- 1.0. biotech conference washington dc: Introduction — Washington 2026 as a "Turning Point" forMarket Accessof High-PricedInnovative Drugs
- 2.0. biotech conference washington dc: New HEOR Challenges and Framework Iterations Amid the 2026 Innovation Wave
- 3.0. biotech conference washington dc: ISPOR 2026 Core & “Hidden” Agenda Highlights — Focus on Actionable Insights
- 4.0. biotech conference washington dc: Practical Conference Guide — Maximize Your Takeaways Like a Seasoned Industry Veteran
- 5.0. biotech conference washington dc: Global Outlook — Lessons from China/Asia for Innovative Drug Expansion
- 6.0. biotech conference washington dc: Conclusion — Take Action to Shape a Sustainable Future for Innovation
- 7.0. biotech conference washington dc: Frequently Asked Questions (FAQ)
1.0. biotech conference washington dc: Introduction — Washington 2026 as a “Turning Point” forMarket Accessof High-PricedInnovative Drugs
1.1 Macro Context: The 2026 Biopharmaceutical Landscape and the Intersection with ISPOR’s Top 10 HEOR Trends
Washington, D.C. in 2026 is the premier destination for any biotech conference washington dc — far more than just an ordinary academic gathering. Imagine this: a marketing manager responsible for ADC product access, holding product materials for a treatment priced at $200,000 per course, facing payers at the healthcare negotiation table who are becoming increasingly “savvy”—that mix of excitement and anxiety is precisely the reality for professionals in the high-cost innovative therapies sector in 2026.If you attended ISPOR Europe 2025, our comprehensive pharmacoeconomics attendee guide covered the foundational HEOR landscape — now see how 2026 takes it further.

In recent years, the IRA’s (Inflation Reduction Act) restructuring of Medicare’s drug price negotiation mechanism, the advancement of the CMMI’s new payment models, and the successive approvals of ADCs, CGTs, and nucleic acid drugs—whose prices are increasingly hitting “ceiling” levels—have propelled the entire industry into an unprecedented era of strategic maneuvering. Payers have become more cautious, HTA agencies’ requirements have grown stricter, and the voices of patient organizations have grown louder.
ISPOR’s annual Top 10 HEOR Trends for 2026 reveal a highly concentrated landscape: AI has surged to the top spot, becoming a core tool for evidence generation and decision support; RWE (Real-World Evidence) remains firmly in second place, continuing to serve as a key instrument for addressing the limitations of randomized controlled trials; Drug Pricing has risen to fourth place, reflecting the heightened tension among market stakeholders under the pressure of IRA policies;Meanwhile, Innovative Therapies ranked fifth, indicating that access challenges for high-cost therapies such as CGT, ADC, and nucleic acid therapies have moved from specialized discussions into the mainstream agenda. These four major trends align closely with the theme of theISPOR 2026conference, “HEOR at the Forefront of Policy, Access, and Value,” making this year’s Washington event particularly worthy of serious attention from every professional in the field.
Table 1-1: Analysis of the Correlation Between the 2026 ISPOR Top 10 HEOR Trends and Market Access for High-Cost Innovative Therapies
| Trend Ranking | HEOR Trend Theme | Relevance to High-Cost Innovative Therapies | Practical Value for Attendees |
| No. 1 | AI and Machine Learning Automation | Accelerating RWD Mining, PRO Analysis, and HEOR Modeling | Learn how AI tools can reduce the cost of evidence generation |
| No. 2 | Real-World Evidence (RWE) | Long-term benefit tracking for CGT, supplementary analysis of actual efficacy for ADC | Master Regulatory-Grade RWE Production Methodologies |
| 4th | Drug Pricing Pressure | IRA Negotiations Trigger Pricing Restructuring, QALYs Under Scrutiny | Understand the latest pricing negotiation strategies and countermeasures |
| No. 5 | Access to Innovative Therapies | Barriers to One-Time Cure Payments, Market Access Pathways | Aligning with Cutting-Edge Practices in CGT/Nucleic Acid Drug Access |
| 7th | Health Equity | Do High-Cost Drugs Widen Health Disparities? | Understanding the Regulatory Requirements for Equity-Informative HTA |
| 9th | Patient-Centered Value | The Rising Importance of PROs in HTA | Turning Patient Voices into Leverage in Reimbursement Negotiations |
As this comparison table shows, the 2026 HEOR agenda is not an isolated academic exploration, but a direct response to real-world market pressures. Behind every trend stands a group of practitioners struggling to navigate product launches, access negotiations, or policy advocacy. This is precisely why this year’s Washington event deserves special attention.
1.2 Setting the Tone for the Conference: HEOR at the Forefront of Policy, Access, and Value
ISPOR 2026 (May 17–20, Washington, D.C.) is no longer the traditional conference confined to an academic ivory tower discussing QALYs. To put it more bluntly: this is the “value-proofing mock exam” for high-cost innovative therapies before they actually enter the healthcare reimbursement system.
HEOR is undergoing a profound role transformation: shifting from “remedial work after the fact” (supplementing economic evidence after a product’s market launch) to “early strategic intervention” (designing RWE studies and building value frameworks as early as Phase II). This shift is not driven by the industry itself, but rather by the combined pressure from payers, regulators, and HTA review bodies.
The conference theme, “HEOR at the Forefront of Policy, Access, and Value,” precisely captures the essence of this shift—HEOR’s role has evolved from that of a post-hoc evaluator to a “bridge-builder” between scientific breakthroughs and sustainable reimbursement.In the specific context of 2026, this means: when a CGT product claims to offer a “one-time cure,” HEOR must present a long-term benefit model that convinces commercial insurers; when an ADC moves from second-line to first-line treatment, HEOR must demonstrate that its economic value justifies a higher willingness to pay; and when a nucleic acid drug expands from rare diseases to large populations with chronic conditions, HEOR must find a new balance between health equity and cost-effectiveness.
Multidisciplinary dialogue, real-world case studies, and in-depth engagement with policymakers ensure this conference combines academic depth with practical policy value. Hosting the event in Washington, D.C., in particular, provides a natural boost in policy sensitivity—as the home of the FDA, CMS, and Congress, policymakers may be listening closely behind the scenes of every session.
Table 1-2: Comparison of Roles at the ISPOR 2026 Conference (Traditional Model vs. 2026 New Paradigm)
| Dimension | Traditional HEOR Conference Model | ISPOR 2026 New Paradigm |
| Timing | Generating post-market economic evidence | Value framework design beginning in the early clinical stages |
| Key Topics | QALY Calculation, Cost-Effectiveness Analysis | RWE, OBCs, AI-assisted modeling, Equity-HTA |
| Participant Composition | Primarily scholars and consultants | Policy makers, payers, and patient organizations actively involved |
| Application Areas | Academic publications and supplementary materials for HTA submissions | Directly influences pricing negotiations and decisions on the national health insurance formulary |
| Core Value Proposition | Demonstrates the product’s cost-effectiveness | Build a multidimensional value framework that balances science, reimbursement, and equity |
1.3 Unique Value of This Article: A Guide to Transitioning from Clinical Success to Commercial and Reimbursement Success
This article is not a straightforward summary of the conference agenda. The market is never short of conference previews; what’s missing is a practical framework that helps you translate those abstract session titles into “insights I can use in my meetings tomorrow.”
