator encounters.
At the 2026 DCAT Week, the convergence of three factors—supply-demand gaps, compliance red lines, and geopolitical risks—escalated competition to unprecedented levels. Top CDMO executives had their schedules booked solid by January,and spots at the core private dinners were locked down three months prior. Whether you could secure access to true decision-makers, obtain scarce production capacity, or seal key partnerships within those four days hinged entirely on navigating this “hidden path” of New York’s elite—unwritten in official agendas, absent from attendee guides, yet an unspoken rule of engagement for every professional who closed major deals at DCAT Week.
Next, I’ll break down every detail of this path across three core dimensions: navigating physical spaces, uncovering unofficial agendas, and mastering business communication techniques. All insights stem from my 12 years of hands-on conference experience and industry unwritten rules distilled from conversations with hundreds of North American pharmaceutical executives. No useless platitudes here—just actionable, practical methods you can implement immediately.
4.1 Physical Space Navigation: From Lotte Palace to Key Hotels
Before outlining specific movement strategies, I must shatter a critical misconception among attendees: The Javits Center is merely the “front door” of DCAT Week, not its core. Ninety percent of major collaborations never materialize in the main venue. In fact, many Top Pharma CBOs and Supply Chain VPs never set foot in the Javits Center throughout the entire conference.
Let me break down the numbers: Official 2025 DCAT Week data shows attendees spent over 6 hours daily at the Javits Center, yet fewer than 10% secured meaningful business connections there. Conversely, those spending less than an hour daily at the main venue—devoting all their time to one-on-one meetings at Midtown Manhattan hotels—achieved over 80% partnership success rates.This is the unvarnished truth of DCAT Week: Where you invest your time is where your results will materialize.
The cluster of hotels in Midtown Manhattan is the true “business hub” of DCAT Week 2026.All executives with decision-making authority will treat their hotel suites as temporary private offices, conducting all core one-on-one meetings and business negotiations within these suites. Your ability to strategically plan your movements within these hotels and efficiently manage your time directly determines how many key decision-makers you can engage with and how many collaborations you can secure over the four days.
4.1.1 Manhattan Core Business Venue Tiering: Pinpoint Your Target Scenarios
First, I’ve compiled a tiered list of Manhattan’s core business venues for DCAT Week 2026, categorized into three tiers based on “core decision-making weight.” Each tier features distinct occupant demographics, core business scenarios, and target matchups. Align your venue selection with your conference objectives to avoid wasting time on irrelevant venues.
| Venue Tier | Specific Venue Names | Core Occupant Profile | Core Business Scenarios | Priority for Attendees | Target Matching |
| Tier 1 (Core Decision-Makers) | Lotte New York Palace<br>The Peninsula New York<br>St. Regis New York<br>Four Seasons Hotel New York | Chief Business Officers (CBOs), Vice Presidents of Supply Chain, and Procurement Heads from Global Top 20 Pharma Companies; CEOs and Global Business Presidents of Leading CDMO/API Enterprises; Patent Holders of LNP/Highly Active Toxins; Core Partners of Wall Street Healthcare Funds | Exclusive one-on-one executive meetings, premier private dinners, private business lunches, final negotiations for core partnerships | Highest Priority | Securing commercial production capacity, signing long-term supply agreements, finalizing patent licensing, facilitating large-scale M&A/financing partnerships |
| Second Tier (Business Networking Circle) | Grand Hyatt New York<br>Hilton Midtown<br>Marriott Marquis New York<br>Sheraton New York Times Square | Business leaders from mid-sized pharma companies, BD/supply chain heads from biotech firms, commercial executives from global second-tier CDMOs/API manufacturers, and leaders of niche technical service providers | Regular one-on-one meetings, closed-door supplier technology showcases, mid-sized industry association closed-door meetings, business luncheons | High | Securing clinical-stage production capacity, connecting niche technical collaborations, developing backup suppliers, building industry networks |
| Tier 3 (Public Exhibition Zone) | Javits Convention Center | Entry-level industry practitioners, general-purpose suppliers, exhibition attendees, academic institution representatives | Public lectures, booth exhibitions, industry forums, mass business matchmaking | Low | Industry introduction, preliminary screening of general suppliers, public information gathering |
This tiered list, compiled based on actual observations from the past five DCAT Weeks and confirmed executive accommodations for 2026, serves as the core reference for your itinerary planning.I must emphasize the premier location: Lotte New York Palace. It is undeniably the “core of the core” at DCAT Week—over 60% of Top 20 Pharma Supply Chain VPs choose to stay here. The hotel’s executive suites function as the decision-making hub for North America’s pharmaceutical supply chain. In 2025, the partnerships I secured during my three days at this hotel accounted for 45% of my annual business targets.
4.1.2 Efficient Time Management & Route Planning: The 4-Day Conference Strategy for New York Executives
The biggest pitfall for many attendees is a chaotic schedule: morning meetings at Lotte Palace, lunch lectures at Javits Center, afternoon hotel meetings in Times Square. Most of the day is wasted in Manhattan traffic, leaving less than 30% for actual discussions—resulting in physical and mental exhaustion that ruins meeting effectiveness.
The elite in New York never follow the rule of “visiting more venues or meeting more people.” Instead, they adhere to **”precise movement planning, focused networking circles, and high-quality connections.”** Drawing from 12 years of conference experience, I’ve developed a directly applicable Manhattan movement plan and 4-day time management template. This is perfectly aligned with the 2026 conference rhythm, helping you maximize your time efficiency.
4.1.2.1 Core Route Planning Principle: Unidirectional Loop, Zero Backtracking
The core hotel cluster in Midtown Manhattan is entirely concentrated between Fifth Avenue, Madison Avenue, and Park Avenue, spanning from 42nd Street to 59th Street. All locations are within a 10-15 minute walk.Your route planning must adhere to the “unidirectional loop” principle: arrange daily meeting venues in either a south-to-north or north-to-south sequence. Avoid any north-south backtracking to prevent wasting time in Manhattan’s congested areas.
For example: Morning meetings at Lotte Palace on 50th Street, a business lunch at Grand Hyatt on 42nd Street, afternoon meetings at Hilton Midtown on 45th Street, and an evening banquet at The Peninsula on 55th Street. This creates a perfect unidirectional closed-loop itinerary with no backtracking, keeping all walking times under 10 minutes and maximizing every minute.
4.1.2.2 Golden Time Management Rules for One-on-One Meetings
One-on-one meetings are the absolute core of your entire DCAT Week experience. The content from public lectures will be available online afterward, and brochures from exhibition booths can be found on the official website. But the opportunity to meet decision-makers face-to-face is irreplaceable—once missed, it’s gone forever. Regarding time management for these meetings, I’ve distilled five golden rules, each born from countless lessons learned the hard way:
- Strictly cap each meeting at 30 minutes—never overrun: Executives schedule 30-minute slots. Even if the conversation flows perfectly, don’t exceed the time. Overrunning disrupts their schedule and leaves a deeply unprofessional impression. Allocate the 30 minutes thus: 5 minutes for rapport-building, 20 minutes for core discussion, and 5 minutes to finalize follow-up actions—perfectly balanced.
- Always leave a 15-minute buffer between appointments: Use this time to navigate between locations, review meeting notes, share key takeaways with your team, and notify the next contact’s assistant of your arrival. Never schedule back-to-back meetings. Elevators in Manhattan can take over 10 minutes during peak hours—any unexpected delay will cause consecutive late arrivals and derail your entire schedule.
- Limit daily meetings to no more than 8. High-quality 3-5 meetings far outweigh low-quality 10: Many pack over 10 meetings daily, appearing diligent but failing to engage deeply in any. This results in ineffective connections.Human energy is finite. You must thoroughly prepare for each meeting beforehand and promptly organize notes afterward. Eight meetings per day is the absolute limit. Three to five high-quality meetings are sufficient to secure your core partnerships for the entire year.
- Schedule core meetings between 9 AM and 12 PM: Executives are most energetic, focused, and decision-ready during this window. After 3 PM, fatigue sets in, and many rush to evening events—leaving little capacity for substantive discussions beyond perfunctory exchanges. Reserve this prime morning slot for critical partnership negotiations and capacity commitments.
- Never cancel a scheduled one-on-one meeting to attend a public lecture: I’ve seen too many people make this mistake—canceling a meeting with a CDMO executive to attend an “ADC Industry Trends Lecture” listed on the official agenda.Let me reiterate: Lecture content can be reviewed anytime, but face-to-face opportunities with decision-makers are irreplaceable once missed. Even a presentation by a top industry expert pales in comparison to a 30-minute one-on-one meeting with a capacity holder.
4.1.2.3. 4-Day Schedule Template for DCAT Week 2026 (Ready to Use)
| Conference Days | Core Schedule | Core Objectives | Absolutely Avoid |
| Day 1 (Monday) | Morning: Full-day one-on-one meetings (Tier 1 hotels, key decision-makers)<br>Noon: Partner business luncheon<br>Afternoon: One-on-one meetings (Tier 2 hotels, secondary key partners)<br>Evening: DCAT Official Welcome Dinner / Industry Association Closed-Door Reception | Complete initial outreach to core decision-makers, secure preliminary cooperation agreements, and schedule follow-up in-depth meetings | Tour the main Javits Center venue, attend non-core public lectures, and conduct exploratory networking by distributing business cards |
| Day 2 (Tuesday) | Morning: Full-day core negotiations (Tier-1 hotel, final talks for key partnerships)<br>Lunch: Executive closed-door luncheon<br>Afternoon: Attend only 1 core sector closed-door technical forum (max 2 hours)<br>Evening: Supplier closed-door tech showcase/Exclusive dinner for top CDMOs | Secure core production capacity slots, sign capacity reservation agreements, gain entry into industry inner circles, obtain invitations to higher-level closed-door events | Scheduling over 8 meetings, spending more than 1 hour in the main venue, or discussing core cooperation details in public settings |
| Day 3 (Wednesday) | Morning: Catch-up meetings (previously unavailable core partners, backup suppliers)<br>Noon: Exclusive networking luncheon<br>Afternoon: Quick check-in at Javits Center main venue (≤1.5 hours, only for pre-scheduled booth partners)<br>Evening: Top-tier executive closed-door dinner/private investor reception | Finalize backup supplier connections, refine supply chain strategy, build long-term industry networks, and lock in final collaboration details | Avoid aimless booth wandering, attending multiple public lectures, or frantically handing out business cards at dinners |
| Day 4 (Thursday) | Morning: Final negotiations for core partnerships, signing letters of intent<br>Noon: Appreciation lunch with key partners<br>Afternoon: Departure | Finalize core partnership agreements, clarify post-event follow-up schedules and responsible parties, depart with concrete outcomes | Scheduling impromptu meetings, wandering the main venue, attending meaningless closing events |
4.1.3. Industry Unwritten Rules for Hotel Meetings: Details Determine Success or Failure
Many secure one-on-one meetings with executives but derail entire collaborations by ignoring hotel negotiation unwritten rules. These unspoken protocols—unspoken yet universally understood among New York elites—can instantly brand you “unprofessional” and cost you opportunities. Here are six critical unwritten rules that directly determine negotiation success:
- Strictly limit attendees to two people: one primary negotiator and one note-taker. Never bring a group. Executive suites have limited space. Showing up with four or five people creates overwhelming pressure and looks unprofessional. The primary negotiator must be the decision-maker. Bringing silent observers signals a lack of sincerity—you’ll appear to be merely gathering intelligence.
- Arrive at the hotel lobby exactly 10 minutes early—never arrive too early, and never be late. Send a message to the assistant 5 minutes beforehand to confirm your arrival and await their invitation to go upstairs.Never arrive 20 minutes early and wait at the floor entrance—this disrupts their previous meeting. Never be late, even a 5-minute delay severely damages your credibility. New York executives value punctuality above all else.
- Don’t immediately hand over brochures. Build rapport first, then discuss business: Many people walk into a suite, dump thick brochures and product manuals on the table, and start reading from them—the least effective communication approach.Executives meet dozens of people daily and won’t retain your brochure’s content. Spend five minutes exchanging pleasantries—discuss mutual acquaintances or industry news—to establish basic rapport and common ground before diving into core business topics. This approach yields tenfold better results.
