- 1.0. biotech events san diego: Introduction: 2026—The First Year of Execution: Moving from "Telling Stories" to "Delivering Results"
- 2.0. biotech events san diego: 2026 Golden Track Map: Modality Convergence and Cross-Boundary Opportunities in the BIO Agenda
- 3.0. biotech events san diego: 70 Days Before the Event: Biopartnering and the Extreme Optimization of Personal ROI
- 4.0. biotech events san diego: On-Site Execution: Efficiently Identifying High-Value Signals Amid the Noise of 20,000 People
- 5.0. biotech events san diego: Post-Event Follow-Up: Quickly Turn Business Cards into Tangible Business Progress
- 6.0. biotech events san diego: Conclusion: BIO 2026 is not the end goal, but a purpose-driven long-term lever
- 7.0. biotech events san diego: Appendix: Frequently Asked Questions (FAQ)—A Practical Guide to Attending BIO 2026
1.0. biotech events san diego: Introduction: 2026—The First Year of Execution: Moving from “Telling Stories” to “Delivering Results”

As one of the most anticipated biotech events san diego offers each year, BIO 2026 brings together 21,000+ global biopharma leaders. If you heard someone say, “We’ll be able to submit an NDA next year,” at the 2023 BIO conference, most people would have responded with a polite nod, already mentally bracing themselves for yet another “story.” Three years later, the situation is completely different.After the mRNA frenzy cooled, the GLP-1 sector deflated, and the ADC target space became oversaturated, the capital markets have recalibrated their expectations—no longer paying for “pipelines on PowerPoint slides,” but only writing checks for assets that “deliver on their data.”The 2026 BIO International Convention is not just another industry gala, but a true “midterm exam.”
This article is not a traditional conference guide.We do not intend to list daily sessions in chronological order, nor do we plan to simply copy and paste the official agenda for our readers. We have only one goal: to help professionals at the San Diego venue—those with specific business objectives (BD licensing, fundraising, supply chain partnerships, regulatory insights)—make the most of the next 70-day preparation window, find their precise coordinates among the 21,000 attendees, and return home with tangible results.
1.1 2026 Macro Landscape: The “Data Realization” Window for ADCs, CGT, and Nucleic Acid Therapeutics Amid a Rational Rebound in Capital
In 2025, global biopharma M&A volume will return to the $150 billion range, rebounding by approximately 40% from the 2023 trough; however, buyer logic has undergone a fundamental shift.BD teams at major pharmaceutical companies (MNCs) are no longer keen on “buying concepts,” but are instead focusing their efforts on three areas: ADC assets that have entered Phase II clinical trials and possess preliminary proof-of-concept (POC) data; CGT projects with a clear path to clinical benefits; and small nucleic acid drugs with validated delivery technology platforms. This means that 2026 is a critical juncture for these three sectors to “deliver results” to capital markets and strategic partners.
Specifically in the ADC sector, the success of Enhertu (DS-8201) has raised the competitive bar for mature targets like TROP2 and HER2 to a level where “latecomers must demonstrate at least non-inferiority.”At the 2026 BIO ADC session, the focus of discussion will no longer be “whether we can develop ADCs,” but rather “in which indications does our linker-payload combination offer a truly differentiated advantage.” This is particularly important for Chinese players—over the past five years, Chinese ADC outbound transactions have accounted for approximately 35% of global ADC business development deals, but the clinical data for most licensed assets has been insufficient, leading to significantly lower valuations during later-stage negotiations.
The CGT sector faces similar data pressures. The FDA has significantly tightened its review standards for CGT products between 2024 and 2025, lowering the ICER threshold from $150,000 to $100,000 per QALY, which directly impacts payers’ reimbursement expectations.The 2026 BIO CGT session will focus heavily on “how to demonstrate therapeutic value in the real world,” rather than merely presenting Phase I clinical safety data. For LNP platform companies focused on in vivo delivery, progress in extrahepatic delivery will be the key highlight—if targeted delivery data to non-liver tissues is presented at BIO, it will be one of the most significant signals of the entire conference.
The core challenge in the nucleic acid therapeutics sector (siRNA, ASO, mRNA) has shifted from “drug-ability” to “scalability.” Alnylam’s siRNA technology platform has validated the commercialization pathways for multiple drugs, but delivery costs, health insurance reimbursement, and patient access remain unresolved commercial challenges.The 2026 BIO Nucleic Acid Therapeutics Forum is expected to focus on cost-reduction strategies for N-acetylgalactosamine (GalNAc) conjugation technology, as well as the potential for LNP delivery platforms to expand into extrahepatic tissues. For Chinese nucleic acid therapeutics companies, BIO 2026 presents an excellent opportunity to engage with established players such as Wave Life Sciences, Ionis, and Sanofi.