If you’re leading market access efforts for ADCs, CGTs, or nucleic acid therapeutics, or if your team is mapping out an international expansion strategy, this article aims to help you achieve three goals: Precise pre-conference planning—knowing which sessions to prioritize and which speakers to research in advance; Efficient information gathering during the conference—extracting key insights most relevant to your pipeline from complex HEOR models and policy signals;Post-conference rapid implementation—transforming those “sounds-good” methodologies into a decision-making framework you can present to your team as early as next week.
Next, we’ll start by examining the new challenges facing HEOR, delve into the specific market access pain points for ADCs, CGTs, and nucleic acid therapeutics, provide a strategic interpretation of the conference’s core agenda, and finally offer concrete, actionable recommendations for attending the conference. We hope you’ll head to Washington in May with questions and return with a framework.
2.0. biotech conference washington dc: New HEOR Challenges and Framework Iterations Amid the 2026 Innovation Wave
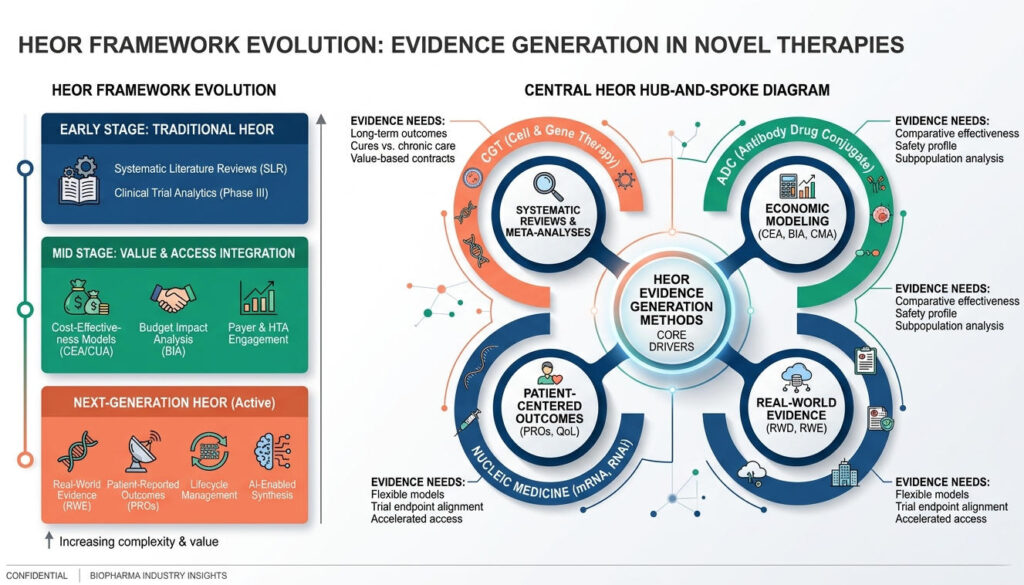
2.1 Common Pain Points for New Modality Therapies: High Costs, Uncertainty, and Pressure for Health Equity
Before delving into specific therapies, we must first acknowledge a fundamental reality: although ADCs, CGTs, and nucleic acid therapeutics differ significantly in mechanism and indications, they face strikingly similar structural challenges at the market access level.
The first challenge is the dilemma of demonstrating “price rationality.”When the price of an ADC (antibody-drug conjugate) reaches $150,000–$250,000 per course of treatment, or the one-time cost of a CAR-T therapy exceeds $500,000, payers are no longer satisfied with data such as “clinical trials show an X-month improvement in overall survival.” They need to know: What is the corresponding change in quality of life for these X months? In the long term, is it truly more cost-effective than existing standard treatments? Can these results be replicated in real-world populations?
The second challenge is the trust gap stemming from “uncertainty regarding long-term benefits.” For the vast majority of innovative therapies, the follow-up period is relatively short at the time of HTA submission (RCT data spanning 18–24 months is particularly common in the CGT field). For therapies claiming to be “curative,” payers naturally ask: What happens if the disease recurs after five years? Are the assumptions underlying extrapolation models reliable? This is not merely a technical issue, but a matter of trust.
The third challenge is the political pressure surrounding health equity. It is no coincidence that equity was explicitly included in ISPOR’s Top 10 Trends.One of the original legislative aims of the IRA was to make innovative medicines affordable for ordinary patients. When a high-cost therapy is shown to be most effective for high-income white patients but has extremely low usage rates among people of color or low-income patients, HTA review agencies find it increasingly difficult to issue a fully positive evaluation. How to systematically incorporate the equity dimension into the value framework has become a core issue that market access teams must confront in 2026.
Table 2-1: Matrix of Common Access Challenges for High-Cost Innovative Therapies
| Challenge Dimension | ADC Drugs | Cell and Gene Therapy (CGT) | Nucleic Acid Drugs |
| Demonstration of Pricing Rationale | Incremental cost-effectiveness is difficult to assess independently in combination therapies | One-time large expenditures are difficult to accommodate within annual budget systems | Conflicting pricing logic between siRNA/ASO for rare diseases and pricing for broad indications |
| Evidence of long-term benefits | Insufficient maturity of OS data; significant controversy surrounding surrogate endpoints | Follow-up data spanning more than 5 years is extremely scarce | Lack of data on long-term adherence and real-world efficacy in the chronic disease sector |
| Health Equity Issues | Uneven distribution of resources among ADC treatment centers for cancer | CAR-T treatment centers are highly concentrated in major cities | Poor geographic accessibility for patients with rare diseases |
| Limitations of Traditional HEOR Frameworks | Markov model health state classifications are not suitable for complex combinations | QALY underestimates the economic value of a one-time cure | Lack of methods for constructing cost-effectiveness models for long-term medication |
Traditional cost-effectiveness analysis (CEA) frameworks were designed during an era dominated by chronic disease management: they assume patients require continuous annual medication, generating ongoing health benefits and healthcare costs each year. While this framework works well for monthly injections of antihypertensive or diabetes medications, the cracks in traditional models become glaringly apparent when applied to cell and gene therapies (CGTs)—which involve a “single injection with lifelong benefits”—or CAR-T therapies, which achieve “complete remission of refractory hematologic malignancies in a single treatment course.”
This is why the evolution of HEOR frameworks is not an option, but a necessity for survival. The 2026 ISPOR conference is one of the most concentrated venues for discussions on this evolution.
2.2 In-Depth Analysis of Technological Fields
2.2.1 ADC Drugs: Next-Generation Technology Trends and RWE-Driven Demonstration of Economic Advantages
If first-generation ADCs (represented by emicizumab) validated the feasibility of the “antibody + toxin” concept, then the new generation of ADCs set to launch or enter late-stage clinical trials between 2024 and 2026 demonstrate a radically different technological ambition.The precision of linker engineering, the diversification of payload toxin profiles, and the emergence of bispecific ADCs are shifting ADCs from being viewed as “enhanced chemotherapy” to positioning them as “precision-targeted weapons.”
However, technological progress does not automatically resolve the challenges of economic positioning. On the contrary, increasing technological complexity brings new challenges to evidence generation:
First, the incremental value of combination therapies is difficult to quantify in isolation. When an ADC is designed by researchers for use in combination with PD-1/PD-L1 inhibitors, payers ask: How much of the improvement in overall survival (OS) is attributable to the ADC itself? What would the efficacy of the ADC be if used alone, without the combination therapy? This question seems simple, yet it is nearly impossible to obtain a clear answer from either single-arm or combination studies.This is where RWE comes into play: by using historical controls, propensity score matching, and other methods to construct a “control group without the ADC,” thereby isolating the ADC’s incremental contribution.