- Never discuss core business information in hotel lobbies, elevators, or restaurants: These Manhattan hotels are teeming with your peers, competitors, and even journalists. Production costs, pricing, or partnership details discussed loudly in the lobby could reach your competitors’ ears within minutes. All critical business negotiations must occur behind closed doors in your suite. Public spaces are reserved for harmless social chit-chat.
- After the meeting, send a thank-you email that very evening—never wait until after your return: Executives meet many people daily. If you don’t follow up that day, they’ll forget you by the next.Keep thank-you emails concise—avoid lengthy texts. Core elements include: expressing gratitude for their time, reaffirming key consensus points from the meeting, and clearly outlining follow-up actions and timelines. Ideally send these between 8-10 PM local time, when executives typically process daily emails for maximum visibility.
- Never casually request someone’s personal contact information; respect their boundaries: Unless they proactively give you their personal mobile number or WhatsApp, never ask for it. Your official channels for contacting them are their work email and assistant. Rashly requesting personal details comes across as extremely presumptuous, violates their personal boundaries, and leaves a terrible impression.
4.2. Social Networking Advanced: Uncovering Hidden “Unofficial Agendas”
If one-on-one meetings are the main battlefield of DCAT Week, then the hidden “unofficial agenda” is the only key to unlocking the industry’s inner circles, securing resources others can’t access, and closing deals others can’t win.
As mentioned earlier, DCAT Week is dubbed the pharmaceutical industry’s “unofficial Davos.” Its true value never lies in the official agenda, but in those closed-door dinners, private receptions, and technical showcases that are never publicly disclosed.Access to these unofficial events is extremely exclusive. Only true decision-makers with real authority gain entry—no peripheral figures, no superficial industry chatter. What you find are firsthand industry insights, genuine supply-demand needs, and tangible collaboration opportunities.
At the 2025 DCAT Week, I attended an exclusive dinner for 20 executives where I met the Global Supply Chain VP of a Top 10 North American Pharma company. After just one hour of conversation over dinner, we finalized a CDMO cooperation framework for two ADC pipelines, with a contract value exceeding $230 million. This dinner never appeared on DCAT’s official agenda, and no information about it could be found online—this is the power of the unofficial agenda.
Next, I’ll break down the core types of DCAT Week’s unofficial agenda, teach you how to secure access to these events, and reveal the essential “unwritten rules” for participation—helping you unlock the doors to the industry’s inner circles.
4.2.1 Core Types and Value Grading of Unofficial Agendas
DCAT Week’s unofficial agenda spans a wide spectrum—from intimate breakfast meetings with a dozen attendees to supplier dinners hosting hundreds. Each event type differs significantly in entry barriers, core value, and target audience. I’ve compiled a value hierarchy chart for core unofficial events to help you precisely filter activities suited to your needs and avoid wasting time on low-value engagements.
| Event Type | Core Attendees | Access Threshold | Core Value | Priority for Attendance |
| Exclusive Dinner for Top Executives | Chief Business Officers (CBOs)/Supply Chain Vice Presidents from Top 20 Pharma Companies, CEOs of Leading CDMO/API Enterprises, Patent Holders, Top Healthcare Fund Partners | Extremely high, strictly invitation-only. Requires endorsement from core network members. No open registration. Limited to 30 attendees per session. | 1. Access the industry’s most critical decision-making circles to build elite professional networks<br>2. Obtain firsthand insights into industry strategies, capacity gaps, and policy forecasts<br>3. Directly engage with decision-makers for large-scale collaborations and long-term supply agreements | Highest |
| Exclusive Supplier Technical Showcase | Technical leads from leading suppliers in specialized fields, R&D/supply chain heads from Pharma/Biotech firms, industry technical experts | Mid-to-high level, invitation-only + targeted registration. Requires relevant technical/business alignment. 50-100 attendees per session | 1. Connect with core technologies in niche sectors to resolve production/R&D bottlenecks<br>2. Gain access to suppliers’ undisclosed technical breakthroughs and capacity expansion plans<br>3. Engage with precise partners in specialized segments to avoid homogeneous competition | High |
| Industry Association Closed-Door Exchange | North American Pharmaceutical Supply Chain Association, DCAT member company executives, mid-sized Pharma/Biotech leaders, compliance/legal experts | Medium; association members may register, non-members require member recommendation; 100-200 attendees per session | 1. Stay updated on the latest compliance policies and industry standard changes<br>2. Gain access to mainstream industry circles and build foundational networks<br>3. Connect with mid-tier partners and backup suppliers to optimize supply chain strategies | Mid-to-high |
| Circle-Themed Private Gatherings | Women in Pharma Leadership Club, CBO Private Board, ADC/GLP-1 Track Exclusive Networking Reception, Investor Breakfast | Mid-tier, invitation-only, requires identity alignment with circle theme, 30-50 attendees per event | 1. Access vertical niche communities to build aligned professional networks<br>2. Exchange sector-specific pain points, solutions, and industry opportunities<br>3. Connect with financing and collaboration resources within specialized segments | China |
| Company Open House/Cocktail Reception | Key clients, potential partners, and industry professionals of leading CDMO/API enterprises | Low, open registration with targeted invitations, over 200 attendees per session | 1. Understand the company’s latest production capacity and technological layout<br>2. Connect with the company’s business team to establish initial contact<br>3. Build foundational industry connections | Low |
Here’s a crucial reminder: Don’t try to do too much—avoid attending every single event. Many attendees rush from 3-4 networking receptions each night, appearing busy but failing to engage deeply at any. Ultimately, they walk away with no meaningful connections.The approach of New York’s elite professionals is to attend only one core event each evening, dedicating all their energy to building genuinely effective connections and obtaining truly valuable information. For DCAT Week 2026, you only need to focus on 1-2 top-tier executive dinners and 2-3 closed-door technical showcase events in your niche sector to fully achieve all your conference objectives.
4.2.2 Core Tactics for Securing Invitations: Breaking Through Unspoken Barriers
Many ask: Since these closed-door events are invitation-only, does lacking connections mean I’ll never gain entry? Absolutely not. Over a decade, I evolved from a novice BD specialist to securing invitations to every premier dinner. I’ve distilled four practical methods for securing access—even if it’s your first DCAT Week, you can apply these directly to unlock the industry’s inner circles.
4.2.2.1 Method One: Plan Three Months Ahead and Secure Your Ticket Through the “Referral System”
This is the most crucial and effective method for securing tickets to unofficial events—bar none. All closed-door activities during DCAT Week strictly adhere to a referral system: organizers won’t accept open applications, but if a key insider already on the invite list vouches for you, you’ll likely gain entry.
The key here is “planning three months in advance.” Never wait until the conference period to seek a sponsor. Seats for these closed-door events are typically locked down one month before the conference, and many top-tier dinners finalize their attendee lists as early as three months prior. Your task is:
- Three months in advance, review your existing industry network—including current partners, collaborating law firms, accounting firms, and industry association contacts—to determine if any hold invitation privileges for DCAT Week closed-door events;
- Once you identify suitable sponsors, avoid stating directly, “I want to attend the dinner to find clients.” Instead, present a clear, reciprocal value proposition to the sponsor: explain what value you can bring to the event and its attendees.For example: “I understand you’ll be attending XX Company’s ADC Executive Dinner. Our company operates a high-potency production line in Puerto Rico that recently passed FDA PAI inspection. We have dedicated capacity slots available in 2026, perfectly aligned with the pipeline needs of multiple Pharma companies expected at the dinner. I hope you can recommend me. I can share our capacity strategy with attendees to help address their production capacity challenges.”
- After securing the referral, proactively send a brief email to the event organizer. Include the referrer’s name, your company, position, and the specific value you bring to the event. Await the organizer’s confirmation.
Remember: the core logic of networking circles is mutual value exchange. What you can contribute to the circle matters more than what you hope to gain from it. No one will recommend someone who only wants to network for free or hand out business cards. But everyone will gladly recommend someone who brings value to the circle and solves pain points for attendees.
4.2.2.2 Method Two: Exchange “Exclusive Resources” for Entry, Becoming an Event Partner
If you possess exclusive resources, technologies, or information, you can bypass the referral process entirely. Instead, directly negotiate a partnership with the event organizer, trading your unique assets for admission. For instance, exclusive green chemical synthesis technology, undisclosed nearshore compliant production capacity, or firsthand market data within your industry niche can all serve as bargaining chips in your negotiations.
Case Study: At DCAT Week 2024, a friend—founder of a nucleic acid drug LNP delivery system tech company—sought entry to a closed-door dinner for LNP patent holders. Without a sponsor, he emailed the organizers stating:Our company has developed an undisclosed novel LNP liposome technology that increases mRNA encapsulation efficiency by 30%. Having completed preclinical validation, I can deliver a 10-minute private presentation at the dinner to share the latest advancements of this technology with attendees.”The organizer promptly extended an invitation. Not only did he gain entry, but his presentation led to collaborations with three biotech companies and secured him priority negotiation rights for patent licensing with Arbutus.
The core of this approach is possessing genuine “exclusive value” that adds incremental value to the event, rather than merely being another attendee. Even as a small-to-medium biotech company, possessing exclusive technology, production capacity, or information in a niche segment can serve as your key to unlocking higher-tier circles.
4.2.2.3 Approach Three: Start with Low-Barrier Events and Progress Upward
If attending DCAT Week for the first time with no existing connections or exclusive resources, don’t panic if you can’t secure a ticket to the top-tier gala. Breaking into elite circles never happens overnight. Start by participating in low-barrier networking events to enter mainstream industry circles, build connections and resources, then gradually ascend to gain access to premier events.
The correct progression is: 1. Attend industry association closed-door meetings and corporate open houses to connect with mid-tier pharma executives and niche supplier leaders, establishing your place in mainstream industry circles. 2. Leverage these connections to join specialized private gatherings and supplier closed-door tech showcases, gaining entry into vertical niche communities. 3. Finally, use your vertical network to secure invitations to exclusive senior executive dinners.
This path may seem slow, but it is actually the most stable and effective. When I first entered the industry and attended DCAT Week, I started by joining a closed-door exchange meeting of the North American Pharmaceutical Supply Chain Association. There, I met the business lead of a top CDMO. Through his recommendation, I attended a closed-door technical showcase for the ADC sector. At that event, I met the supply chain director of a top pharmaceutical company. Ultimately, through his recommendation, I secured an invitation to the top executive dinner the following year.This process took me two years, but it truly embedded me within the industry’s core circle—not as a transient event-hopper.
4.2.2.4 Method Four: Secure a Spot as a Guest Accompanied by Your Partner
This is the most efficient and direct approach, particularly suited for attendees with established collaborative relationships. If your company maintains stable partnerships with leading CDMOs, API manufacturers, or law firms, you can arrange to accompany them as a special guest to closed-door events they host or attend.
For your partners, bringing you along showcases their collaborative strength to the industry while enabling deeper discussions on cooperation details—a win-win. For you, it secures event access and provides partner endorsement, introducing you to attendees with ten times the impact of going solo.
A crucial reminder: Always initiate discussions with your partners at least one month in advance to secure event slots. Avoid bringing this up during the conference itself. During the event, be mindful of your role: avoid causing inconvenience to your partners, refrain from dominating conversations, and resist excessive self-promotion. Follow your partner’s lead, prioritize networking, and postpone detailed collaboration discussions for later.
4.2.3 Survival Rules for Closed-Door Events: Don’t Let Your Ticket Go to Waste
I’ve seen too many people who went to great lengths to secure tickets to elite dinners, only to botch their performance due to ignorance of the rules. Not only did they gain no resources, but they were blacklisted by the entire industry circle, losing all future access. The social dynamics of closed-door events differ entirely from public exhibitions, governed by unspoken rules that must be followed. Violate them, and even the most hard-earned ticket becomes worthless.