[Table 1-1] Comparison of Global Biopharmaceutical BD Transaction Trends, 2024–2026
| Indicator | 2024 | 2025 | 2026 (Forecast) | Trend |
| Global BD Total Transaction Value (USD billion) | 1,080 | 1,520 | 1,650 | ↑ Recovery but with greater rationality |
| Share of Transactions in the ADC Sector | 18% | 22% | 25% | ↑Continuing to rise |
| CGT Sector Transaction Share | 9% | 11% | 14% | ↑Regulatory pressure drives consolidation |
| Share of Transactions in the Nucleic Acid Drug Sector | 7% | 10% | 13% | ↑Breakthroughs in Delivery Technologies |
| Median Upfront Payment (in $10,000) | $2,100 | 2,800 | 3,200 | ↑Higher Data Threshold |
| Share of Chinese overseas asset transactions | 32% | 35% | 33% | [‘→ Stabilizing’, ‘↓ Impact of insufficient data’] |
[Interpretation of Table 1-1] As shown in the table above, the total value of global BD transactions is projected to exceed $165 billion in 2026, but the underlying driving force has shifted from “sector popularity” to “data validation capabilities.”The combined share of transactions in the ADC, CGT, and nucleic acid therapeutics sectors is projected to exceed 50%, indicating that these three sectors will be the undisputed focal points of BD discussions at the 2026 BIO conference. The share of outbound transactions involving Chinese assets is expected to remain flat or even decline slightly, primarily due to international buyers’ increasingly stringent requirements regarding the quality and integrity of Chinese companies’ clinical data—the logic of “delivering results” applies equally to Chinese companies expanding overseas.
[Table 1-2] Forecast of Core Topics for the 2026 BIO ADC/CGT/Nucleic Acid Therapeutics Sessions
| Therapeutic Area | Core Topics | BIO Session | Expected Interest |
| ADC | Differentiated Strategies for Next-Generation Payloads (TOP1i, PROTAC, BEC-1) | Oncology: Next-Generation ADCs | ★★★★★ |
| ADC | AOC (Antibody-Oligonucleotide Conjugate) Delivery Breakthrough | Platform Technologies | ★★★★☆ |
| ADC | Expansion Pathways for Non-Oncology Indications (Autoimmune, Metabolic) | Immunology & Metabolic Diseases | ★★★★☆ |
| CGT | In vivo LNP extrahepatic delivery clinical data | Gene Therapy & Genome Editing | ★★★★★ |
| CGT | Scaling Challenges for iPSC-Derived Universal Cell Therapies | Cell Therapy Manufacturing | ★★★★☆ |
| Nucleic Acid Therapeutics | Declining GalNAc Delivery Costs and Health Insurance Negotiations | Nucleic Acid Therapeutics Forum | ★★★★★ |
| Nucleic Acid Therapeutics | CMC and Commercialization Pathways for the AOC Platform | Business Development | ★★★☆☆ |
| Nucleic Acid Drugs | Advances in mRNA Cancer Vaccines and Therapeutic mRNA | Cancer Immunotherapy | ★★★★☆ |
1.2 San Diego’s Unique Appeal: Supply Chain Restructuring and California’s Biomedical Ecosystem Signals Behind the 21,000 Attendees
BIO’s selection of San Diego as the host city for 2026 was by no means a coincidence. As the core city of the world’s third-largest biopharmaceutical industry cluster, San Diego is home to approximately 1,200 life sciences companies, forming a complete ecosystem spanning from early-stage discovery to commercial production.The density of Big Pharma and biotech clusters—represented by companies such as Illumina, Biogen, BMS (La Jolla campus), Gilead, and Vertex—is second only to the San Francisco Bay Area on the U.S. West Coast, while the supply chain’s nearshoring advantages significantly outperform those of traditional East Coast clusters.