Second, the economic logic is being restructured as treatments expand from second-line to first-line settings. Pricing for second-line therapies can be based on “rescue value following failure of existing treatments,” but the pricing logic for first-line therapies is entirely different—it must demonstrate the “economic rationality of replacing the current standard of care.” As T-DXd expands from second-line to first-line treatment for HER2-positive breast cancer, this pricing restructuring issue becomes particularly urgent.
Third, the expansion into non-oncology indications creates an entirely new reimbursement landscape. Some ADCs are beginning to explore non-oncology fields such as autoimmune diseases and ophthalmology, implying fundamental shifts in payers, HTA regulations, and patient value frameworks. The 2026 ISPOR conference will provide early signals regarding this transition.
Table 2-2: Comparison of ADC Market Access Challenges and HEOR Solutions
| Market Access Challenges | Core Issue | HEOR Solutions | Key Data Sources |
| Incremental Value of Combination Therapy | Inability to Quantify the Contribution of ADCs to Overall Survival (OS) in Isolation | RWE Synthetic Control Arm | EHRs, Cancer Registry Databases |
| Pricing Logic for First-Line vs. Second-Line Treatment | Economic Rationale for Replacing Standard First-Line Therapy | Incremental Cost-Effectiveness Analysis (ICEA) | Head-to-Head RCT + RWE Extrapolation |
| Long-term survival extrapolation | Lack of OS Data Beyond 36 Months | Parametric survival modeling, Bayesian extrapolation | Historical natural history data |
| Real-world efficacy discount | Discrepancy between RCT population and real-world patients | Adjusted real-world HEOR studies | Commercial claims data, hospital data |
| Non-oncology market access | Significant Variations in Reimbursement Rules Across Disease Areas | Development of Disease-Specific Value Frameworks | Patient-reported outcome (PRO) data, patient registry studies |
At the 2026 ISPOR conference, multiple sessions are expected to focus on the intersection of ADC pricing and RWE strategies. For teams responsible for ADC product access, this presents a rare opportunity: not only to observe how competitors are addressing similar challenges, but also to engage in direct dialogue with regulatory representatives to understand the boundaries of their acceptance of the strategy of “RWE supplementing RCT evidence.”
2.2.2 CGT (Cell and Gene Therapy): Reimbursement Challenges for “One-Time Cures” and OBCs/RWE Solutions
The concept of a “one-time cure” is scientifically exciting but a ticking time bomb for the payment system.
Let’s get to the heart of the matter: most commercial insurance plans in the U.S. are renewed annually; Medicaid is constrained by state budget pressures; and under the traditional Medicare model, large one-time expenditures are difficult to budget for.When a CGT product is priced at $2–3 million, the real challenge for payers is: “If I pay this amount this year, but the patient switches insurers next year, who will reimburse my investment?” This is known as the “payer spillover” problem—the most unique and intractable structural barrier in the field of CGT market access.
The industry is currently exploring three potential solutions:
- Outcome-Based Contracts (OBCs): Linking payment to clinical outcomes. For example, if a patient relapses within three years of treatment, the manufacturer refunds a portion of the costs. This model has been piloted for some CGT products, but implementation is highly challenging—the criteria for determining clinical outcomes, responsibility for data monitoring, and contract continuity after an insurance switch all present potential negotiation hurdles.
- Installment/Annuity Payments: This approach breaks down a single large upfront payment into payments spread over multiple years to align with insurers’ annual budget cycles. CMS has begun testing similar models under the CMMI framework, but widespread adoption still faces resistance due to insurers’ cash flow management concerns.
- New CMMI Access Models: The CGT Access Model introduced by the Centers for Medicare and Medicaid Innovation (CMMI) in recent years aims to systematically address this issue at the federal level by reducing systemic barriers through national-level data tracking, outcome verification mechanisms, and multi-payer coordination.
Special Recommendation: If you are attending ISPOR 2026, be sure to prioritize the short course “Using RWE to Inform the Value and Affordability Assessment of Cell and Gene Therapies.”This course will systematically cover three core applications of RWE in CGT evaluation: long-term benefit modeling (how to extend the 24-month observation window of RCTs using real-world follow-up data), definition of natural disease progression (how to describe the disease trajectory of “no treatment” using historical cohorts in the absence of an RCT control group), and gene therapy safety monitoring (a long-term RWE monitoring framework for adverse events).
Table 2-3: Evolutionary Path of CGT Payment Models and HEOR Support Requirements
| Reimbursement Model | Core Logic | Key Challenges | HEOR Support Requirements |
| Lump-Sum Payment | One-time payment, lifetime benefits | Significant budget impact and high risk of spillover effects on payers | Long-term QALY extrapolation models, cost-effectiveness evidence |
| Outcome-Based Contracts (OBCs) | Refunds or reductions if outcomes fall short | Complex outcome definitions and high data tracking costs | Long-term real-world evidence (RWE) follow-up systems and standardized outcome assessment metrics |
| Annuity payments in installments | Spreading large expenditures over multiple years | Significant challenges in coordinating across insurance cycles | Actuarial models, discount rate assumptions, and financial sustainability analysis |
| CMMI New Enrollment Model | Federal-level multi-payer coordination | Uncertain timeline for policy implementation | RWE data infrastructure development and national patient registries |
Based on our observations in actual projects, CGT access teams often do not begin systematically building their HEOR evidence package until 1–2 years before product launch, which is a severely insufficient timeframe.The 2026 Washington conference will provide an excellent opportunity for teams that have already begun planning early on to validate and iterate their evidence strategies, while teams that have not yet started should view this conference as a wake-up call—competitors’ HEOR preparations may be further ahead than you imagine.
2.2.3 Nucleic Acid Therapeutics: Pricing Logic from Rare Diseases to Large Patient Populations and the Potential of AOC Platforms
Nucleic acid therapeutics (siRNA, ASO, mRNA, and the emerging antibody-oligonucleotide conjugates, AOC) have followed a unique market trajectory:From their origins as “orphan drugs” targeting rare genetic diseases, to their entry into the market for rare but increasingly prevalent conditions such as transthyretin amyloidosis (ATTR), and finally to targeting chronic disease markets affecting hundreds of millions of people, such as hypercholesterolemia, hypertension, and non-alcoholic steatohepatitis (NASH). This path of expansion from niche to mainstream reflects a profound evolution in pricing logic.
The core logic of rare disease pricing is “scarcity premium + disease severity”: with a small patient population, R&D costs cannot be spread across a large enough scale, and there are often no alternative therapies, making high pricing inherently justifiable. Patisiran (an siRNA treatment for ATTR) was initially priced at nearly $450,000 per year, and the market largely accepted this.However, when the siRNA technology platform was used to develop inclisiran (administered via two injections per year) for hypercholesterolemia, it faced an entirely different set of reimbursement logic: PCSK9 inhibitors had already established a mature value benchmark, and the affordability of proton pump inhibitors and statins had become deeply ingrained in the public consciousness. Consequently, payers’ expected price for an “injectable lipid-lowering drug” was far lower than in the rare disease sector.