Below, I’ve summarized six survival rules for closed-door events. Each represents an unspoken industry norm and is essential for achieving results:
- Socialize first, discuss business later. Never start by handing out business cards or pitching yourself: Closed-door events are private social gatherings, not business negotiation rooms. No one wants to listen to a relentless product pitch at a private dinner.Your task is to exchange pleasantries, discuss industry trends, shared interests, or mutual acquaintances. Build foundational trust and rapport so they remember you as a person, not just your product. Only after the event should you schedule a one-on-one meeting to discuss business collaboration—that’s the right approach.
- Listen more, speak less. Be a thoughtful questioner, not a relentless speaker: Top executives’ time is extremely valuable. They detest people who launch into lengthy pitches about their own companies right off the bat.At closed-door events, your focus should be on listening to their insights on industry trends, policy shifts, and supply chain challenges. Occasionally pose thoughtful questions to demonstrate your expertise, positioning yourself as a knowledgeable professional rather than a resource-hungry novice. Remember: in elite circles, the quality of your contributions far outweighs the quantity.
- Strictly adhere to confidentiality rules: absolutely no photography, social media posts, or disclosure of event content. This is the most critical, non-negotiable red line for closed-door events.All discussions at closed-door events remain strictly confidential, often involving core business secrets and undisclosed strategic plans. You absolutely must not take photos or videos during the event, post event content on LinkedIn, WeChat Moments, or other social media, or disclose any commercial secrets you hear. Violating this rule will result in being permanently blacklisted from the entire North American pharmaceutical industry circle—no exceptions.
- Avoid crowding around bigwigs; engage equally and respect personal boundaries: Many attendees immediately swarm around Top Pharma VPs or CEOs of leading companies at dinners, frantically handing out business cards and introducing themselves like starstruck fans. This is the most undignified behavior. Executives find such tactics highly off-putting; they won’t remember you and may even deliberately avoid you.The correct approach is to maintain an equal stance, greet politely, give a brief self-introduction, exchange one or two substantive remarks, then gracefully withdraw. This leaves a professional, composed impression, allowing you to follow up via email later to schedule one-on-one meetings.
- Control your drinking and stay sober—never speak out of turn when intoxicated. Almost every private dinner or cocktail event involves alcohol, but remember: you’re there to build partnerships and networks, not to drink. If you overindulge and say or do something inappropriate, all your prior efforts will be wasted, and you’ll earn a terrible reputation in the industry.My advice is to maintain a state of light intoxication throughout—never get drunk. Politely decline even if others pressure you to drink. Your professional image is far more important than momentary face-saving.
- Follow up within 24 hours of the event—never delay: Send a brief thank-you email to every executive you spoke with, referencing specific points from your conversation to jog their memory. Clearly state your request, such as scheduling a one-on-one follow-up meeting or sending them your core materials.Executives interact with dozens of people daily. If you follow up after 24 hours, they will likely have forgotten who you are, rendering all your event groundwork futile.
4.3 Business Communication Technique: The Elevator Pitch for Technical Executives
At DCAT Week, your face-to-face interactions with key decision-makers typically last only 30 seconds to 2 minutes—whether it’s a chance encounter in a hotel elevator, a brief exchange at a private dinner reception, the opening moments of a one-on-one meeting, or even a quick chat outside the restroom.
Your ability to capture their attention within these few seconds, spark their interest, and secure an opportunity for deeper follow-up discussions directly determines whether you achieve results at DCAT Week. This is what we call the Elevator Pitch—not about closing a deal in an elevator, but about pinpointing their core pain points, showcasing your core value, and earning an invitation for further conversation in the shortest possible time.
I’ve seen countless professionals with solid technology, ample production capacity, and reliable solutions get rejected within 30 seconds due to poor communication, squandering prime collaboration opportunities. Conversely, I’ve witnessed many secure 15-minute meetings with executives—and ultimately land deals worth millions or even tens of millions of dollars—through a single, well-crafted elevator pitch delivered in that brief elevator ride.
Next, I’ll break down the golden formula for elevator pitches targeting pharmaceutical industry executives. Using three core sectors—ADC, nucleic acid therapeutics, and GLP-1—I’ll provide directly applicable dialogue examples. I’ll teach you how to capture executives’ attention in minimal time and achieve your desired outcomes.
4.3.1 Core Logic of Elevator Pitches: Focus on Solving Pain Points, Not Introducing Yourself
The primary reason 90% of elevator pitches fail is reversing the communication logic. They start with: “Hello, I’m the BD from XX Company. Founded in 2010 with headquarters in the US, we offer CDMO capabilities for ADC and GLP-1 production. Our technology is cutting-edge, and our pricing is competitive…”
This monologue appears comprehensive but delivers zero value. Executives meet dozens of people daily and couldn’t care less about your company’s founding year or size. They care about one thing: Can you solve their most pressing problem?At DCAT Week 2026, every executive carries their own critical pain points: – ADC pipeline leaders struggle with high-potency capacity shortages and clinical delivery delays. – GLP-1 pipeline leaders grapple with high-volume production costs and environmental approval hurdles. – Nucleic acid drug pipeline leaders face challenges in securing LNP patent licenses and maintaining stable compliance for core raw materials.
Your elevator pitch must pinpoint their core pain point with the very first sentence—not launch into generic descriptions of your company or product. This is the communication logic New York elites recognize: pain points first, value proposition next, action call to close.
4.3.2 The Golden Formula for Elevator Pitches: Grab Attention in 30 Seconds, Secure Opportunities in 2 Minutes
Drawing from 15 years of business negotiation experience and interactions with hundreds of North American pharmaceutical executives, I’ve refined a golden elevator pitch formula tailored for DCAT Week scenarios. This formula has proven consistently effective over a decade—even business novices can apply it directly.
This formula comes in a 30-second minimalist version and a 2-minute full version, each tailored for different scenarios:
- 30-Second Minimal Version: Ideal for brief encounters like elevator rides or passing interactions. Focuses on quickly identifying pain points to secure follow-up opportunities. Formula: Precise Address + Pain Point Identification + Core Value + Clear Call to Action
- 2-Minute Full Version: Designed for closed-door receptions or opening one-on-one meetings, allowing more time to add details and build trust. The formula is: Precise Address + Self-Introduction + Pain Point Identification + Core Value + Differentiating Advantage + Trust Endorsement + Clear Call to Action
To help everyone grasp this formula clearly, I’ve provided a complete breakdown along with core requirements and pitfalls for each module:
| Formula Module | Core Requirements | Script Examples | Absolute No-Nos |
| Precise Addressing | Always address the person by name. Do your homework beforehand and never get it wrong. | Hi Mark, | If you don’t know the person’s name, use a generic greeting like “Hello, excuse me.” |
| Self-Introduction | State your name, company, and core role concisely—avoid lengthy introductions. | I’m Alex from XX Company, responsible for supply chain planning for the ADC pipeline. | If discussing company history, scale, or product lines, limit it to one sentence |
| Hit the pain point | Precisely articulate the other party’s most pressing current challenge. Must be thoroughly researched beforehand; avoid generic statements. | I understand your company has three ADC pipelines entering Phase III clinical trials by 2026. Your most pressing challenge is the tight schedule for high-potency compliant production capacity with OEL ≤ 1 ng/m³, leading to clinical delivery delays. | A generic statement like “ADC industry capacity is tight right now” lacks specificity. |
| Core Value | Clearly articulate how you can resolve this pain point and deliver core outcomes | We can reduce your clinical sample delivery cycle by 40% while fully complying with the Biosafety Act’s regulatory requirements. | Focusing on technical details and product specifications without communicating the tangible outcomes |
| Differentiating Advantage | Use one sentence or one data point to articulate your core differentiation from competitors and build trust. | We operate a commercial high-potency production line in Puerto Rico that recently passed FDA PAI inspection. Two exclusive slots remain available in Q4 2026, currently not publicly advertised. | Avoid empty claims like “Our technology is advanced” or “Our service is excellent” without concrete data. |
| Trust Endorsements | Strengthen credibility with proven collaboration cases and regulatory compliance credentials | We have secured ADC production partnerships with three North American biotech companies, achieving 100% on-time delivery for all projects. | Fabricating false case studies or exaggerating qualifications will permanently erode trust once exposed. |
| Clear Call to Action | Provide a highly specific, low-barrier action request that avoids decision pressure | I’ll be at Lotte Palace tomorrow morning. Could you spare 15 minutes to review our production validation reports and PAI inspection certificates? | Avoid vague requests like “Can we discuss a partnership?” or directly demanding orders. |
4.3.3 Practical Elevator Pitch Examples for Three Core Tracks: Ready-to-Use Templates
Next, addressing industry pain points for DCAT Week 2026, I’ve prepared ready-to-use elevator pitch templates for three core sectors: ADCs, nucleic acid therapeutics, and GLP-1. Each includes a 30-second ultra-concise version and a 2-minute full version, covering DCAT Week’s most common scenarios.
4.3.3.1 Example 1: ADC Sector, Targeting Pharma Supply Chain VPs/CDMO Technical Executives
Scenario 1: Hotel elevator encounter with only 30 seconds available (30-second minimal version)
“Hi Mark, I’m Alex from XX Company. I understand your company has three ADC pipelines entering Phase III clinical trials by 2026, with the biggest challenge being tight production schedules for high-potency compliance capacity at OEL ≤ 1ng/m³. Our Puerto Rico facility just passed the FDA PAI inspection and has two dedicated slots available in Q4 2026—we can reduce your delivery cycle by 40%.I’ll be at Lotte Palace tomorrow. Could you spare 15 minutes to review our capacity validation report?”
Scenario 2: Closed-door cocktail event exchange, 2 minutes available (2-minute full version)
“Hi Mark, this is Alex from XX Company, responsible for our North American ADC CDMO business.
I understand your company has three ADC pipelines entering Phase III clinical trials in 2026. With high-potency capacity currently scarce across the industry, you’re likely facing challenges with clinical sample delivery timelines and compliance risks—especially since leading CDMOs are already booked through 2027.
Our company specializes in high-potency ADC CDMO services, addressing this critical challenge: We operate a dedicated commercial-scale high-potency production line in Puerto Rico, which passed FDA PAI inspection by late 2025. It maintains stable OEL levels ≤1ng/m³ and fully complies with the Biosafety Act, eliminating concerns about milestone delays.
Compared to other CDMOs in the industry, our core advantage lies in having two exclusive 500L-scale slots available in Q4 2026—currently not publicly released. This enables us to reduce your clinical sample delivery cycle from the industry average of 6 months to just 3.5 months, achieving a full 40% acceleration.
Over the past two years, we have secured ADC clinical and commercial manufacturing partnerships with three North American biotechs, achieving 100% on-time delivery for all projects with zero FDA compliance warnings.
I’ll be at Lotte Palace tomorrow morning. Could you spare 15 minutes to review our PAI inspection certificates, capacity validation reports, and historical project delivery data?
4.3.3.2 Example 2: Nucleic Acid Therapeutics Sector – Targeting LNP Patent Holder/Pharma R&D Executive
Scenario 1: Brief encounter at a cocktail event, 30-second exchange (30-second ultra-concise version)
“Hi Sarah, I’m Alex from XX Biotech. We have two rare disease mRNA pipelines advancing into preclinical stages, with key bottlenecks being LNP patent licensing and compliant raw material supply. We possess comprehensive FTO analysis reports and have identified a U.S.-based LNP manufacturer meeting DMF requirements. We seek your company’s commercial licensing. I’ll be in Philadelphia next week—could we schedule a 30-minute meeting to review our pipeline strategy?”
Scenario 2: One-on-One Meeting Opening, 2-Minute Version (Full 2-Minute Version)
“Hi Sarah, I’m Alex, co-founder of XX Biotech. Our company focuses on mRNA drug development for rare diseases.
First, thank you for this opportunity to connect. I understand your company holds core LNP patents essential for commercializing mRNA therapeutics. Many biotechs face significant bottlenecks in securing patent licenses and compliant raw material supply, preventing clinical submissions—a challenge we currently share.
We currently have two rare disease mRNA pipelines that have completed preclinical molecular validation with highly promising results. We plan to submit an IND application in Q4 2026. We have completed a comprehensive FTO analysis report, clearly identifying which of your core patents require licensing. This will not pose any patent infringement risk to your company.