Since 2025, the global biopharmaceutical supply chain has been undergoing a dual restructuring toward “nearshoring” and “friendshoring.” The vulnerabilities of trans-Pacific supply chains exposed during the pandemic have prompted multinational corporations (MNCs) to begin shifting manufacturing bases for critical consumables and intermediates to allied nations and regions.San Diego’s geographic advantages align perfectly with this trend—forming a nearshore manufacturing network with the Baja California corridor in Mexico, where costs are approximately 30–40% lower than local production in California, while maintaining consistency in quality regulation.For Chinese CDMO companies, this implies that requests to establish “nearshore partnership” relationships—a model distinct from the traditional “Made in China, Sold Globally” approach—may increase during the 2026 BIO conference.
Beyond the figure of 21,000 attendees, what deserves greater attention is the shift in the attendee demographic. According to official BIO data, at the 2025 San Diego event, the proportion of business development and licensing professionals rose from 22% in 2023 to 28%, while the share of financing and investment professionals increased from 15% to 19%.The increased concentration of these two groups suggests that the networking intensity (deal-making efficiency) at BIO 2026 will be significantly higher than in previous years. For readers hoping to achieve specific business development goals at the event, this means you need to enter “operational mode” at least six weeks in advance—your Partnering Profile, target company list, and meeting schedule must be finalized by then.
[Table 1-3] Comparison of Key Metrics: San Diego vs. Other BIO Host Cities
| Dimension | San Diego (2026) | Boston (2024) | San Francisco (2023) | Advantage Analysis |
| Total Attendees | 21,600 | 19,800 | 20,200 | Largest |
| Percentage of BD/BD-Licensing Professionals | 28% | 24% | 25% | ↑Highest in San Diego |
| Investors/VCs | 19% | 18% | 17% | ↑Continuing to rise |
| Share of CDMO/Supply Chain Service Providers | 14% | 11% | 12% | ↑Driven by nearshore manufacturing |
| Number of local biotech companies in California | ~1,200 | ~900 | ~950 | Most in San Diego |
| Accessibility to nearshore manufacturing corridors | ★★★★★ | ★★☆☆☆ | ★★☆☆☆ | Unique to San Diego |
| Average Hotel Rate During the Conference (per night) | $280–350 | $320–$400 | $350–450 | San Diego is relatively affordable |
| Biopartnering system activity (2025 data) | 15,000+ searches per day | 12,000 daily | 13,000+ daily | ↑Continuing to grow |
[Interpretation of Tables 1–3] As the host city for BIO 2026, San Diego offers significant advantages in terms of the density of BD professionals and the nearshore supply chain ecosystem. For readers whose primary goal is BD licensing, San Diego’s conference efficiency (BD dialogue density per total attendee) is approximately 17% higher than that of Boston.Furthermore, the presence of top-tier research institutions such as the University of California, San Diego (UCSD), Scripps Research, and the Salk Institute means you have the opportunity to engage with a large number of translational medicine projects in the early discovery stage on-site—a unique value proposition that other host cities struggle to offer.
1.3 Core Value of This Article: A Perspective from a U.S. Biopharma BD Professional—Moving Beyond Generic Conference Summaries to Focus on How to Truly Capture Commercial Value at BIO 2026
The author’s stance is clear: we do not endorse BIO officially, we do not produce clickbait articles like “Top 10 Session Recommendations,” and we have no intention of telling you how exciting “AI-driven drug discovery” is—you can find that information for free on the official website. What we aim to do is more specific, more pragmatic, and more disruptive: help you maximize your ROI (Return on Investment) from BIO 2026 in just 70 days.
How do you measure the ROI of attending? For most readers, the answer boils down to four key outcomes:
① Closing at least one business development licensing or acquisition deal;
② Validate or refute your pipeline hypotheses through high-quality conversations;
③ Establish 3–5 contacts that can be developed into long-term partnerships;
④ Bringing back valuable industry insights that convince company management the trip was worth the expense.
What sets this article apart is our commitment to a “Modality Convergence” perspective. Cross-disciplinary insights spanning the four golden sectors—ADC, CGT, nucleic acid therapeutics, and GLP-1—often represent high-value opportunities that are easily overlooked by the average attendee.Technical expertise from an oral GLP-1 delivery platform could be precisely the key that nucleic acid drug companies need to solve their extrahepatic delivery challenges—this cross-modality connection is your core weapon for achieving differentiated insights at BIO.
In the following five chapters, we will provide actionable frameworks, templates, and checklists following a comprehensive timeline: “Pre-Event Preparation (Chapter III) → On-Site Execution (Chapter IV) → Post-Event Follow-Up (Chapter V).”We focus specifically on the unique challenges Chinese and Asian players face on the global stage—including practical details of IP protection, supply chain security in a geopolitical context, and how to use “purpose-driven” narratives to resonate with European and American partners accustomed to Western discourse.