The emerging AOC (antibody-oligonucleotide conjugate) technology represents a bridging strategy: by conjugating a targeted antibody to an oligonucleotide, it addresses the tissue delivery limitations of nucleic acid drugs (currently, the vast majority of siRNA/ASO can only be efficiently delivered to the liver), expanding the scope of nucleic acid drugs to tissues such as muscle, the central nervous system, and tumors.The economic potential of AOCs lies in the fact that, once extrahepatic delivery is unlocked, they can access a broader range of therapeutic indications, theoretically enabling economies of scale while maintaining high technological barriers.
Table 2-4: Comparison of Pricing Logic Across Different Indication Markets for Nucleic Acid Drugs
| Market Type | Representative therapies | Patient Population | Key Factors in Pricing Logic | Key Focus Areas for HEOR Evidence |
| Rare Genetic Diseases | siRNA (ATTR, PH1) | Thousands to tens of thousands | Disease severity + lack of alternatives + orphan drug incentives | Natural history data, PROs, improvement in quality of life |
| Moderate-sized diseases | ASO (SMA, HD) | Tens of thousands to hundreds of thousands | Comparison with existing therapies + magnitude of long-term improvement | Long-term RWE follow-up, comparative effectiveness studies |
| Chronic diseases in large populations | siRNA lipid-lowering (inclisiran) | Tens of millions | Pricing Reference Framework + Health Insurance Affordability + Population Benefits | Budget impact analysis, cost-effectiveness modeling |
| New therapeutic area for AOC | Muscle/CNS-targeted nucleic acids | To be determined (platform-level) | Technological scarcity + First-mover premium | Early-stage proof of concept + patient-centered value framework |
For teams planning to bring nucleic acid therapeutics to the global market, ISPOR 2026 will offer a rare opportunity to gauge U.S. payers’ acceptance thresholds for pricing nucleic acid therapeutics targeting large patient populations. Concurrently, discussions on the Patient-Centered Value Framework will help teams understand how to translate treatment conveniences—such as “two injections per week instead of daily oral dosing”—into monetized value that payers can accept.
2.3 Necessary Iterations of HEOR Evaluation Frameworks: Transitioning Toward Patient-Centered and Societal Value
Traditional cost-effectiveness analysis (CEA) centers on QALYs (Quality-Adjusted Life Years) as its core metric. While this system functioned well in the era of chronic disease management, it is now facing challenges from multiple fronts.
First are the inherent limitations of QALY. QALY multiplies health utility values (numbers between 0 and 1) by years of survival; while this appears to be an objective measure of utility, it systematically underestimates the value of life for patients with rare diseases, pediatric conditions, and disabilities.Even if a rare disease patient’s utility value improves from 0.3 to 0.6 after treatment, their QALY gain remains far lower than that of a patient with a common chronic disease whose utility improves from 0.7 to 0.9. This is not a scientific issue, but a matter of values.
Second is the growing call for distributional cost-effectiveness analysis (Distributional CEA). Building on traditional CEA, Distributional CEA analyzes disparities in the distribution of cost-effectiveness across different population groups (stratified by income, race, geographic location, etc.), clearly identifying whether a given intervention exacerbates health inequalities. The inclusion of “Equity” in ISPOR’s Top 10 Trends reflects this very call.
Third is the rise of the societal valuation framework.This framework expands the healthcare system perspective (direct costs of hospitals and insurance) to a societal perspective, incorporating non-medical expenditures by patients and their families (transportation, care), lost productivity, and the burden on caregivers. For one-time curative therapies such as CGT, the societal valuation framework often yields a more favorable cost-effectiveness ratio than traditional CEA, as it includes values such as “patients returning to work” and “the elimination of the family caregiving burden” in its calculations.
Table 2-5: Roadmap of HEOR Evaluation Framework Evolution
| Framework Type | Core Metrics | Key Advantages | Limitations | Applicable Scenarios |
| Traditional CEA | QALY/ICER | Standardized and highly comparable | Underestimates the value of rare diseases; does not include the equity dimension | Chronic and common diseases |
| Distributional CEA | QALY allocation + equity weights | Demonstrates the equity impact of interventions | High data requirements; policy application is not yet mature | Equity-sensitive diseases |
| Social Value Framework | Social cost-effectiveness ratio | Comprehensively reflects overall economic value | Data collection is complex and difficult to standardize | CGT, one-time curative therapies |
| Patient-centered framework | PROs, patient preferences | Reflecting patients’ true value judgments | Difficult to quantify; HTA acceptance varies widely | Rare Diseases, Pediatric Diseases |
For industry professionals, the key recommendation is: do not wait for HTA agencies to mandate a specific framework before beginning preparations; instead, proactively design a multidimensional HEOR evidence package based on product characteristics and target markets during the early clinical stages. At ISPOR 2026, the latest application cases of these frameworks will be extensively presented, offering a rare opportunity to catch up and recalibrate understanding.
3.0. biotech conference washington dc: ISPOR 2026 Core & “Hidden” Agenda Highlights — Focus on Actionable Insights

3.1 Plenary Sessions and Policy Dialogues: CMS/FDA Signals and Value Framework Applications
What does hosting the event in Washington, D.C. mean? It means policymakers from agencies such as CMS (Centers for Medicare & Medicaid Services), the FDA, and PCORI can attend the plenary sessions without having to fly in. Among all past ISPOR conferences, the density of policy signals at the Washington, D.C. venue is unmatched by any other city.
Pay close attention to the plenary speaker lineup, particularly Inmaculada Hernandez (former Senior Advisor at the FDA and a key figure in real-world evidence policy) and Indranil Bagchi from GSK’s Global Health Economics Department—the latter offering a frontline perspective on “how multinational pharmaceutical companies are reshaping the value narrative of innovative therapies under policy pressure.” Public statements from such figures often serve as crucial signals for understanding the boundaries of the regulatory-industry dynamic.
Regarding the application of value frameworks, discussions at the plenary session are expected to focus on two key issues: first, whether the ICER (Institute for Clinical and Economic Review) framework can be adapted to accommodate the one-time cure nature of CGTs; and second, whether global HTA harmonization (the similarities and differences between the frameworks of ICER, NICE, G-BA, and HAS) might see a landmark convergence in 2026–2027.
The extended value of the Washington/East Coast policy and economic network should also not be overlooked. Attending ISPOR is not merely about listening to presentations; it is a rare opportunity to engage with CMS drug policy officials, FDA CDER economic advisors, and CMS team members currently leading the practical implementation of IRA negotiations—individuals who typically require significant time to schedule meetings with. For teams responsible for market access strategies, the value of policy insights gained through these informal interactions often far exceeds that of the formal sessions themselves.