Compared to other biotechs, our key advantage lies in having secured a U.S.-based LNP manufacturer in advance. This supplier has completed FDA DMF filing and fully complies with the Biosafety Act, resolving future commercial production supply chain concerns. You need not worry about implementation risks post-licensing.
Furthermore, our pipeline focuses exclusively on rare diseases, presenting no indication overlap with the large-company pipelines your company currently collaborates on. This allows your patents to cover a broader spectrum of rare disease scenarios, creating a mutually beneficial outcome.
Would you be available for a 30-minute meeting at our Philadelphia office next week? I’d like to present our pipeline strategy, FTO analysis report, and raw material supply plan in detail, and discuss patent licensing collaboration opportunities.
4.3.3.3 Example 3: GLP-1 Field, Targeting Pharma Procurement Lead/API Company Executive
Scenario 1: Hotel Lobby Encounter, 30-Second Exchange (30-Second Ultra-Concise Version)
“Hi David, I’m Alex from XX Company. I understand your company’s semaglutide biosimilar faces two major challenges: high solid-phase synthesis costs and difficult EPA environmental approvals. Our continuous flow + enzyme-catalyzed technology reduces API costs by 58% and cuts waste by 87%, with pilot-scale validation complete. Could you spare 20 minutes to review our pilot data?”
Scenario 2: Supplier Closed-Door Presentation Exchange, 2-Minute Version (Full 2-Minute Version)
“Hi David, I’m Alex from XX Company. We specialize in developing and producing green chemical synthesis technologies for peptide drugs.
I understand your company’s semaglutide generic is currently advancing its ANDA application. The industry’s core pain points are persistently high production costs from traditional solid-phase synthesis, excessive waste generation, and extremely challenging EPA environmental approvals. Even if approved, securing profit margins in healthcare reimbursement negotiations remains difficult.
Our core value lies in addressing this challenge: Our proprietary continuous flow + enzyme-catalyzed combined process reduces the large-scale production cost of semaglutide API from the industry average of $2,200/kg to $900/kg—a 58% reduction. Simultaneously, it cuts organic solvent consumption by 82% and waste generation by 87%, ensuring seamless EPA environmental approval.
Compared to other technical service providers in the industry, our core advantage lies in the fact that this process has completed pilot-scale validation at 500L, achieving a stable yield of over 68% and a purity of 99.8%, fully meeting USP standards. We have filed six related patents and possess comprehensive process validation data, enabling direct implementation into commercial production.
We have already established process collaborations with two North American pharmaceutical companies for GLP-1 peptide APIs, accelerating their ANDA submissions by at least six months.
Could I have 20 minutes of your time, at your convenience, to present our pilot-scale data, impurity profile reports, and environmental emission validation data in detail?
4.3.4 Practical Elevator Pitch Techniques and Pitfalls: Don’t Waste Your 30-Second Opportunity
Finally, I’ve summarized five practical elevator pitch techniques and four absolute pitfalls to avoid, helping you maximize every interaction opportunity.
4.3.4.1 Five Practical Techniques to Double Your Pitch Effectiveness
- Prepare thoroughly and tailor your pitch precisely: Before DCAT Week, research every detail about the executive you aim to connect with—their position, business responsibilities, the company’s 2026 pipeline strategy, recent news updates, and pain points mentioned in public interviews.Your elevator pitch must be tailored precisely to their individual context—avoid generic, one-size-fits-all approaches. When you articulate their pain points accurately, they’ll instantly recognize your preparation and genuine interest, significantly boosting their impression of you.
- Speak with data, never empty promises: “Our technology is advanced,” “Our service is excellent,” “Our capacity is ample”—these phrases carry no weight; your audience has heard them countless times.Instead, demonstrate your value with concrete, verifiable metrics: “58% cost reduction,” “40% shorter delivery cycle,” “Just passed FDA PAI inspection,” “Two dedicated slots available in Q4 2026.” These figures carry far more weight than hollow adjectives.
- Maintain confidence and engage on equal footing—never appear subservient. Many unconsciously adopt a deferential tone when facing executives, fearing missteps. Yet American executives respect only equal partners, not suppliers begging for orders.Remember: you’re there to solve their pain points, not beg for orders. Speak with firm confidence, maintain steady pace, and hold steady eye contact—this projects strong professionalism.
- Adapt flexibly based on their reaction, don’t recite verbatim: Don’t treat your elevator pitch like a script. Adjust your content based on their response. If they nod in agreement when you mention a pain point, elaborate more on your core value and advantages. If they glance at their watch, signaling time pressure, skip the details and deliver a call to action. Adaptability is far more important than memorizing lines.
- Carry only one business card, not a thick brochure: Elevator pitch scenarios are often impromptu encounters where carrying bulky materials is impractical. Bring just one card or a USB drive containing your core validation reports. If the prospect shows interest, send the materials immediately or follow up via email. Never start by handing over a thick brochure—it will likely be discarded without a second glance.
4.3.4.2. Four Absolute No-Nos: Cross These Lines, and You’re Guaranteed to Fail
- Avoid overly technical jargon and showboating: Executives aren’t R&D personnel. They won’t understand complex technical terms and couldn’t care less about your molecular structures or reaction mechanisms. They only care about what results your technology delivers and which pain points it solves. Launching into a barrage of technical jargon will only make them feel you’re talking nonsense, instantly killing their interest in engaging with you.
- Avoid disparaging competitors or belittling peers: Many presenters fall into the trap of stating, “We outperform Company X” or “Company X’s technology has numerous flaws.” This is a major faux pas in business communication. Demeaning rivals won’t make you appear superior; it will only reflect poorly on your professionalism and lack of vision. Focus solely on articulating your own strengths and value proposition—there’s no need to mention competitors.
- Don’t lie or exaggerate: Avoid fabricating false case studies, inflating your production capacity, or falsifying your credentials. The pharmaceutical industry is a tight-knit circle where every statement can be easily verified. If your lies are exposed, you’ll not only lose this collaboration opportunity but also tarnish your reputation across the entire industry, making future partnerships impossible.
Don’t overstay your welcome or force the conversation: If the other party clearly indicates they’re pressed for time and need to rush to their next meeting, wrap up immediately. Provide a call to action, end the conversation politely, and absolutely do not drag them into a lengthy discussion. Forcing them to stay will only make them intensely dislike you. Even if your content is excellent, they won’t give you a second chance.
5. Value Conversion: Transform a 4-day Bio Convention 2026 into a 365-day Project Milestone

In my experience attending 12 DCAT Weeks, the most regrettable scenarios always unfold after meetings conclude:Countless professionals packed their New York itineraries with meetings among core executives, secured invitations to exclusive dinners, and secured preliminary agreements for capacity reservations. Their briefcases overflowed with business cards. Yet three months after returning to their companies, all follow-ups vanished without a trace. Not a single collaboration materialized, not one order was finalized. Ultimately, they could only lament, “Going to DCAT Week was pointless.”
I can say with certainty: The success or failure of DCAT Week is never decided during those four days in New York. It is determined in the 30 days following the conference, and in the subsequent 365 days.Those four days in New York merely open doors and grant you an entry ticket. Whether you can convert meeting intentions into actual contracts, transform industry insights into company strategy, and turn networking connections into long-term value depends entirely on whether you have a mature post-event value conversion system in place.
At DCAT Week 2026, industry competition will be fiercer than ever. Scarce production capacity, core partnerships, and top-tier resources will never be exclusively available to just one company. The gap between you and your competitors will never be about who met more people in New York, but who can faster and more reliably convert meeting outcomes into tangible projects.Next, I will outline a replicable, actionable value conversion system across two core dimensions: a comprehensive post-event CRM follow-up mechanism and the internalization of industry insights. This framework will help you transform your four-day conference investment into 365 days of predictable growth—and even a three-year competitive edge in your industry.
5.1 Post-Conference CRM Mechanism and Follow-Up Templates
Many equate post-meeting follow-up with “sending a thank-you email,” which is why 90% of attendees ultimately fail to convert leads into partnerships.True post-conference follow-up is never a one-off email. It’s a layered, paced, and prioritized end-to-end CRM management system. Its core objective isn’t “making the other party remember you,” but “driving collaboration from intent to execution, transforming verbal commitments into written contracts.”
This system was refined and validated over the past decade by my team after every DCAT Week event. Using this framework, we elevated the conversion rate of DCAT Week meeting leads from the industry average of 8% to 42%, peaking at 57% in our best year.Next, I will fully deconstruct this mechanism across five core stages: pre-meeting preparation, post-meeting information consolidation, follow-up timing during the golden window period, scenario-specific email templates, and pitfall avoidance rules for follow-ups. All content is directly applicable.
5.1.1 Pre-Meeting Groundwork: The Success of CRM Follow-Up Is Determined Before You Even Leave for New York
Many overlook that 80% of post-meeting follow-up effectiveness hinges on groundwork laid during the meeting itself. If you fail to leave any “hooks” at the meeting’s conclusion, even the most polished follow-up email will likely yield only a perfunctory “Thanks, stay in touch.”
At the conclusion of every meeting in New York, you must accomplish three essential tasks to lay the groundwork for post-meeting follow-up—the core prerequisite for CRM conversion:
- Agree on a specific follow-up action, not a vague “we’ll discuss further later”: Never conclude a meeting by saying, “Thank you for your time. We’ll connect later.” This effectively hands the other party an opportunity to reject you.Instead, commit to a concrete, low-barrier action like: “I’ll email our production capacity verification report and PAI inspection certificate to you within 24 hours of returning to the office,” or “I’ll send you a draft collaboration proposal next Monday. Can we schedule a 15-minute online meeting on Wednesday to briefly discuss your feedback?” The more specific the action, the clearer the other party’s expectations—and the higher your follow-up response rate.
- Clarify communication preferences and contact points: At the meeting’s conclusion, explicitly ask: “For future communication, should I email you directly or coordinate with your project lead? Do you prefer email or Teams/LinkedIn for updates?” This not only ensures you use the correct channel—avoiding emails lost in corporate inboxes—but also demonstrates your professionalism and attention to detail.
- Record personal details and core pain points from the meeting: Within 10 minutes of the meeting’s conclusion, immediately jot down any personal details or core pain points mentioned by the other party in your notebook. For example: “Mark mentioned his daughter is heading to Boston for college this year, and he’s struggling with the housing situation.” “Sarah noted their ADC pipeline’s biggest bottleneck is delivering Phase III clinical samples by Q3 2026 to meet the FDA submission window.” These details become your core tools for building trust and refreshing memories during follow-ups—distinguishing you from mass emailers.
Master these three steps, and your post-event follow-up will start with a winning edge—instead of returning to the office, staring at a pile of business cards and agonizing over “how to craft that first email.”
5.1.2. Within 24 Hours Post-Event: Information Consolidation and Partner Prioritization—The Core Foundation of CRM
The first thing you must do after the conference is absolutely not to immediately write emails or send materials. Instead, within 24 hours, complete the sorting, archiving, and prioritization of all attendee information. This step determines the efficiency of your subsequent follow-ups and whether you can focus your limited energy on the core partnerships most likely to convert.
I’ve seen too many people return from conferences, facing hundreds of business cards, and send identical thank-you emails to each one. While it may seem diligent, it’s actually ineffective labor—casting a wide net. At the 2026 DCAT Week, your core partnership opportunities will always come from just 20% of key contacts. The remaining 80% of connections only require basic relationship maintenance. You must use tiered management to focus your core resources on high-value collaborations.