[Table 1-4] Five Key Differences Between This Guide and Standard BIO Conference Guides
| Dimension | Standard BIO Conference Guide | This Article’s Methodology |
| Content Focus | Replication of session schedules + general recommendations | BD/Licensing Goal-Driven Execution Framework |
| Depth of Technical Analysis | Overview of the Industry Sector | “Map + Opportunity + Risk” Three-Dimensional Analysis, Including a Cross-Boundary Modality Integration Perspective |
| Practical Tools | Selected Lists | Partnering Profile Template + Information Gathering Template + Email Follow-up Template |
| Perspective for Chinese/Asian Readers | Virtually Ignored | Dedicated sections on IP protection, supply chain compliance, narrative differences, and other relevant topics |
| Pre-meeting/during-meeting/post-meeting coverage | Typically covers only pre-meeting | 70-day preparation → 4-day on-site event → 48-hour follow-up: a fully closed-loop process |
2.0. biotech events san diego: 2026 Golden Track Map: Modality Convergence and Cross-Boundary Opportunities in the BIO Agenda

Technological trends do not exist in isolation. Within the four golden tracks—ADC, GLP-1, CGT, and nucleic acid therapeutics—and across them, profound technological migration and paradigm convergence are taking place. Understanding these cross-modal connection points is key to achieving cognitive differentiation at BIO 2026.This chapter adopts a three-part structure—”Trend Analysis → BIO Agenda Mapping → Cross-Boundary Opportunity Extraction”—providing actionable on-site observation points for each track.
2.1 ADCs and Their Evolved Forms (XDCs): Expansion from Oncology to Autoimmune and Metabolic Diseases
2.1.1 Target Differentiation: Next-Generation Competitive Strategies for Mature Targets Such as TROP2, c-Met, and B7-H3
Competition for TROP2-targeted ADCs has entered a “data arms race” phase. Goscatuzumab (Trodelvy), as the world’s first TROP2 ADC, has established a firm foothold in three major indications—breast cancer, urothelial carcinoma, and triple-negative breast cancer (TNBC)—driven by overall survival (OS) benefit data from the Phase III ASCENT study.In 2025, DS-1062 (a TROP2 ADC) co-developed by AstraZeneca and Daiichi Sankyo suffered a setback in the NSCLC field (the Phase III TEAM-DT trial failed to meet the primary PFS endpoint), casting a shadow over the entire TROP2 ADC pipeline.
The direct lesson for Chinese TROP2 ADC developers is this: for the NSCLC indication, they must re-evaluate their differentiation strategies. The approach of following Trodelvy’s path in breast cancer has proven to be a dead end, while the negative results for TROP2 in NSCLC have actually opened a new window of opportunity—the target itself is sound; the issue lies in the optimization of DAR for the TOP1i payload in lung tissue and insufficient tumor penetration depth.For Chinese companies, a new direction worth focusing on in the Oncology session at BIO 2026 is the combination therapy of TROP2 ADCs with immune checkpoint inhibitors (ICIs), as well as last-line treatment options for patients resistant to PD-1 therapy.
The competitive landscape for the c-Met target is characterized by the entry of “major players.” Following its approval in 2022, AstraZeneca’s Teliso-V (a c-Met ADC) has seen slow expansion of its indications, primarily due to the extremely high expression heterogeneity of c-Met—an oncogene—across various tumor types.The 2026 BIO Oncology Forum is expected to focus on combination regimens of c-Met ADCs with EGFR inhibitors, as well as the potential for c-Met-targeted delivery to bypass the blood-brain barrier (BBB)—the latter of which holds significant therapeutic implications for patients with brain metastases.Notably, dual-target ADCs targeting c-Met and HER3 (B7-H3 × c-Met) are emerging as a new technological frontier, with multiple Chinese companies already positioning themselves in this area.
ADC development targeting B7-H3 (CD276) is set to enter a boom phase in 2025.MacroGenics’ MGC-018 (vobramitamab duocarmazine) has demonstrated promising ORR data in a Phase II study for prostate cancer, and the combination therapy of Seagen’s (now Pfizer) PSMA-ADC (PSMA-targeting ADC) with a B7-H3 ADC has emerged as a new hot combination.At BIO 2026, the key questions to focus on are: Do these B7-H3 ADCs in early clinical stages exhibit significant differences in therapeutic window and PK/PD characteristics? What clinical differences do the various companies’ linker-payload technology platforms (BEC-1 vs. TOP1i vs. DNA alkylating agent) actually yield?