Table 3-1: Matrix of Policy Signals to Watch at the ISPOR 2026 Annual Meeting
| Policy Topic | Focus | Impact on ADCs/CGTs/Nucleic Acids | Recommended Conference Actions |
| IRA Drug Price Negotiations | Medicare Negotiation Trigger Mechanism and Innovative Drug Pricing | Potential to Squeeze Profit Margins for High-Priced Innovative Therapies | Understanding Negotiation Exemptions and Value-Based Requirements |
| CMMI New Payment Model | Latest Developments in the CGT Access Model | Impact on CGT Reimbursement Pathways and OBC Design | Assessing Whether the CMMI Model Is Suitable for Your Product |
| RWE Regulatory Guidelines | Updates to the FDA RWE Framework | Factors Affecting Whether RWE Can Be Used as Regulatory-Grade Evidence | Focus on criteria for eligibility to generate evidence |
| Global HTA Harmonization | Similarities and Differences in ICER/NICE/HAS Frameworks | Impact on the Consistency of Global Market Access Strategies | Planning Common Elements for Multi-Country HEOR Evidence Packages |
3.2 High-Value Short Courses and Workshops
Short Courses are ISPOR’s most distinctive learning format—compact in duration (typically 3–6 hours), led by leading practitioners in the field, and focused directly on market access challenges rather than general academic reviews. The following courses are particularly worth prioritizing for registration in 2026:
3.2.1 Applications of Generative AI in HEOR (Introductory + Advanced Dual-Track)
“Introduction to Applied Generative AI for HEOR” and “Applied Generative AI for HEOR: Advanced Architectures” form a dual-track system. The introductory course focuses on the foundational applications of LLMs (Large Language Models) in automating literature reviews, assisting with PRO scale design, and cost-effectiveness modeling, making it suitable for HEOR professionals who have not yet systematically explored AI tools.The advanced course delves into the implementation of RAG (Retrieval-Augmented Generation) and agent systems within HEOR evidence generation pipelines, as well as the compliance applications of AI model interpretability in regulatory submissions—providing direct decision-making insights for teams considering the integration of AI tools into product approval processes. It is particularly recommended for: teams exploring how AI can reduce HEOR research costs, or those needing to demonstrate the feasibility of AI tool applications to internal decision-makers.
3.2.2 Using Real-World Evidence (RWE) to Assess the Value and Affordability of Cell and Gene Therapies
“Using RWE to Inform the Value and Affordability Assessment of Cell and Gene Therapies” — This is one of the most directly valuable short courses for CGT market access teams in 2026. The course will systematically break down: how to use patient registry data to describe the long-term benefit curve of CGTs; how to use EHR data to construct natural history controls; and how to design RWE monitoring systems to support outcome assessment clauses in OBC contracts.Highly recommended for: teams responsible for market access, medical affairs, and health economics for CAR-T and gene therapy products.
3.2.3 Strategic Application of PROs in Clinical Trials
“PROs in Clinical Trials: Endpoint Selection, Regulatory Strategy, and Label Claims” — The importance of Patient-Reported Outcomes (PROs) in high-cost innovative therapies is rapidly increasing. Payers are increasingly requiring companies to provide evidence that “patients themselves perceive the treatment as effective,” rather than relying solely on clinical metrics assessed by physicians. This course will provide comprehensive guidance on the entire process of PRO endpoints, from design to regulatory submissions and HTA claims.Highly recommended for: clinical development teams, medical affairs, and research teams designing Phase II/III endpoints for CGT/ADC products.
3.2.4 Societal Valuation of Innovative Medicines
“Societal Valuation of Innovative Medicines” — This course will systematically introduce how to monetize “soft values” such as caregiver burden, lost productivity, and social support needs beyond the traditional health economics perspective, and integrate them into HTA submission materials. For CGTs and gene therapies, such societal value arguments are often critical supplementary evidence for persuading payers to accept high pricing.
3.2.5 Developing Decision-Grade Real-World Evidence
“Developing Decision-Grade Real-World Evidence” — Unlike “observational studies,” Decision-Grade RWE adheres to explicit research design standards, data quality requirements, and methodological guidelines, making it suitable for inclusion in regulatory submissions or HTA dossiers. This course provides framework tools directly applicable to projects and is particularly suited for CRO partners and HEOR research teams at pharmaceutical companies.
Table 3-2: 2026 ISPOR Key Short Course Recommendations and Attendee Matching Guide
| Course Title | Core Content | Ideal Attendees | Direct Relevance to High-Cost Innovative Therapies |
| Generative AI for HEOR (Introductory/Advanced) | Applications of LLM/RAG in HEOR Modeling and Reviews | HEOR Analysts, Market Access Strategy Teams | AI Tools to Reduce Evidence Generation Costs for ADCs and CGTs |
| RWE for CGT Evaluation | Long-term benefit modeling, natural history definition, and OBC outcome monitoring | CGT Market Access and Medical Affairs Teams | Core Evidence Tools for CAR-T/Gene Therapy Reimbursement Negotiations |
| PROs Strategy | PRO endpoint design, regulatory submissions, HTA statements | Clinical Development, Medical Affairs | ADC/CGT Patient-Centered Value Framework Development |
| Social Value Assessment | Monetization of Caregiver Burden and Productivity Loss | HEOR Researcher, Health Economics Consultant | Demonstrating the Social Value of a One-Time Cure with CGT |
| Development of Decision-Support RWE | RWE Design Standards, Data Quality, Regulatory Acceptability | CROs, Corporate HEOR Teams | Regulatory-Grade RWE to Support ADC/Nucleic Acid Drug Submissions |
3.3 Special Sessions and Cross-Disciplinary Discussions: Digital Tools, Early Payer Engagement, and Equity-Informative HTA
Beyond the high-profile plenary sessions and popular short courses, ISPOR’s most valuable content is often found in the “hidden agenda”—those panel discussions and Issue Panels that don’t make the front page of promotional materials but bring together the field’s true decision-makers.
AI-driven automated literature reviews and systematic reviews are reshaping the timeline for evidence synthesis.Traditional systematic reviews take 3–6 months to complete, whereas LLM-based automated screening tools can compress the initial screening phase to just a few days. ISPOR 2026 will showcase multiple AI-driven review tools already deployed in real-world access projects, along with regulatory agencies’ criteria for evaluating the acceptability of these tools’ outputs. For teams striving to catch up with competitors’ evidence packages, this represents a technical insight that can be immediately put into practice.
Synthetic Control Arms (SCAs) are gaining increasing prominence in HTA submissions for single-arm clinical trials. When a rare disease or CGT product cannot design a traditional RCT control group due to ethical or feasibility constraints, SCAs—which construct “hypothetical controls” using historical data and statistical methods—have become an accepted means of supplementary evidence for some regulatory and HTA review bodies.ISPOR 2026 is expected to showcase multiple real-world case studies of CGT and nucleic acid therapeutics utilizing SCAs, as well as variations in the acceptance criteria for SCAs across different HTA agencies.
Early Payer Engagement is another under-the-radar topic worth exploring this year. Traditionally, companies engage in formal pricing negotiations with insurers only after Phase III completion; however, for high-cost therapies such as CGT, payers wish to participate in discussions regarding the design of value frameworks and outcome assessment metrics as early as the Phase II stage. This “early co-creation” model has been piloted in some European HTA systems, and its path to adoption in the United States is expected to be discussed in depth at ISPOR.
Equity-Informative HTA is one of the fastest-growing methodological trends over the past three years. Both Canada’s CADTH and the UK’s NICE have incorporated some form of equity analysis requirements into their respective frameworks, while the U.S. ICER is also exploring how to systematize equity considerations.Based on our practical experience in these projects, companies that proactively present “subgroup effect differences” and “access improvement plans” in their access submissions often gain greater initiative in HTA discussions, rather than passively responding to questions from review agencies.