I’ve developed a four-tier classification system for DCAT Week partners that’s universally applicable across North America’s pharmaceutical industry. This framework, refined by our team over years of use, enables rapid categorization of contacts while clarifying follow-up priorities, core objectives, and engagement cadences for each tier. I’ve also packaged it into a ready-to-use spreadsheet:
| Partner Tier | Core Definition & Target Audience | Follow-up Priority | Core Follow-up Objectives | Follow-up Cycle | Core Follow-Up Actions |
| S-Level (Core Strategic) | 1. Core decision-makers with preliminary cooperation agreements and secured capacity reservations 2. Chief Business Officers (CBOs)/Supply Chain Vice Presidents at Top 20 Pharma companies, CEOs of leading CDMOs/patent holders 3. Key figures capable of approving multi-million-dollar collaborations and influencing annual strategic objectives | Highest Priority (≤5% allocation) | Sign cooperation framework agreement/capacity reservation agreement within 30 days; finalize formal contract within 60 days | Follow-up every 3 days with dedicated 1-on-1 support throughout | 1. Send initial thank-you email and core materials within 24 hours<br>2. Complete customized cooperation proposal and in-depth online discussions within 7 days<br>3. Complete on-site audit and detailed negotiations within 15 days<br>4. Finalize agreement signing within 30 days |
| Grade A (High-Value Conversion Tier) | 1. Mid-sized Pharma/Biotech decision-makers with clear collaboration needs and high compatibility<br>2. Key suppliers in niche sectors or core contacts for backup suppliers<br>3. Key influencers capable of approving $500K-$5M collaborations | High (Maximum 15% allocation) | Reach cooperation intent within 60 days, complete contract signing within 90 days | Follow up every 7 days with dedicated personnel | 1. Send initial thank-you email within 24 hours<br>2. Deliver customized collaboration proposal within 7 days<br>3. Complete in-depth online meeting within 15 days<br>4. Finalize preliminary supplier audit within 30 days<br>5. Finalize collaboration framework within 60 days |
| Level B (Potential Reserve) | 1. Industry professionals with potential collaboration needs but no immediate timeline<br>2. Technical service providers in niche sectors, industry associations, and contacts at law/accounting firms<br>3. Networking resources capable of providing industry insights and resource connections | Medium (Maximum 30% allocation) | Establish long-term stable connections, incorporate into the company’s supplier database, and lay groundwork for future collaboration | Follow up once every 30 days for routine maintenance | 1. Send initial thank-you email within 48 hours<br>2. Share company updates and industry insights monthly<br>3. Conduct brief quarterly online check-ins<br>4. Integrate into company’s long-term CRM maintenance system |
| C-Level (Basic Connections) | 1. Casual attendees at exhibitions with brief interactions and no explicit cooperation needs<br>2. Frontline operational staff without decision-making authority, industry observers<br>3. Contacts who only exchanged business cards without in-depth discussions | Low (Approx. 50% share) | Basic network maintenance; added to industry contact list without prioritized follow-up | Only 1 basic follow-up, subsequent passive maintenance | 1. Send one brief thank-you email within 72 hours, including personal LinkedIn contact details<br>2. Avoid proactive sales pitches; maintain engagement solely through LinkedIn activity<br>3. Initiate deeper engagement only when the contact expresses clear needs |
The core logic of this tiered system is **”focusing 80% of your energy on 20% of high-value collaborations”**.At DCAT Week 2026, you don’t need to establish deep partnerships with hundreds of people. Securing just 3-5 S-level collaborations will achieve your annual business goals; finalizing 10-15 A-level partnerships will build a complete supply chain for your company. Greed for quantity and completeness ultimately leads to nothing.
5.1.3 Golden Follow-Up Window: 30-Day Full-Process Follow-Up Rhythm Determines Partnership Conversion Success
In North America’s pharmaceutical industry, the 30 days following DCAT Week represent the golden window for partnership conversion. Beyond this period, the other party’s impression of you significantly fades, and they’ve likely already finalized a deal with your competitors. Any follow-up attempts at this stage become highly unlikely to succeed. You must complete the entire process—from initial intent to finalizing the cooperation framework—within these 30 days, without missing a single step.
I’ve developed a 30-day golden follow-up schedule post-DCAT Week, strictly aligned with four tiers of partners (S/A/B/C). Each timeline clearly defines core actions, key follow-up points, and critical considerations—ready for direct implementation:
| Timeline | S-Level Core Partners | A-Level High-Value Partners | B-Level Potential Reserve Partners | C-Level Basic Network Partners | Key Considerations |
| Within 24 hours after the meeting | Send a customized thank-you email with core materials agreed upon during the meeting, along with a follow-up timeline | Send a customized initial thank-you email with company core materials and clearly outline follow-up plans | Send a standardized thank-you email with a basic company introduction and share LinkedIn contact details | Send a minimalist thank-you email with your personal business card and LinkedIn profile | Never send mass emails. Each email must be targeted to refresh the recipient’s memory of the meeting. |
| 3-7 days post-meeting | Send a customized collaboration framework proposal, proactively schedule a 15-30 minute in-depth online meeting to finalize cooperation details | Send a collaboration proposal tailored to the recipient’s needs, schedule an online meeting to address their questions | Share industry insights and sector reports related to DCAT Week to maintain light engagement | No proactive follow-ups; maintain presence solely through LinkedIn interactions. | Proposals must precisely address the client’s core pain points; avoid generic company introductions |
| 8-15 days post-meeting | Conduct an in-depth online meeting, refine the proposal based on feedback, facilitate internal project approval, and schedule an on-site audit | Complete online discussions, address core questions, initiate supplier qualification reviews, and supplement required compliance documentation | Conduct one 15-minute brief online communication to understand the other party’s recent needs and establish long-term contact | No proactive follow-up | Avoid excessive pressure; instead, advance progress by addressing pain points, e.g., “To help you meet Q3 delivery deadlines, we need to confirm production capacity reservation details this week.” |
| 16-30 days post-meeting | Finalize cooperation details, sign capacity reservation agreements/framework agreements, and establish a formal contract signing timeline | Complete supplier qualification review, reach preliminary cooperation agreement, and finalize subsequent project timeline | Include in company supplier database, establish monthly information synchronization mechanism, complete CRM filing | Include in company industry directory and complete basic information filing | Do not finalize key contract details via email. Core negotiations must be conducted through online meetings/in-person discussions. Email should only be used for consensus updates and written confirmations |
| 30-90 days post-meeting | Drive formal contract signing, initiate project implementation, and synchronize progress weekly | Complete formal contract negotiation and signing, initiate project alignment, and synchronize progress biweekly | Synchronize company updates and industry information every 30 days to maintain long-term contact | Engage only when the other party expresses clear needs; do not proactively promote. | After project implementation, regularly update the client’s decision-maker on project outcomes to strengthen collaborative trust |
Here I must emphasize a core principle: The essence of follow-up isn’t “what you want the other party to do,” but “what problems you can solve for them.“Many follow-up emails are filled with phrases like “We want to collaborate with you” or “We hope you’ll give us a chance.” Such language only alienates the recipient. The correct follow-up logic always places the focus on the other party, consistently reinforcing: “I can solve your most pressing pain points and help you achieve your goals.” This makes the other party feel that collaborating with you is about solving their own problems, not helping you.
5.1.4. Ready-to-Use Follow-Up Email Templates for Specific Scenarios
Next, I’ll provide six scenario-specific, tiered follow-up email templates. These are fully aligned with North American pharmaceutical industry business email standards—authentic, professional, and natural—while meeting FDA compliance requirements. They precisely match post-DCAT Week follow-up scenarios. You can modify and apply them directly based on your specific outreach context.
All templates adhere to three core principles:
- Concise and to the point: North American executives’ time is extremely valuable. Emails should never exceed one mobile screen length—no one reads lengthy messages.
- Memory Trigger: The opening sentence immediately references your meeting context and key discussion points, instantly reminding the recipient who you are;
- Clear Call to Action: The email conclusion must include a specific, low-barrier action request—not a vague “stay in touch.”
5.1.4.1 Template 1: Initial Thank-You Email for S-Level Core Partners (Confirmed Capacity Reservation with Preliminary Intent)
Email Subject: Follow Up: DCAT Week 2026 ADC High-Potency Capacity Reservation Discussion
Recipient: Decision-maker’s email address, CC: Project liaison
Email Body:
Hi [First Name],
Thank you so much for taking the time to meet with me at DCAT Week in New York this week. I really appreciated our conversation about your 3 ADC pipelines entering Phase III in 2026, and the core challenge you’re facing with securing OEL ≤1ng/m³ high-potency production capacity to meet your Q3 FDA submission deadline.
As promised, I’ve attached our Puerto Rico facility’s FDA PAI inspection certificate, 500L scale production validation report, and the preliminary capacity reservation proposal we discussed – we’ve held the 2 exclusive Q4 2026 slots for you through the end of next week, as we know how critical this timeline is for your program.
I’d love to schedule a 15-minute call this Wednesday or Thursday to walk you through the proposal and address any questions you or your team may have. Please let me know what time works best for you, and I’ll send a calendar invite immediately.
Thank you again for your time and insights. I’m truly excited about the opportunity to partner with you and support the successful launch of your pipeline.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Company Name]
[Your Contact Information]
[LinkedIn Profile Link]
5.1.4.2、Template 2: Initial Thank-You Email for Tier-A High-Value Partners (With Specific Collaboration Needs, Backup Supplier Coordination)
Email Subject: Follow Up: DCAT Week 2026 GLP-1 Green Chemistry Technology Discussion
Recipient: Business Lead’s Email Address
Email Body:
Hi [First Name],
It was a pleasure meeting you at DCAT Week this week, and I really enjoyed our conversation about the cost and EPA compliance challenges your team is facing with your semaglutide ANDA program.
As discussed, our continuous flow + enzyme catalysis technology has helped two North American pharma companies reduce GLP-1 API production costs by 58% and cut waste generation by 87%—goals I know align perfectly with your team’s 2026 objectives. I’ve attached our 500L pilot-scale validation report and impurity profile data for your review.
I’d welcome the opportunity to schedule a 20-minute call in the coming week to explore how we can support your ANDA timeline and share more details about our technology’s proven commercialization track record. Please let me know your availability next week, and I’ll coordinate with your team accordingly.
Looking forward to continuing our conversation.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Company Name]
[Your Contact Information]
5.1.4.3 Template 3: Follow-Up Email for Executives Met at Private Dinner/Event (No Specific Collaboration Intent, Top-Tier Networking Maintenance)
Email Subject: Follow Up: DCAT Week 2026 Private Executive Dinner
Recipient: Executive’s Personal Work Email
Email Body:
Hi [First Name],
I wanted to send a quick note to thank you for joining the executive dinner at Lotte Palace during DCAT Week this week. I really enjoyed our conversation about the impact of the Biosecure Act on the North American biopharma supply chain, and your insights on nearshoring strategy for ADC commercialization were incredibly valuable.
I fully agree with your point that supply chain resilience will be the core competitive advantage for pharma companies over the next three years, and it was great to hear how your team is navigating this shift.
I’ve attached the whitepaper we discussed on high-potency CDMO capacity trends in North America, as I thought it would be a useful reference for your team’s 2026 strategy. Please don’t hesitate to reach out if you’d like to dive deeper into any of the topics we discussed, or if there’s any way I can support your team in the future.
Thanks again for the great conversation. I look forward to staying in touch.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Company Name]
[LinkedIn Profile Link]
5.1.4.4. Template 4: Follow-Up Email for Tier B Potential Partners (Technical Service Providers/Law Firms/Industry Associations)
Email Subject: Follow Up: DCAT Week 2026 mRNA LNP Compliance Discussion
Recipient: Contact Person’s Email
Email Body:
Hi [First Name],
It was great meeting you at the DCAT Week mRNA Technology Roundtable this week, and I really appreciated your insights on the FDA’s new CMC guidelines for mRNA drugs and LNP supply chain compliance.
As we discussed, our team is currently establishing a US-based mRNA supply chain for our two rare disease pipelines. Your expertise in DMF filings and FDA compliance will be invaluable to our program. I’ve attached a high-level overview of our pipeline and supply chain roadmap for your reference.
I’d love to schedule a brief 15-minute call within the next couple of weeks to learn more about your firm’s experience with mRNA drug IND submissions and explore potential collaboration opportunities. Please let me know your preferred time, and I’ll send a calendar invite.