[Table 2-1] Analysis of the Competitive Landscape and Differentiation Opportunities for Major ADC Targets in 2026
| Target | Approved ADCs | Key Development Milestones | Differentiation Opportunities | Key Focus Areas for BIO 2026 |
| TROP2 | Trodelvy (Gilead), sacituzumab govitecan | DS-1062 (AZ) Faces Setbacks in Phase III; Multiple Chinese TROP2 ADCs Enter Phase II/III | Combination with ICI strategies, PD-1-resistant populations, and combinations of ADCs with bispecific immune antibodies | Clinical data on TROP2 ADC + PD-(L)1 combination therapy |
| HER2 | Kadcyla (Roche), Enhertu (AZ/Daiichi) | Post-trastuzumab resistance lines, expansion to low HER2 expression | Non-breast cancer indications (GC/GEJ, biliary tract cancer), dual-target ADCs | Phase III data on HER2 ADCs in neoadjuvant treatment of gastric cancer |
| c-Met | Teliso-V (AbbVie) | In combination with EGFR inhibitors; dual-target c-Met×HER3 ADC | Brain metastases in NSCLC; biparatopic design | Phase III results for c-Met ADC in combination with osimertinib |
| B7-H3 | No approved products | MGC-018 Phase II data; multiple Chinese companies have entered Phase I/II | PSMA+B7-H3 dual-target combination; multi-tumor indications being advanced in parallel | Phase I/II data for B7-H3 ADC in prostate cancer and NSCLC |
| FRα | Elahere (ImmunoGen/AbbVie) | Late-line treatment for platinum-resistant ovarian cancer, in combination with bevacizumab | Patients with low FRα expression; in combination with an ADC | Elahere real-world evidence and latest Phase III data |
[Interpretation of Table 2-1] Differentiated competition in mature targets has entered uncharted waters. The setbacks with the TROP2 target in NSCLC remind us that the payload’s tumor penetration is one of the key variables determining the clinical success or failure of ADCs, rather than merely the choice of the target itself.At BIO 2026, examining the linker stability of various companies’ ADC technology platforms (the rationale behind choosing cleavable vs. non-cleavable linkers) and DAR optimization strategies will be more valuable than simply comparing the prominence of target molecules.
2.1.2 Core of Modality Convergence: Delivery Breakthroughs in Antibody-Oligonucleotide Conjugates (AOCs), Linker Technology Innovations, and CMC Manufacturing Challenges
AOC (Antibody-Oligonucleotide Conjugate) represents the extension of ADC technology into the field of nucleic acid therapeutics.Unlike traditional ADCs, which use cytotoxic payloads (cytotoxins), AOCs employ therapeutic oligonucleotides (siRNA, ASO, antimiR) as payloads, leveraging the tissue-specific targeting of antibodies to address the delivery challenges associated with oligonucleotide drugs.The core logic of this technological approach is to leverage the proven tissue and cell specificity of antibody drugs to bypass the limitations of LNP or GalNAc delivery systems, thereby achieving precise extrahepatic delivery.
Avidity Biosciences is a pioneer in the AOC field. Its anti-CD71 antibody-siRNA AOC (desiranib) has demonstrated encouraging data in clinical studies for Duchenne muscular dystrophy (DMD) and DM1: following a single intravenous administration, the siRNA uptake rate in muscle tissue was significantly higher than that of free siRNA-LNP formulations, and the risk of immunogenicity was manageable.In November 2025, Avidity received FDA Regenerative Medicine Advanced Therapy (RMAT) designation in the AOC field, marking the regulatory agency’s initial recognition of the AOC platform.At BIO 2026, the key questions to consider are: Which antibody scaffold (full-length IgG vs. F(ab)2 vs. scFv) is best suited for a specific oligonucleotide payload? Will the immunogenicity of AOCs become a bottleneck for large-scale clinical application?
Innovations in linker technology represent another key area where AOCs differ from traditional ADCs. The vc-PABC (valine-citrulline-p-aminobenzyloxycarbonyl) linker commonly used in traditional ADCs can cleave to release the payload in the enzyme-rich tumor microenvironment (TME), which is abundant in cathepsin B.However, the molecular weight of oligonucleotide payloads (6–15 kDa) is significantly greater than that of small-molecule cytotoxins, requiring longer linker spacers and more precise design of cleavage sites.In 2025, a noteworthy new strategy emerged: the combined design of a pH-sensitive linker and a cathepsin dual-responsive system, which enables more precise control of payload release following endocytosis by tumor cells (endosome/lysosome stage) and reduces premature release in the bloodstream.