Table 3-3: Key Insights and Practical Applications from the ISPOR 2026 “Hidden Agenda”
| Topic | Core Insight | Practical Applications |
| AI-Automated Literature Reviews | LLM tools can reduce the time required for the initial screening of systematic reviews from months to days; regulatory acceptance criteria are currently being established | Evaluate and adopt AI-powered review tools to accelerate the development of evidence packages |
| Synthetic Control Arm (SCA) | Multiple HTA agencies have accepted SCAs as supplementary evidence for single-arm CGT trials, and methodological standards are gradually converging | Assess the feasibility of incorporating SCAs when designing clinical evidence strategies for nucleic acid/CGT products |
| Early payer engagement | Engage insurers starting in Phase II to co-design value frameworks, shortening market access negotiation cycles | Develop an early-stage payer advisory board strategy and design outcome assessment metrics |
| Equity-Informative HTA | Canada, the UK, and ICER are all introducing equity analysis requirements; proactive disclosure of subgroup efficacy differences is becoming a trend | Include an equity section in the HEOR evidence package and proactively present data on accessibility |
| Multi-stakeholder Value Assessment | A joint value assessment model involving patient organizations, caregiver representatives, and payers is gaining traction | Establishing a collaborative mechanism for creating a multi-stakeholder value framework |
4.0. biotech conference washington dc: Practical Conference Guide — Maximize Your Takeaways Like a Seasoned Industry Veteran
4.1 Pre-Conference Preparation: Selecting Tracks Suited to Your Background and Researching Speakers

The biggest efficiency killer at ISPOR is failing to clarify beforehand, “What problem am I here to solve?” With three days and hundreds of sessions, without pre-screening based on specific goals, it’s easy to fall into a fragmented state where every session feels somewhat relevant but none is fully understood.
Below are personalized preparation recommendations for attendees with different backgrounds:
Pharmaceutical Company Market Access Teams: Focus on pricing and RWE sessions related to your therapeutic area. Review the speakers’ latest published literature in advance (Google Scholar / PubMed), and prepare questions specific to your pipeline’s indications (e.g., “Our Phase II data has an OS follow-up of only 18 months—what is the HTA agency’s threshold for accepting extrapolation models?”).
CRO/Consulting Firm Colleagues: Treat this conference as an opportunity for intensive intelligence gathering. Monitor the evolving evidence standards for competitive technologies, document the latest statements from regulators and payers, and compile these into industry briefings to share with your internal teams and clients. Also, seize opportunities for informal discussions with pharmaceutical companies’ market access teams to understand where their most pressing evidence gaps currently lie.
Academic institutions and payer representatives: Focus on the latest advancements in methodological frontiers (distributional CEA, societal valuation, AI-driven HEOR tools) and policy developments regarding multinational HTA coordination mechanisms. Actively participate in the discussions and interactions during the Issue Panels, as these are highly effective venues for building academic networks.
Table 4-1: Guide to Selecting Tracks Based on Attendee Background
| Attendee Background | Preferred Session Types | Key Speaker Types to Focus On | Pre-Conference Preparation |
| Pharmaceutical Companies: Market Access | ADC/CGT Pricing Sessions, RWE Workshops | CMS representatives, ICER analysts, competitive product teams | Review the target speakers’ latest publications and prepare specific questions |
| CROs/Consulting Firms | Methodological Innovation Sessions, Issue Panels | Academic Methodology Experts, Payer Representatives | Organize competitive intelligence frameworks and design intelligence gathering templates |
| Researcher at an academic institution | Frontier Methodology Session, Plenary Presentation | Leading HEOR Scholars and Policy Researchers | Browse the latest articles in the ISPOR journal to prepare topics for academic discussions |
| Payer/HTA representatives | Value Framework discussions, Equity HTA | Pharmaceutical company market access strategy teams, HEOR consultants | Stay informed on the latest trends in evidence design for pharmaceutical companies’ market access submissions |
Speakers of particular interest: Dawn Hershman (expert in the intersection of oncology and HEOR) and Stacie B. Dusetzina (Vanderbilt University, a leading scholar specializing in drug access and pricing research). Their sessions often focus on real-world access issues for high-cost innovative drugs, providing firsthand insights into the practical pressures of the U.S. payer ecosystem.
4.2 Conference Strategy: Value Exchange in Networking, Prioritize Short Courses and Issue Panels
The biggest misconception about networking at conferences is equating “exchanging business cards” with “building relationships.” In high-density professional settings like ISPOR, truly effective networking is a “value exchange”: Before you arrive, clearly define what you can offer the other person (a unique data insight in a specific field, a problem framework not yet widely discussed, or an invitation for a follow-up in-depth conversation)—rather than simply handing over a business card and saying, “Nice to meet you.”
Short Courses and Issue Panels are the two most efficient types of events. The benefits of Short Courses include: concentrated time slots, comprehensive content, and instructors who are typically practitioners in the field rather than purely academic scholars; the quality of interaction during the Q&A session is far superior to that of large plenary sessions. Issue Panels, on the other hand, focus on controversial topics, presenting multiple perspectives on the same stage, making them an excellent venue for understanding the “true thoughts of different stakeholders beyond the mainstream consensus.”
How can you find potential CRO or consulting partners on ISPOR’s Social Media Wall and app? We recommend actively searching attendee profiles using specific keywords (such as “CGT value framework” or “real-world evidence ADC”) and scheduling a 15-minute coffee meeting in advance—this is often far more efficient than making impromptu connections on the conference floor.
Take full advantage of Washington’s geographical advantages: the area surrounding the Pennsylvania Convention Center offers numerous venues suitable for small meetings. Since CMS officials and congressional policy advisors are based in DC, arranging an informal lunch meeting during ISPOR is far easier than usual. Make the most of this window of opportunity to connect with key policymakers who typically require formal appointments to meet.
Table 4-2: Recommended Time Allocation for the Conference (Optimal Strategy for a 3-Day Event with 5,000 Attendees)
| Activity Type | Recommended Allocation | Key Takeaways | Considerations |
| Short Courses | 30–35% | Mastery of systematic methodologies, acquisition of practical tools | Register early—popular courses often sell out quickly |
| Issue Panels | 20–25% | Gain insights from multiple perspectives and identify policy signals | Select controversial topics relevant to your product pipeline |
| Full Plenary | 10–15% | Macro trends, policy signals, industry consensus | Ideal for capturing the conference’s core narrative; full attendance is not required |
| Networking/Social Events | 20–25% | Identifying partnership opportunities, connecting with CROs/consultants | Attend with specific questions in mind; avoid general networking |
| Poster Session | 10–15% | Latest research data, competitive intelligence gathering | Mark posters of interest in advance; authors’ time on-site is limited |
4.3 Report Interpretation and Implementation Techniques: Extracting Decision-Making Insights from Complex Models
The technical barriers of HEOR research can sometimes become obstacles for executives and investors seeking insights—a densely packed Markov model transition matrix may actually conceal a very simple strategic conclusion. How can you quickly extract the information you need from complex posters and reports?
We recommend a three-step extraction method:
- Step 1—Ask “So What”: After reading an HEOR report, ask yourself, “What does this conclusion mean for the product I’m responsible for?” If a real-world evidence (RWE) study for an ADC product shows that the median overall survival (OS) in the real world is 20% lower than in an RCT, this means your HTA extrapolation model needs to incorporate a real-world discount factor, and you’ll need to prepare a set of arguments for reimbursement negotiations to address the issue of “discounted real-world efficacy.”