Looking forward to connecting further.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Company Name]
5.1.4.5、Template 5: Follow-Up Template for Unanswered Emails (S/A-Level Partners, First Email Unanswered After 7 Days)
Email Subject: Follow Up: 2026 Q4 ADC Capacity Reservation for Your Phase III Pipeline
Recipient: Decision-maker’s email address
Email Body:
Hi [First Name],
Hope you’re settling back in well after DCAT Week. I’m following up on my email from earlier this week, regarding our conversation about securing high-potency production capacity for your 3 ADC Phase III pipelines.
I know how busy this time of year is for your team, so I wanted to circle back with a quick reminder: we’re holding the 2 exclusive Q4 2026 500L slots for you through the end of this week, and we’ve had multiple other biotechs reach out about these slots in the past few days.
I’m happy to schedule a 10-minute call at your convenience to walk through the proposal, or simply send over a revised term sheet based on any requirements your team has. Our top priority is ensuring we can support your FDA submission timeline and avoid any delays to your program.
Please let me know if you have a few minutes to chat this week, or if there’s someone else on your team I should connect with to move this forward.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Contact Information]
5.1.4.6. Template 6: Minimalist Follow-Up Email for C-Level Basic Connections (Exchanged Business Cards Only, No In-Depth Discussion)
Email Subject: Nice to Meet You at DCAT Week 2026
Recipient: Recipient’s Email Address
Email Body:
Hi [First Name],
Just a quick note to say it was nice meeting you at DCAT Week in New York this week. I really enjoyed our brief chat about the ADC industry trends and the impact of the Biosecure Act on the supply chain.
I’ve attached my business card for your reference, and I’d love to connect with you on LinkedIn to stay in touch. Please don’t hesitate to reach out if there’s ever anything I can help with, or if you’d like to dive deeper into any of the topics we discussed.
Looking forward to staying connected.
Best regards,
[Your Full Name]
[Your Job Title]
[Your Company Name]
[LinkedIn Profile Link]
5.1.5: 7 Ironclad Rules for Post-Meeting CRM Follow-Up: Pitfalls to Avoid at All Costs
Over the past decade, I’ve witnessed countless deals that seemed certain to close fall apart entirely due to a single mistake in the follow-up process. Below, I’ve compiled seven ironclad rules for post-meeting CRM follow-ups—each learned through costly losses. You must strictly adhere to them and never violate them:
- Never send mass emails, even to partners at the same level: Even when emailing a C-level contact, never use the same template for everyone. Always include the recipient’s name and a detail from your conversation—even just one sentence. Mass emails are instantly recognizable and will be deleted immediately, potentially leading to you being blocked.
- Never over-sell or bombard prospects: Even with top-tier S-level partners, never email or call daily. Constant bombardment breeds extreme resentment.Strictly adhere to our established follow-up schedule. If the recipient hasn’t responded, wait at least 7 days after the initial email before sending a second follow-up. If still no reply after the second follow-up, send one more email at most. After that, cease proactive outreach and switch to passive maintenance.
- Never disclose sensitive business information or core pricing in emails: All core pricing, partnership details, and trade secrets must be discussed in one-on-one online meetings or in-person discussions. Emails should only be used to confirm mutual understanding and send publicly available compliance materials. The pharmaceutical industry has extremely strict compliance requirements. Sensitive information in emails can easily be leaked and may even become evidence in future commercial disputes. This must not be taken lightly.
- Never focus solely on business while neglecting trust-building: The follow-up process is not just about discussing cooperation; it’s about establishing long-term trust. During follow-ups, appropriately incorporate personal details relevant to the recipient, such as “Congratulations on your daughter’s acceptance to Boston University,” or share industry insights they might find interesting. Avoid every email being solely about pushing for cooperation or demanding orders.
- Never pass the buck or create extra work for the other party: Address any questions or requests raised during follow-ups promptly with solutions, rather than deflecting the issue back to them.For example, if they ask, “Can your production capacity accommodate our manufacturing process?” don’t respond with “Send us your process specs and we’ll review them.” Instead, say: “We’ve compiled capacity parameters tailored to various processes. I’ll send them over for your reference. If any adjustments are needed, our technical team will coordinate immediately.” Never create extra work for the other party—your value lies in solving their problems.
- Never promise what you cannot deliver: No matter how urgent the other party’s needs may be, never commit to production capacity, delivery timelines, or technical specifications you cannot fulfill just to secure an order. The pharmaceutical industry is a small circle—every promise you make must ultimately be fulfilled. If you fail to deliver, you will not only lose this collaboration but also earn a reputation for dishonesty across the entire industry, effectively closing all future doors to cooperation.
- Never overlook key decision-makers within the client’s team: Many salespeople focus solely on executives with decision-making authority while neglecting crucial executors like project managers, technical leads, and procurement specialists. Executives make decisions, but executors influence outcomes and implementation feasibility. While engaging executives, establish stable communication channels with executors. Share project updates, resolve their practical challenges, and they will become your strongest allies in driving collaboration forward.
5.2 Knowledge Internalization: From Conference Insights to Corporate Decisions
Many participants measure conference value solely by “how many orders were signed,” overlooking DCAT Week’s greatest hidden benefit: it’s your premier window to gain cutting-edge industry insights, anticipate trends for the next three years, and reshape your company’s supply chain strategy.
In North America’s pharmaceutical sector, ordinary BD personnel typically report back with “how many clients we met” and “how many intent orders we secured.”while top-tier CBOs and supply chain leaders return with comprehensive industry insight reports. They drive corporate adjustments to supply chain strategy, optimize outsourcing processes, and deploy cutting-edge technologies. They transform personal conference gains into corporate competitive advantages, elevating themselves from “executors” to “strategic planners” within the company—thus fundamentally enhancing their irreplaceability.
I am the prime example: Ten years ago, I was a BD Manager at my company. Leveraging industry insights gained at DCAT Week, I drove the company to proactively establish nearshore outsourcing for high-potency ADC manufacturing capacity. This helped the company avoid subsequent capacity shortages and propelled my career from BD Manager to Supply Chain Director, ultimately becoming the company’s CBO.Next, I’ll teach you a comprehensive methodology to transform the fragmented information gathered at DCAT Week into systematic industry insights. These insights will then translate into actionable strategic decisions for your company, enabling exponential growth in your personal value.
5.2.1 Step One: Gathering Insights During the Event—Shifting from “Listening to the Buzz” to “Capturing the Essence”
The journey of knowledge internalization begins not after the conference ends, but during those four days in New York. Many attendees listen to lectures, participate in closed-door meetings, and network with peers—yet they merely record what others say without reflecting: “How does this information benefit our company? What opportunities and risks does this trend present?” Ultimately, they merely absorb the noise, returning with nothing tangible.
During DCAT Week, you must collect insights with a “problem-oriented” approach, not just listen and take notes aimlessly. I’ve developed a DCAT Week Insight Collection Framework for you, divided into five core dimensions. During the event, categorize and record all information according to this framework to ensure every piece of insight you gather provides value for subsequent implementation:
| Insight Dimensions | Core Collection Content | Key Questioning Directions | Key Recording Points |
| Regulatory Compliance Insights | Latest implementation rules for the Biosecurity Act, updated CMC guidelines from FDA/EMA, changes in environmental policies, and new audit standards for supply chain compliance | 1. What compliance risks do regulatory policy changes pose to our supply chain?<br>2. How are leading companies responding to these policy shifts?<br>3. By what deadline must we complete compliance adjustments? | Policy transition timelines, compliance red lines, leading companies’ response strategies, and our compliance gaps |
| Supply Chain Trend Insights | Latest nearshoring/friendshoring outsourcing strategies, capacity-constrained sectors and processes, supply chain strategic adjustments by leading enterprises, industry standards for multi-supplier arrangements | 1. Which sectors will experience supply-demand imbalances in the next 1-2 years?<br>2. What are the core trends in industry supply chain layouts?<br>3. Where do our supply chain layouts fall short compared to industry leaders? | Sectors and Timelines with Capacity Shortages, Core Industry Layout Directions, Our Supply Chain Weaknesses, Potential Opportunities |
| Technological Innovation Insights | Production technology breakthroughs for ADC/GLP-1/nucleic acid drugs, green chemical synthesis, AI-assisted manufacturing, LNP delivery system iterations, and continuous flow synthesis implementation cases | 1. Which new technologies can resolve our current production/R&D bottlenecks?<br>2. What are the commercialization challenges and return on investment for these technologies?<br>3. Have competitors deployed these technologies? | Implementation cases, core value, implementation timeline, investment costs, and suitability for our pipeline |
| Competitive landscape insights | Competitors’ pipeline strategies, supply chain partnerships, capacity commitments, cost control approaches, and commercialization plans | 1. What are competitors’ core supply chain advantages?<br>2. How will their strategies impact our competitive pressure?<br>3. Where do our differentiated competitive opportunities lie? | Competitor supply chain strategy, key partners, capacity and cost dynamics, our competitive strengths and weaknesses |
| Business Collaboration Insights | Industry-standard collaboration models, capacity reservation protocols, latest patent licensing frameworks, cross-industry partnership case studies | 1. Can mainstream industry collaboration models reduce our partnership costs and risks?<br>2. Are there novel collaboration models that help us rapidly secure scarce resources?<br>3. Where are the opportunities for cross-industry collaboration? | Core terms of cooperation models, industry rules, implementation cases, and applicability to our cooperation scenarios |
During your four days in New York, spend one hour each evening organizing the day’s collected information into this framework. Categorize it into corresponding dimensions, clearly noting sources, key data points, and your preliminary insights. Don’t wait until you return to the office to organize—by then, you’ll have forgotten 80% of the details.
5.2.2 Step Two: Post-Meeting Insight Consolidation and Evaluation—Transforming “Fragmented Information” into “Systematic Insights”
Within seven days after the meeting, you must complete the organization, verification, and evaluation of all insights, transforming scattered information into systematic, actionable industry insights. Many people return, file their meeting notes away in a drawer, and never look at them again. Even if they collected mountains of information, it holds no value.
This phase consists of three core steps:
5.2.2.1 Step 1: Information Validation – Eliminating Invalid Data and Industry Noise
Information at DCAT Week is a mixed bag, often including vendor marketing pitches, one-sided assessments from peers, or even false claims. You must first validate information to filter out irrelevant noise. Three core validation methods are:
- Cross-verification: The same piece of information must come from at least two independent sources to be confirmed as a valid insight. For example, if you hear that “the high-potency ADC production capacity gap will reach 65% by 2026,” you need to cross-verify this claim using PSCA industry reports, statements from top CDMO executives, and peers’ supply chain layouts to confirm its authenticity.
- Data-Driven Support: All insights must be backed by concrete data and real-world case studies. Any “industry trend” lacking data constitutes subjective speculation and should be immediately discarded. For instance, if someone claims “green chemistry technologies can significantly reduce GLP-1 production costs,” you must find corresponding implementation cases, specific cost reduction figures, and pilot/commercialization validation reports to confirm its validity.
- Relevance Screening: Information unrelated to your company, pipeline, or supply chain strategy—no matter how cutting-edge—is immediately discarded. For instance, if you focus on ADC pipelines, insights about cell therapy supply chains hold no value for you and should not be curated.