CMC (Chemistry, Manufacturing, and Controls) manufacturing is the biggest “hidden landmine” on the path to commercializing AOCs. AOC production involves three major complex processes: antibody manufacturing, oligonucleotide synthesis, and site-specific conjugation between the two.Scale-up at each stage presents unique challenges: batch-to-batch consistency in antibody titers, oligonucleotide purity (quality standard of HPLC purity >95%), and precise control of the drug-to-antibody ratio (DAR) during the conjugation reaction.Process fluctuations in any of these steps directly impact the quality attributes of AOCs, which in turn affect clinical data and regulatory filing timelines. At BIO 2026, if you are evaluating AOC platform companies, CMC due diligence should be one of the core topics of discussion.
[Table 2-2] Technical Comparison of AOC vs. ADC vs. Free Oligonucleotides
| Dimension | Traditional ADC | GalNAc-siRNA | LNP-mRNA | AOC |
| Payload Type | Small-molecule cytotoxins (e.g., TOP1i) | siRNA/ASO | mRNA | siRNA/ASO/antimiR |
| Delivery system | Antibody-mediated endocytosis | GalNAc → Hepatocytes ASGPR | LNP → Hepatocytes | Antibody-mediated → Target tissue/cells |
| Extrahepatic delivery capacity | Strong (depends on the antibody target) | Very weak | Weak | Strong (depends on antibody specificity) |
| Dosage frequency | Q3W or Q2W intravenous infusion | Subcutaneous injection every 3 months | IV infusion every 3 months to 1 year | IV infusion every 2–4 weeks (initially) |
| CMC Complexity | High | Medium | Very high | Extremely High (Three-Component Conjugation) |
| Commercial Maturity | Mature (20+ marketed products) | Fairly mature (6 approved products) | Mature (COVID-19 vaccines) | Early-stage (0 approved products) |
| Cost per Course (Estimated) | $10,000–50,000 | $3,000–15,000 | $100–500 (spiked after COVID) | $20,000–80,000 (early estimate) |
| 2026 BIO Popularity | ★★★★★ (Mature market) | ★★★★☆ (Highly competitive) | ★★★★★ (mRNA cancer vaccines) | ★★★☆☆ (Emerging Proof of Concept) |
[Interpretation of Table 2-2] AOC represents the cutting edge of the convergence between ADC and nucleic acid drug technologies. Although no products have been approved yet, the progress made by companies such as Avidity Biosciences, Denali Therapeutics (in collaboration with Secarna), and Tallac Therapeutics indicates that this sector is transitioning from “proof of concept” to “proof of clinical concept.”For BD licensing decision-makers, the greatest appeal of AOC lies in its potential to extend the drugable space of any antibody-validated tumor/disease target into the nucleic acid therapeutics domain. This means that if you have a clinically validated antibody candidate but face bottlenecks in payload development, AOC may be a differentiated pathway worth exploring.
2.2 The “Post-GLP-1” Storm in the Metabolic Therapeutics Sector
2.2.1 Multi-Target Synergy: Business Logic and Risk Assessment for Mechanisms Such as GIP, Pancreatin, and Glucagon
The GLP-1 sector has entered the “post-single-target era” in 2025–2026.Eli Lilly’s tirzepatide (a dual GIPR/GLP-1R agonist) surpassed $5 billion in global sales in 2024, overtaking dulaglutide and semaglutide to become the new leader in the metabolic market.However, MNCs have long since shifted their strategic focus to the next generation—triple-target agonists targeting GIPR/GLP-1R/glucagon receptor (GCGR),GLP-1R/amylin co-agonists, and GLP-1R/GIPR/amylin triple-combination regimens—are set to be the central themes of the BIO Metabolism session in 2026.
The scientific rationale behind multi-target synergy is clear: GLP-1R agonism addresses insulin secretion and appetite suppression; GIPR agonism enhances glucose-dependent insulin secretion and may improve lipid metabolism; glucagon receptor (GCGR) agonism increases energy expenditure (thermogenesis); and amylin synergistically slows gastric emptying and increases satiety.In Eli Lilly’s Phase II SURPASS-Plus study, Retatrutide (a triple-target GLP-1R/GIPR/GCGR agonist) achieved a median weight loss of 24.2% at 48 weeks, making it the multi-target candidate with the most aggressive weight loss data to date.