- Step 2 — Identify the “Key Assumptions”: Any cost-effectiveness model is built on a set of key assumptions (discount rate, choice of OS extrapolation model, source of utility values). At the meeting, what you should focus on most is not the model’s ICER conclusion, but rather “what assumptions they used, why they chose those assumptions, and how much a change in these assumptions would alter the conclusion, as shown by sensitivity analyses.”
- Step 3 — Translate into an Internal Reporting Framework: Within 48 hours of the meeting, distill the key takeaways into a briefing of no more than one page. Recommended format: Background (Why we are focusing on this topic) → Key Findings (3–5 most important insights) → Impact on Our Pipeline (specific implications for action) → Recommendations for Next Steps. The audience for this briefing is not HEOR experts, but team leaders with decision-making authority.
Table 4-3: Quick Checklist for Identifying Key Assumptions in the HEOR Model
| Model Elements | Key Issues to Address | Impact on Access Decisions |
| Survival Extrapolation Model | The selected parameter distribution (exponential/Weibull/log-logistic) and the reasonableness of the extrapolation period | Different extrapolation assumptions can result in ICER differences of 50% or more |
| Source of utility values | Which population does the EQ-5D data come from? Is there cross-validation with PRO data? | Overestimated utility values may artificially inflate QALY benefits |
| Discount rate assumptions | Are the discount rates used for costs and benefits consistent with the target country’s HTA standards? | Discount rates influence the cost-effectiveness analysis of long-term therapies |
| Selection of comparators | Is the comparator a true standard of care, or was a comparator with weaker efficacy deliberately selected? | The reference baseline that directly determines the ICER |
| Real-world discount | Assumptions regarding the translation of RCT efficacy to the real world Is the discount factor reasonable? | This directly influences payers’ assessment of the model’s credibility |
4.4 Recommendations for Collaboration Between the Washington On-Site Network and the East Coast
For teams planning a global footprint, Washington is a particularly strategic hub city.Not only does ISPOR itself take place here, but a large number of events organized by federal agencies, think tanks, and patient advocacy organizations also occur during the same timeframe before and after the conference. By fully utilizing the ISPOR travel and hotel budget to arrange a one-day “policy visit day” before or after the conference—visiting representatives from PCORI (Patient-Centered Outcomes Research Institute), PHRMA (Pharmaceutical Research and Manufacturers of America), or relevant patient organizations—you can significantly enhance the intelligence and networking value of a single trip.
If your team also has operations in Boston (biotechnology R&D), Washington offers an excellent opportunity to shift your “R&D perspective” to an “access perspective”: while discussions in Cambridge/Kendall Square focus on scientific feasibility, conversations in Washington center on whether “this scientific breakthrough has a sustainable pathway to reimbursement.” The sooner you make this shift in perspective, the better.
Tip: The ISPOR conference app typically allows you to browse attendee profiles. Once this feature is available, we recommend using the filter criteria “Director level or above, from pharmaceutical company access/HTA departments” to pre-select 30–50 target contacts. Spend a week using LinkedIn or the conference messaging system to schedule 15-minute coffee meetings. Securing these meetings in advance is 3–5 times more efficient than relying on chance encounters at the event.
5.0. biotech conference washington dc: Global Outlook — Lessons from China/Asia for Innovative Drug Expansion
5.1 The Applicability of U.S. HEOR Experience in Global Market Access
For teams planning global expansion—especially those preparing to bring Chinese ADCs, nucleic acid therapeutics, or CGT products to markets in Europe, the U.S., Japan, and South Korea—ISPOR 2026 offers an exceptionally rare opportunity to “calibrate their reference framework.”
The strength of the U.S. HEOR system lies in its highly standardized methodological framework (ICER, QALY) and robust real-world data infrastructure (commercial claims databases, EHR networks, patient registries). While this system offers profound insights for establishing a high-quality value framework for innovative therapies, directly transplanting it to Asian markets presents the following key gaps:
First, there are significant differences in the review criteria and decision-making frameworks of HTA agencies.The UK’s NICE, Canada’s CADTH, Germany’s G-BA, and France’s HAS differ in methodological preferences, QALY thresholds, and the types of evidence they accept. The standards of Japan’s PMDA and the National Health Insurance Bureau also differ fundamentally from those in the U.S. An evidence package that passes ICER review in the U.S. cannot be directly applied to European HTA. For teams hoping to reduce HEOR costs by “reusing a single dataset across multiple countries,” this is a risk factor that requires early, unified planning.
Second, the availability and quality of RWD (Real-World Data) vary significantly across markets. The U.S. possesses the world’s most comprehensive commercial claims databases and the most mature EHR interoperability system. China’s RWD infrastructure is rapidly developing, but compared to the U.S., it still faces challenges such as low data standardization and difficulties in cross-hospital data exchange. This means that RWD accumulated in China often requires additional data quality validation efforts if it is to be used to support HTA in the U.S. or Europe.
Third, the maturity and influence of patient advocacy organizations in Asian markets lag significantly behind those in the U.S. In the U.S., patient organizations have become a force to be reckoned with in HEOR framework design, HTA dialogues, and pricing negotiations. In Asian markets, while patient organizations are gradually growing in strength, institutional mechanisms for their participation in market access processes still require time to establish. Establishing partnerships with patient organizations early on is an often-underestimated strategic investment for innovative Asian pharmaceutical companies expanding into international markets.
Table 5-1: Comparison of Methodological Frameworks in Major HTA Markets (U.S. Perspective)
| Market/Organization | Core Evaluation Framework | QALY Threshold | RWE Acceptability | Equity Considerations |
| United States (ICER) | Cost-Effectiveness + Budget Impact | $100,000–$150,000 per QALY (reference value) | Limited adoption; regulatory standards are currently being established | Limited inclusion; policy implementation underway |
| United Kingdom (NICE) | Cost-effectiveness (QALY-based) | £20,000–£30,000/QALY (threshold) | Explicit acceptance; dedicated RWE guidelines available | Preliminary inclusion in the MCDA framework |
| Canada (CADTH) | Clinical benefit + cost-effectiveness | CAD$50,000/QALY (reference value) | Accepted, subject to meeting quality standards | Equity analysis has become standard practice |
| Germany (G-BA) | Additional clinical benefit grading | No uniform QALY threshold | Limited acceptance; strict methodological requirements | Equity has been incorporated into discussions in recent years |
| Japan (MHLW) | Cost-effectiveness analysis (pilot phase) | Approx. ¥5 million–7.5 million per QALY | Standardization of RWE applications is still evolving | Limited systematic consideration |
| China (National Healthcare Security Administration) | Clinical benefits are the primary focus; economic evaluation is used as a reference | Approx. ¥400,000–600,000/QALY (for reference) | Accelerated use of RWD; standards are still evolving | Relevant policy frameworks are being established |
5.2 Synergistic Pathways for RWE, Patient-Centered Evidence, and Multinational HTA
For teams planning global market access, the most critical insight is: “A multi-country HTA submission package is not a mere aggregation of separate documents; rather, it requires systematic evidence architecture design from the very beginning of the project.”
At the RWE level, the core of a collaborative strategy is “shared core data with differentiated analytical frameworks”: designing an RWE study protocol that simultaneously meets the requirements of the FDA, EMA, and NMPA. Although this increases upfront design costs, it can significantly reduce the overall cost of conducting duplicate independent studies. This requires inviting HEOR consultants from multiple markets to participate jointly during the study design phase, using the “intersection of the most stringent requirements” as the standard for data collection.