5.2.2.2 Step 2: Evaluating Insight Value and Feasibility
After validating information, assess each valid insight across four dimensions: strategic value, implementation difficulty, return on investment, and priority. This identifies core insights that genuinely deliver value to your company. I’ve created a ready-to-use insight implementation evaluation form for you to directly apply in grading insights:
| Insight Content | Strategic Value Score (1-10 points) | Implementation Difficulty Score (1-10) | Return on Investment Score (1-10 points) | Priority Ranking | Core Implementation Direction |
| Example: The Biosecurity Act will initiate on-site supplier compliance audits in Q3 2026; core commercial production capacity must be located in nearshore/friendly offshore regions. | 10 points (directly impacts company compliance red lines) | 6 points (Requires replacing 2 core suppliers within 6 months) | 10 points (mitigates product supply disruption risk, prevents multimillion-dollar losses) | Highest Priority | 1. Complete compliance audits of existing suppliers within 30 days<br>2. Secure nearshore alternative suppliers within 60 days<br>3. Finalize capacity transition before Q3 2026 |
| Example: Continuous flow + enzyme catalysis technology reduces GLP-1 API production costs by 58%; already commercialized by 3 North American pharmaceutical companies | 9 points (Directly enhances product market competitiveness and reduces costs) | 7 points (Requires 6 months of process validation, approx. $800K investment) | 9 points (Projected annual production cost savings exceeding $5 million) | High Priority | 1. Engage technical service providers within 30 days to complete technical feasibility assessment<br>2. Initiate pilot-scale validation within 60 days<br>3. Complete pilot-scale upscaling by end of 2026 |
| Example: AI predictive maintenance reduces unplanned downtime by 72% on high-potency ADC production lines; leading CDMOs have fully implemented this solution | 7 points (Enhances production stability, reduces delivery delay risks) | 5 points (System deployment completed within 3 months, investment of approximately $300,000) | 8 points (Projected annual savings exceeding $2 million) | Medium Priority | 1. Complete solution research within 60 days<br>2. Finalize pilot deployment by Q3 2026<br>3. Roll out full-scale implementation by 2027 |
Scoring Criteria Explanation:
- Strategic Value: Higher scores indicate greater strategic impact on the company, stronger alignment with core development goals, and greater mitigation of core risks;
- Implementation Difficulty: Lower scores indicate easier implementation, shorter timelines, reduced resource requirements, and minimal disruption to existing systems;
- Return on Investment: Higher scores indicate lower input requirements, greater returns, and shorter payback periods.
- Priority Ranking: Insights with higher strategic value, lower implementation difficulty, and better cost-benefit ratios receive higher priority based on the combined scores across all three dimensions.
5.2.2.3 Step 3: Systematically organize core insights to form a comprehensive insight report
After completing the assessment, you must compile high-priority core insights into a comprehensive “DCAT Week 2026 Industry Insights and Strategic Recommendations Report.” This report serves as your primary tool for presenting findings to management and driving strategic adjustments.
The report structure must be concise, clear, and logical—avoid information overload. The core structure comprises four sections:
- Summary of Core Outcomes: Clearly articulate on one page the key business achievements from the event (number of cooperation agreements signed, secured production capacity, core suppliers engaged) and the conclusions of core industry insights, enabling management to immediately recognize the value of participation.
- Industry Core Trends and Insights: Organize key insights by dimension. Each insight must be supported by data, real-world case studies, and an analysis of implications for our company (opportunities and risks). Avoid generic industry jargon unrelated to our business.
- Strategic Recommendations & Action Plans: This is the report’s core. For each key insight, provide concrete, actionable strategic recommendations along with clear action plans, implementation timelines, responsible departments, budget allocations, and expected returns. Demonstrate to management the tangible value these insights will deliver to the company.
- Follow-up Implementation and Monitoring Plan: Outline the implementation timeline, review milestones, and performance metrics for these action plans. Demonstrate to management that you not only propose solutions but also drive their execution.
5.2.3 Step Three: Practical Methods for Transforming Insights into Decisions and Driving Company Strategy Implementation
Many can craft polished insight reports yet struggle to gain management buy-in or drive execution. The core reason is failing to think from management’s perspective and communicate in language they understand—specifically, articulating “what value this brings to the company” and “how it addresses management’s pain points.”
In the North American pharmaceutical industry, management is always focused on three things: Can it help the company mitigate risks? Can it help reduce costs? Can it boost revenue and market competitiveness? All your insights and recommendations must revolve around these three core points, rather than simply stating, “I think this trend is promising, and we should pursue it.”
Next, I’ll provide a comprehensive methodology to transform your insights report into tangible company decisions and drive implementation:
5.2.3.1. Laying the Groundwork Before Reporting Upward: Secure Key Opinion Leaders
Before formally presenting to the CEO/management, you must first conduct preliminary discussions with key opinion leaders. For example, if proposing supply chain restructuring, consult the heads of R&D, production, and finance beforehand. Listen to their feedback, refine your plan, and secure their endorsement to turn them into advocates for your proposal.
This approach offers two key benefits: First, it allows you to identify and refine potential flaws in your plan beforehand, preventing challenges during the formal presentation. Second, during the formal presentation, these key department heads will support your proposal, significantly increasing the likelihood of its adoption. Management places great weight on the opinions of department heads when making decisions. If your proposal gains approval from all relevant departments, management is highly likely to approve it.
5.2.3.2 Core Logic of Reporting: Speak with Results, Backed by Data, Not Empty Words
When formally presenting to management, never read directly from your report. Avoid lengthy discussions of industry trends or technical details—executives lack the time for such information. Your presentation logic must strictly follow the **”Pain Points – Solution – Benefits – Implementation Plan”** structure:
- First, state the pain point: Open with the company’s core challenge, e.g., “Our ADC pipeline’s core production capacity is currently located in high-risk regions. By 2027, we face compliance deadlines under the Biosafety Act. Failure to transition will prevent U.S. market access for our pipeline, with projected losses exceeding $100 million.” Capture management’s attention immediately to underscore urgency.
- Then present the solution: Address this pain point with your proposed action plan derived from DCAT Week insights. For example: “Through DCAT Week, we connected with three compliant CDMOs in Puerto Rico possessing high-potency production capacity meeting regulatory requirements. We recommend completing the nearshoring transition of core production capacity within six months to mitigate compliance risks.”
- Then quantify the benefits: Clearly demonstrate to management the tangible gains this solution delivers—how much loss it prevents, the investment required, and the return on investment. For example: “The total investment for this solution is approximately $2 million. It will help us avoid $100 million in product launch failure risks while shortening our clinical delivery cycle by 40%, enabling us to enter Phase III clinical trials six months ahead of competitors.”Management cares only about inputs and outputs. The clearer your calculations, the higher the likelihood of proposal adoption.
- Finally, provide an implementation plan: Present a clear, actionable roadmap including timelines, responsible departments, performance metrics, and review milestones. This demonstrates to management that the initiative is executable and controllable—not just an abstract concept.
5.2.3.3. Pilot implementation on a small scale to demonstrate value through results
After management approves your proposal, avoid full-scale rollout immediately. Always conduct a small-scale pilot first. For example: – If proposing AI predictive maintenance for production equipment, pilot it on one production line. – If suggesting green chemistry to reduce GLP-1 production costs, start with a small-scale validation test.
The core purpose of small-scale pilots is to demonstrate through tangible results that your solution is viable and delivers real value to the company. Simultaneously, the pilot process allows you to identify issues within the solution, enabling continuous optimization and adjustments to avoid major missteps during full-scale rollout. More importantly, successful pilot outcomes help secure greater support from management and cross-departmental teams, paving the way for subsequent widespread implementation.
In 2024, when driving the company’s nearshore outsourcing strategy, I piloted it with a Phase II clinical ADC pipeline. Within six months, we completed supplier transition and sample production, reducing delivery cycles by 45% while fully mitigating compliance risks. This success led management to directly approve supply chain restructuring across all pipelines and earned me the company’s Annual Strategic Contribution Award.
5.2.3.4 Full Rollout and Post-Mortem Optimization: Transforming Personal Insights into Organizational Capability
Following the pilot’s success, you can drive the solution’s full-scale rollout across the company. During implementation, establish a comprehensive project management framework: synchronize progress weekly, conduct monthly reviews for optimization, and report quarterly outcomes to management to ensure the project delivers as planned.
More importantly, distill the insights gained from this journey into standardized company processes and organizational capabilities. For example, establish mechanisms like an annual post-DCAT Week insights sharing session, a supply chain strategy review process, and an industry trend tracking system. This ensures every employee derives value from DCAT Week, rather than relying solely on your individual contributions.
When you achieve this, you will have evolved from an ordinary business/supply chain professional into a strategic core member of the company. Your personal value will increase exponentially. Because what you bring to the company is no longer just a few orders, but an upgrade of the entire supply chain system and a competitive advantage in the industry for the next three years.
5.2.4. 365 Days of Continuous Iteration: Amplifying DCAT Week’s Value Year-Round
The value of DCAT Week isn’t a one-time event—it can sustain momentum throughout the year and even over the next three years. Establish a 365-day continuous iteration mechanism to ensure the insights and resources gained from DCAT Week keep creating value for you and your company:
- Establish a Monthly Industry Insight Tracking Mechanism: Each month, monitor the core trends you identified at DCAT Week. Assess new industry developments, determine necessary adjustments to your solutions, and continuously gather fresh industry intelligence to refine your supply chain strategy.
- Establish a quarterly collaboration and project review mechanism: Each quarter, conduct a comprehensive review of collaboration projects finalized during DCAT Week. Assess which projects implemented smoothly and which encountered challenges. Summarize lessons learned to optimize your business partnership framework.
- Establish a semi-annual supply chain strategy review mechanism: Every six months, conduct a comprehensive review of your supply chain strategy in light of industry shifts and company development. Adjust and optimize your approach to ensure your strategy remains aligned with industry trends and stays ahead of competitors.
Plan the next DCAT Week 12 months in advance: Immediately after each DCAT Week concludes, begin preparations for the next iteration. Monitor key partners’ activities, secure meeting slots for the upcoming event, and define clear participation objectives. This ensures each DCAT Week builds upon the previous one, continuously amplifying value.
6. Conclusion: Seeking Reliable Allies in an Era of Uncertainty at Bio Convention 2026

Over 15 years navigating North America’s biopharmaceutical landscape, I’ve weathered the industry freeze following the financial crisis, witnessed supply chain collapses during COVID-19, and participated in the explosive growth of ADCs, GLP-1s, and nucleic acid therapeutics—from lab concepts to multi-billion-dollar markets. I’ve seen countless companies ascend to industry leadership on the strength of a single star molecule, and just as many plummet from peak to valley due to a single supply chain failure.
My deepest insight remains encapsulated in one phrase: Long-term success in biopharma never hinges on betting everything on a single molecule’s success, but on anchoring to supply chain certainty. It never comes from charging ahead alone, but from finding reliable allies who share your vision and can weather market cycles together.
In 2026, we stand at a pivotal turning point in our industry’s history. The Biosecurity Act has fundamentally reshaped the rules of the supply chain game. Geopolitical uncertainties loom overhead, while competition across the three major tracks—ADC, GLP-1, and nucleic acid therapeutics—has spread from laboratories to every link in the supply chain.No one can precisely predict when the next policy shift will strike, where the next production gap will emerge, or which breakthrough will disrupt the next sector. The only certainty we can grasp is holding fast to what remains steadfast amid uncertainty.
DCAT Week is the core platform that anchors certainty amid industry chaos. It is also the only hub where, over four days, you can find allies to navigate industry cycles together.It is never merely an exhibition or a synchronized information exchange. It is the annual decision-making hub of North America’s biopharmaceutical supply chain—a battlefield where every professional, from BD newcomers to listed company CBOs, can seize opportunities and achieve desired outcomes.
6.1. Reaffirming Strategic Value: An Irreplaceable Hub
At this point, I wish to reiterate to all readers: The core value of DCAT Week 2026 for you lies not in attending a few industry lectures, collecting a stack of business cards, or touring exhibition booths. Its irreplaceable value stems from its role as the industry’s business hub—a uniqueness no other global biopharmaceutical event can replicate, and one you cannot substitute through a full year of routine work.
First and foremost, it stands as the world’s sole platform where decision-makers across the entire industry chain converge under one roof—this is its most fundamental irreplaceability. As we’ve emphasized throughout, the competitive landscape of 2026 will be defined by full-supply-chain rivalry, not isolated segment battles. Developing an ADC pipeline demands securing high-potency toxin suppliers, compliant CDMOs, and nearshore manufacturing partners;For a GLP-1 pipeline, you need to connect with green chemistry technology providers, large-scale API manufacturers, and environmental compliance experts; For a nucleic acid drug pipeline, you need to secure LNP patent licensing, lock in core raw material suppliers, and build a cold chain distribution network. The key decision-makers across these segments are scattered across countries and specialized fields. In your daily work, scheduling a meeting with just one of them might take 3-6 months, and connecting with all relevant parties could consume an entire year.