However, the business logic behind multi-target strategies also carries risks that cannot be ignored. First is the cumulative safety profile: while GIPR and GCGR agonism provide metabolic benefits, they may also lead to elevated pancreatic enzymes, increased gastrointestinal adverse events, and even a risk of pancreatitis.In Phase II studies, Retatrutide had a Grade 3 or higher gastrointestinal adverse event rate of approximately 12%, significantly higher than the 2–4% seen with single-target GLP-1 agonists. Second is the challenge of dose titration: multi-receptor activation implies an exponential increase in the complexity of pharmacokinetic/pharmacodynamic (PK/PD) curves, placing immense pressure on clinical development and the design of commercial formulations.At BIO 2026, the key question you need to evaluate is: Does this company’s multi-target candidate demonstrate a statistically significant safety advantage—particularly regarding pancreatic and gastrointestinal safety—compared to single-target agents?
[Table 2-3] Competitive Landscape and Clinical Progress of Metabolic Multi-Target Agonists in 2026
| Drug Name | Target Combination | Company | Indications | Latest Clinical Phase | 48-Week Weight Loss Data | Key Risks |
| Tirzepatide | GIPR + GLP-1R | Eli Lilly | T2D/Obesity/NASH | Approved | 22.5% (15 mg) | Gastrointestinal AEs, risk of thyroid C-cell tumors |
| Retatrutide | GIPR + GLP-1R + GCGR | Eli Lilly | T2D/Obesity | Phase III | 24.2% (12 mg) | Grade 3+ gastrointestinal AEs (12%), increased heart rate |
| Cotadutide | GLP-1R + GCGR | AstraZeneca/MedImmune | T2D/NASH/Obesity | Phase IIb | 7.4% (300 μg) | GCGR agonism → risk of elevated liver enzymes |
| Pemvidutide | GLP-1R + GCGR | Altasciences | Obesity/NASH | Phase II | 15.6% (2.4 mg) | GCGR-associated thyroid C-cell carcinoma? |
| Amycretin | GLP-1R + Amylin | Novo Nordisk | Oral obesity | Phase II (oral) | 13.1% (extrapolated from Phase I data) | Subcutaneous injection efficacy vs. oral bioavailability |
| Survodutide | GLP-1R + GCGR | Boehringer Ingelheim/Zealand | NASH/Obesity | Phase III | 16.8% (4.8 mg) | Cardiovascular safety of GCGR agonists remains to be validated |
[Interpretation of Tables 2–3] The competitive landscape for multi-target metabolic drugs has become highly clear: Eli Lilly’s leading position in the weight loss market is virtually unassailable, thanks to its first-mover advantage with the GIPR+GLP-1R dual-target approach and the comprehensive data matrix from the SURMOUNT series of studies. Opportunities for Chinese companies are primarily concentrated in:
① Differentiated indications for GLP-1R-based dual-target combinations (especially GLP-1R + Amylin);
② Delivery technology platforms for oral GLP-1 agonists;
③ Clinical development of GLP-1 agonists for NASH/MASH indications. At the Metabolism track of BIO 2026, data readouts for NASH/MASH indications warrant particular attention—the size of this unmet medical need is 3–4 times that of the obesity market.
2.2.2 The Wave of Oral Small-Molecule Drugs: Disruptive Potential for Biologics and Cross-Domain Lessons from Delivery Experience
Competition among oral GLP-1 agonists has reached a fever pitch.Novo Nordisk’s semaglutide oral tablets (Rybelsus) are currently the only approved oral GLP-1R agonist, but their bioavailability is only about 1% (requiring daily oral administration on an empty stomach, swallowing the tablet whole, and avoiding water for 30 minutes), which severely limits clinical accessibility and patient adherence.Eli Lilly’s Orforglipron (an oral small-molecule GLP-1R agonist) met its primary endpoint in the Phase III ACHIEVE trial in 2025: a 15.3% weight loss over 40 weeks. With the convenience of oral administration significantly superior to injectable formulations, it has the potential to disrupt the entire GLP-1 market landscape.