Regarding patient-centered evidence, the cross-cultural validation of PRO (Patient-Reported Outcome) scales is an often-overlooked task. An EQ-5D scale validated in the U.S., if applied directly to Chinese patients, requires additional validation of the cultural appropriateness of its utility values. Preparing PRO tools in multiple languages and cultural contexts as early as the Phase II stage will save a significant amount of time for subsequent HTA submissions.
Regarding equity and accessibility, the Chinese and Asian markets present unique opportunities for storytelling. When an innovative therapy claims that its global access strategy prioritizes “accessibility,” whether it can achieve genuine accessibility in the Chinese market through tiered pricing or assistance programs often serves as a key indicator for international HTA agencies to assess the credibility of the company’s commitments. Such “equity actions in practice” not only hold public relations value but also carry substantial global HTA credibility.
Table 5-2: Framework for a Multi-Country HTA Collaborative Evidence Strategy
| Evidence Type | Collaborative Strategy | Points of Differentiation | Action Recommendations |
| RWE Study Design | Core Design Meets the Most Stringent Market Requirements (FDA + EMA) | Regulatory compliance and data localization across countries | Establish a multinational HEOR joint research team |
| PRO Scales | Use of internationally recognized scales (EQ-5D, SF-36) | Cross-cultural utility value validation and localized standard adjustments | Initiating multilingual PRO validation during Phase I/II |
| Cost-Utility Model | Development of a generic framework with adjustable parameters | Differences in QALY thresholds, discount rates, and cost inputs across countries | Design of a “modular” cost-effectiveness model to support rapid localization |
| Equity Analysis | Establishing a foundational equity analysis framework | Differences in equity-related regulations and HTA requirements across countries | Identify equity-related market access requirements on a market-by-market basis and prepare data in advance |
6.0. biotech conference washington dc: Conclusion — Take Action to Shape a Sustainable Future for Innovation
6.1 Trend Summary: HEOR as the Deciding Factor in the Competition for High-Cost Innovative Therapies

If we distill all discussions from ISPOR 2026 into a single, essential statement, it is this: clinical data alone is no longer sufficient for high-cost innovative therapies to gain a foothold in the market.
We are at a turning point: the pace of scientific breakthroughs in ADCs, CGTs, and nucleic acid therapeutics far outstrips the pace of evolution in reimbursement systems. The direct consequence of this gap is that even if a therapy’s clinical data is flawless, it may still suffer a crushing defeat at the access negotiation table due to an inability to demonstrate economic value, reach a consensus of trust with payers, or pass health equity reviews.
The role of HEOR is precisely to build a trusted bridge between scientific breakthroughs and sustainable reimbursement.At the Policy level, HEOR provides the evidence base to support value-based pricing; at the Access level, HEOR quantifies barriers to access for innovative therapies and designs solutions; at the Value level, HEOR translates the multidimensional benefits to patients, families, and society into a monetized value narrative that payers can accept. The synthesis of these three elements is the defining challenge of our era, as represented by ISPOR 2026.
This 2026 conference in Washington comes at just the right time. It is not a mere routine industry gathering, but a strategic venue where practitioners of high-cost innovative therapies collectively chart the course for competitive success. The conceptual frameworks you establish here, the partnerships you forge, and the evidence-generation methodologies you bring back will continue to play a pivotal role in market access competition over the next 2–3 years.
6.2 Call to Action: Go to Washington with Questions, Return with a Framework
If there’s one thing you should take away from this article, it’s this: before booking your flight and hotel, write down the three questions you most want to find answers to in Washington.
It could be “Which OBC structure should our CGT product adopt?” or “What does the most payer-accepted HEOR evidence package currently look like for first-line ADC use?” or even “How do we design a multinational RWE study protocol that works in both the U.S. and Europe?” By going with these specific questions, you’ll find that every session and every coffee conversation takes on a clear sense of purpose.
The topics covered in this article are the ones we’re asked most frequently in our daily market access consulting work: ADC pricing negotiations, CGT reimbursement barriers, the logical leap from rare diseases to large patient populations for nucleic acid therapeutics, and the integration of AI and RWE tools in evidence generation…If these topics are highly relevant to your work, please share the innovative therapy issues you’re most interested in in the comments section, or let us know which topics you plan to focus on at the Washington event.
See you in the blog comments section, or in person in Washington this May. HEOR is helping us shape a sustainable future of innovation that delivers scientific breakthroughs while ensuring treatments are truly affordable for patients—a mission every professional in the field should take seriously.
7.0. biotech conference washington dc: Frequently Asked Questions (FAQ)

(The following FAQs are provided for readers’ reference; the English translation will be used for Google’s “People Also Ask” search visibility)
FAQ 1: Is the bar high for attending ISPOR 2026? Who is it suitable for?
ISPOR caters to a diverse audience, including market access teams at pharmaceutical companies, medical affairs professionals, CROs and HEOR consulting firms, academic institutions, payers (public and private health insurance), and patient organizations.Whether you are a novice analyst or a seasoned access director, ISPOR offers learning and networking resources tailored to your level of experience—provided your work involves demonstrating the value of innovative therapies, pricing negotiations, or market access. For first-time attendees, we recommend prioritizing registration for Short Courses as a systematic introduction to the field.
FAQ 2: What key discussions related to ADC drug market access will be featured at the 2026 ISPOR conference?
The 2026 ISPOR conference is expected to feature multiple sessions focusing on the pricing rationale for ADCs, the application of real-world evidence (RWE) in validating real-world efficacy, and incremental cost-effectiveness analyses for expanding from second-line to first-line treatment. Of particular note are the methodological applications of synthetic control arms in single-arm ADC studies, as well as payers’ perspectives on the credibility of the evidence-supplementation strategy that “RWE compensates for the time constraints of RCTs.”
FAQ 3: How can the high pricing of CGT (Cell and Gene Therapy) be accepted by payers?
Currently widely discussed and implemented approaches include Outcome-Based Contracts (OBCs), staged annuity payments, and new federal payment models promoted by CMS through the CMMI framework. The key lies in demonstrating the sustainability of long-term benefits through high-quality RWE, while jointly designing outcome assessment metrics with payers (rather than unilaterally establishing them). Relevant short courses at ISPOR 2026 will provide systematic answers to this issue.
FAQ 4: How can insights from ISPOR 2026 be translated into internal decision-making support?
We recommend a “three-step translation method”: record key takeaways during the conference (not the full volume of information); within 48 hours of the event, compile them into a briefing of no more than one page (background → findings → implications for the pipeline → action recommendations); and hold an internal sharing session within two weeks. The key is to translate HEOR’s technical language into a framework that executives can directly use for decision-making, rather than simply forwarding the conference slides verbatim.
FAQ 5: What insights can China and Asia gain from U.S. HEOR experience regarding the global expansion of innovative drugs?
There are three key takeaways: First, early involvement in HEOR evidence planning (designing RWE studies and PRO tools starting in Phase II); second, designing a multi-country compatible evidence framework (collecting data using the “strictest requirement” standard to reduce duplication costs across markets); and third, establishing partnerships with patient advocacy organizations early on (particularly critical in Asian markets, as this is a strategic asset for market entry that companies expanding overseas often underestimate).