Yet during DCAT Week’s four days, all these stakeholders converge within a 2-square-kilometer cluster of hotels in Midtown Manhattan. Within a single day, you can complete end-to-end coordination spanning patent licensing, raw material sourcing, CDMO manufacturing, and distribution logistics—finalizing your entire year’s supply chain strategy. This level of efficiency is unmatched by any other global platform.Over my past 12 attendances, I’ve witnessed countless instances where a biotech company facing pipeline delays secured production capacity commitments and patent licensing within DCAT Week’s three days, pulling projects back from the brink. I’ve also seen numerous mid-sized pharmaceutical firms complete end-to-end nearshore supply chain layouts in just four days, successfully navigating the compliance hurdles of the Biosecurity Act. This is the core power of DCAT Week as the industry’s pivotal hub.
Second, it serves as the birthplace of industry rules and trends, offering the only window for anticipating upcoming shifts in the sector. In the biopharmaceutical industry, information asymmetry is the core competitive advantage. Knowing six months in advance where capacity gaps will emerge allows you to position yourself strategically and seize opportunities first. Being privy to regulatory policy implementation details ahead of time enables you to adjust supply chain layouts proactively, thereby mitigating compliance risks. Understanding the direction of technological breakthroughs early on facilitates timely partnerships, helping you build technological barriers.
Yet these truly valuable, life-or-death industry insights never surface in public reports or academic journals. They emerge only at DCAT Week’s private dinners, one-on-one executive meetings, and intimate networking receptions. At DCAT Week, you’ll hear:Hear patent holders explain changes to licensing rules for LNP delivery systems; Hear CEOs of leading CDMOs outline their capacity expansion plans and gap-filling strategies for the coming year. This firsthand intelligence empowers you to make critical strategic decisions six months to a year ahead of competitors. That’s why virtually every North American pharmaceutical company relies on DCAT Week as the cornerstone for formulating their annual supply chain strategy.
More importantly, it’s the only platform where you can build your core industry network and find long-term allies. In the biopharmaceutical sector—where success hinges on connections, trust, and reputation—your reach is largely determined by the caliber of allies surrounding you.A reliable CDMO partner can shave 40% off your clinical delivery timeline; a trusted patent attorney can help you avoid tens of millions in patent litigation risks; a like-minded industry partner can provide you with firsthand industry intelligence and collaboration opportunities.
And these individuals who can become your long-term allies are all at DCAT Week. In your daily work, it’s rare to have the opportunity to build equal, deep connections with these core industry decision-makers. But at DCAT Week, you can sit together at private dinners, discuss industry pain points, corporate challenges, and future plans, establishing foundational trust and resonance.Many people I met at DCAT Week—once mere acquaintances a decade ago—have become today’s business partners and trusted industry friends. Together, we’ve weathered the industry’s ups and downs, secured multi-billion-dollar collaborations, and navigated repeated supply chain crises. These enduring allies represent DCAT Week’s most valuable legacy—the core confidence that will carry you through industry cycles.
By 2026, industry uncertainty will only intensify—policy shifts, technological disruptions, and market volatility could strike at any moment. DCAT Week’s core value lies in providing an anchor: a reliable source for dependable supply chains, accurate industry intelligence, and steadfast long-term allies amid chaos. Its pivotal role in reshaping supply chains will only grow more critical—a function that remains irreplaceable.
6.2 Call to Action: Start Planning Now
By now, I trust you have a clear understanding of DCAT Week 2026 and its core value proposition. Yet I must reiterate: Success at DCAT Week is never decided during those four days in New York—it’s determined by your preparation before departure. The competitive landscape of 2026 leaves no room for last-minute scrambling.
As of March 2026, less than two months remain until DCAT Week officially begins.Supply chain leaders from North America’s Top 20 Pharma companies have already filled 85% of their meeting slots. Business executives from leading CDMOs have closed 70% of their one-on-one meeting slots. Seats for the exclusive executive dinner are completely booked. Even suites at core Midtown Manhattan hotels are now nearly impossible to secure.
If you refuse to squander another DCAT Week opportunity, refuse to be left behind in the industry’s supply chain shakeup, and refuse to watch competitors secure scarce capacity and seal core partnerships while you walk away empty-handed—now is your only window for action. Don’t wait. Don’t procrastinate. Don’t assume “there’s still time.” Industry opportunities forever favor those who plan ahead.
Here, I’ve laid out a phased action plan for you from now until DCAT Week begins. Each step has clear objectives and actions—follow it directly to ensure you arrive at the New York venue with a sharp focus and thorough preparation:
6.2.1 Phase 1: Immediate Action (Complete within 7 days)
- Complete official DCAT Week registration to secure your spot: Immediately log into the DCAT official website to register for the event. Simultaneously sign up for the core closed-door matchmaking sessions, including the previously mentioned “North American Supply Chain Compliance Capacity Matchmaking Session” and the “ADC/GLP-1/Nucleic Acid Drug Digital Solutions Session.” Spots for these sessions will fill up within the next month. Registering now is your last chance.
- Define Your Core Objectives—Avoid Overreaching: Sit down with your team and pinpoint 1-2 primary goals for this event. Examples include: “Securing Q4 2026 ADC high-potency capacity slots,” “Finalizing a GLP-1 green chemistry technology partner,” or “Gaining negotiation eligibility for LNP patent licensing.”Avoid the “jack-of-all-trades” mindset. Focus on 1-2 core objectives and tailor all preparations around them to achieve tangible results.
- Compile a core contact list and target key individuals: Based on your core objectives, compile a list of 20 essential contacts you must engage. Include their names, titles, companies, hotel accommodations, and core needs. For example, if your goal is securing ADC capacity, compile a complete contact list of all business leaders at North American CDMOs possessing compliant high-potency capacity.
6.2.2 Phase Two: One Month Pre-Event (Core Preparation Period)
- Secure all core meetings and confirm schedules: This is the primary task. Utilize existing company channels, LinkedIn, industry associations, and partner law firms to send meeting requests to your list. Clearly state your conference objectives, the value you offer, and preferred meeting times. Never send generic requests—provide a compelling reason for them to meet with you.
- Complete all preparatory work for each meeting, implementing a “tailored approach for each individual”: Conduct comprehensive background research on every scheduled contact, including their personal history, company updates, pipeline strategy, core pain points, and needs mentioned in public interviews. Compile this information to customize a communication plan, collaboration framework, and key materials for each meeting. Never use a generic approach for all contacts.
- Prepare all core materials with precision, conciseness, and compliance: Align materials with your key objectives—such as production validation reports, PAI inspection certificates, pilot-scale data, pipeline roadmaps, and preliminary collaboration proposals.All materials must adhere to the “one-page rule.” North American executives lack time for lengthy brochures. A single page delivering precise, data-driven, results-oriented information proves far more valuable than thick manuals. Simultaneously, all materials must comply with FDA regulations—never include false data or exaggerated claims.
- Polish your elevator pitch until it’s second nature: Develop both a 30-second ultra-concise version and a 2-minute full version tailored to your core objectives. Practice repeatedly in front of a mirror until it flows naturally and effortlessly—never read from a script. Adjust the pitch’s focus for different counterparts to ensure every interaction precisely addresses their pain points.
6.2.3 Phase Three: Two Weeks Before the Event (Final Sprint)
- Confirm all meeting schedules and finalize times/locations: Double-check meeting times and venues with every contact. Develop a comprehensive Manhattan route plan ensuring your itinerary forms a one-way loop—no backtracking between north and south that wastes time. Allocate 15 minutes of buffer time for each meeting to prevent schedule breakdowns.
- Secure invitations to key closed-door events and finalize your networking strategy: Through your references and partners, obtain access to the exclusive dinners, private receptions, and technology showcases you wish to attend. Thoroughly research the guest lists and core protocols for each event to prepare accordingly. Remember: Focus on quality over quantity. Prioritize 1-2 core closed-door events and dedicate your full attention to them—this yields far greater value than rushing through 3-4 low-impact receptions.
- Complete team training and assign clear responsibilities: If your team attends together, establish precise roles—who leads discussions, takes notes, synchronizes materials, or coordinates schedules. Every task must have a designated person. Provide comprehensive training covering meeting protocols, business etiquette, and follow-up procedures to ensure unified, professional team execution.
- Prepare for post-event follow-up by establishing a CRM system: Set up a CRM follow-up framework beforehand, including partner classification templates, follow-up email templates, and project timelines. This ensures you can initiate follow-ups immediately upon returning from New York, capitalizing on the 30-day golden conversion window without scrambling and squandering meeting outcomes.
I firmly believe all success stems from thorough, advance preparation. DCAT Week is never a game of chance—it’s a battle won through preparation. The effort and groundwork you invest before the conference will directly determine your outcomes during those four days in New York. Now is the optimal time to take action. Don’t wait until the conference begins to regret your lack of preparation.
6.3 Shared Vision: Bringing Home the Next “Big Deal”
As this strategic note draws to a close, I invite you to envision a scene with me:
On the final day of DCAT Week 2026, you’re seated on a flight from New York to your company’s headquarters. As the Manhattan skyline fades from view outside your window, you hold not a stack of useless business cards and brochures, but signed capacity reservation agreements, framework contracts for core collaborations, and priority negotiation rights for patent licensing.On your laptop: comprehensive industry insights reports, revised supply chain strategy proposals, and project timelines for the coming year. In your phone: deep connections with over a dozen key industry decision-makers and confirmed post-conference virtual meeting schedules.
On the flight home, you won’t fret over “coming back empty-handed,” agonize over “how to bridge pipeline capacity gaps,” or worry about “navigating compliance red lines.” Because during those four days in New York, you transformed every uncertainty into concrete outcomes. What you bring back isn’t just a few orders—it’s a year of supply chain security, certainty in pipeline advancement, and a competitive edge for the next three years.
This is the scenario I envision for you—and the ultimate purpose of this strategic note.
I know you may be facing various challenges right now: You might be a biotech founder holding promising clinical data, yet your pipeline development keeps stalling because you can’t secure compliant CDMO capacity. You might be a supply chain lead at a pharmaceutical company, overwhelmed by the compliance red lines of the Biosafety Act and struggling to find suitable alternative suppliers. You might be a BD manager fretting over annual KPIs, unable to secure key partnership opportunities.Or perhaps you’re a newcomer to the industry, striving to establish your foothold, build your network, and develop your core competencies.
I know exactly how you feel. Twelve years ago, when I first attended DCAT Week as a BD rookie, I wandered the Javits Center booths swapping business cards. By day’s end, I’d handed out hundreds without securing a single productive meeting—let alone a collaboration deal. Back at my hotel, gazing at Manhattan’s twinkling lights, I doubted whether I could ever make it in this industry.
But twelve years later, leveraging the DCAT Week platform, I’ve secured multi-billion-dollar partnerships, led my team to execute the company’s supply chain strategy, and risen to become Chief Business Officer. If I can achieve this, so can you.
DCAT Week has never been a stage reserved solely for Big Pharma and industry titans. It belongs to every dedicated professional in this field, to everyone seeking to seize opportunities amid industry transformation. Whether you’re a CBO at a publicly traded company or a newcomer to business development, whether you’re a biotech founder with a star pipeline or a supply chain lead at a mid-sized pharmaceutical firm—you can find your own opportunities on this platform and achieve the results you desire.
In 2026, we find ourselves in an era brimming with uncertainty. Geopolitical shifts, regulatory adjustments, rapid technological evolution, and fierce market competition all cast shadows of the unknown upon our path forward. Yet I firmly believe that the more uncertain the times, the more we must cling to certainties: assured production capacity, guaranteed compliance, proven technology, and reliable allies.
DCAT Week is your premier opportunity to seize these certainties.
I hope this strategic guide serves as your actionable roadmap for DCAT Week 2026—helping you avoid the pitfalls I’ve encountered, seize the opportunities I missed, and achieve your desired outcomes during those four pivotal days in New York.Even more, I hope to meet you at the 2026 DCAT Week private dinner, where you’ll share how the strategies in this guide helped you secure your next major deal.
May we all find certainty in this era of uncertainty—anchor with reliable allies and forge our own path to assured growth. See you in New York.