The core advantages of oral small-molecule GLP-1 agonists lie in: low production costs (approximately one-tenth that of biologics), low barriers to administration (no need for subcutaneous injections), and relaxed storage and transportation requirements (room temperature vs. cold chain).More importantly, the half-life of small-molecule GLP-1 agonists can be precisely controlled through chemical structural modifications, enabling more flexible dosing regimen design. For China’s GLP-1 market, this presents both an opportunity and a threat—if oral small-molecule GLP-1 agonists reduce monthly treatment costs to the $200–500 range, domestically produced GLP-1 biosimilars, which currently rely on cost advantages, will face intense price competition.
The transfer of experience in oral delivery technology to other drug modalities is one of the key cross-modal considerations in this chapter.The permeation enhancer technology (with SNAC as the core component) relied upon for oral GLP-1 delivery is currently being explored for the oral delivery of other macromolecular drugs. Emisphere’s (now acquired by Novo Nordisk) Eligen technology platform has demonstrated the feasibility of permeation enhancers in oral peptide drugs, and this technical approach may offer valuable insights for the oral delivery of nucleic acid drugs.At BIO 2026, if you are following both the GLP-1 and nucleic acid drug sectors, it is worth seeking out companies with cross-modal strategies in delivery technology platforms—such companies are often prime targets for business development (BD) licensing.
[Table 2-4] Comparison of Key Dimensions Between Oral and Injectable GLP-1 Agonists and Cross-Modality Implications
| Dimension | Oral Small-Molecule GLP-1 (e.g., Orforglipron) | Injectable Peptide GLP-1 (e.g., Semaglutide) | GLP-1 Biosimilars | Cross-Modality Insights |
| Bioavailability | ~5–10% (Eligen technology) | ~70–80% (subcutaneous injection) | Compared to the reference biologic | Permeation enhancers may migrate into nucleic acids Oral delivery |
| Dosage frequency | Once daily by mouth | Once weekly by subcutaneous injection | 1–4 times per week | Patient compliance: Oral > Injection |
| Average monthly cost per treatment course (U.S. market, 2025) | $400–800 (projected post-commercialization) | $900–1,200 (brand-name drug) | $200–400 (biosimilar) | Oral small molecules → A cost war is about to begin |
| Cold chain/storage requirements | Room temperature storage | Refrigerated at 2–8°C (pen-type) | Refrigerated at 2–8°C | Room-temperature stability → Simplified supply chain |
| Clinical Development Risks | Phase III results are positive, but long-term CV risks remain to be validated | Validated (complete CVOT data) | The biosimilar pathway is relatively well-established | Regulatory: Bioavailability bridging for oral formulations is complex |
| Opportunities for Chinese Companies | High likelihood of “me-better” or “me-too” products (patentability of small molecules) | Intense competition in the “me-too” space, with limited differentiation | Cost and production capacity are core competitive advantages | Delivery technology platforms hold greater BD value than individual molecules |
2.3 The Second Half of CGT and Precision Medicine
2.3.1 Evolution of In Vivo Delivery: Extrahepatic Delivery Beyond LNP, Non-viral Vectors, and Scalable Manufacturing
In the second half of the CGT (Cell and Gene Therapy) race, the deciding factor has shifted from “whether gene editing can be achieved” to “whether safe, reproducible, and affordable in vivo delivery can be achieved.”LNP (lipid nanoparticles), as the core delivery technology for mRNA COVID-19 vaccines, has been administered in over 5 billion doses globally, demonstrating its baseline capabilities for large-scale manufacturing and safety. However, LNP’s inherent hepatic tropism—specifically, its uptake by liver cells through binding to apolipoprotein E (ApoE)—constitutes a fundamental limitation for its application in extrahepatic tissue delivery.
In 2025–2026, four non-hepatic delivery technology pathways are emerging as focal points at the CGT conference:
- ① LNP chemical modification: Altering the surface charge of LNPs (by adjusting the pKa of ionizable lipids) and ligand modification (by adding targeting antibodies or peptides) to achieve selective accumulation in tissues such as the lungs, myocardium, and muscles. Ethos Therapeutics (an MIT spin-off) has made notable technological progress in this area.
- ② Exosome delivery: Exosome engineering technologies pioneered by Genethon and Codiak BioSciences (which has undergone bankruptcy reorganization) leverage exosomes’ natural cell fusion capabilities to bypass lysosomal degradation. While extremely costly, this approach offers outstanding targeting potential.
- ③ Virus-like Particles (VLP): The SORT (Selective Organ Targeting) platform developed by ReCode Therapeutics enables precise targeting of different tissues—such as the spleen, liver, and lungs—by adding SORT molecules to the surface of LNPs. Preclinical data from 2025 is encouraging.
- ④ Virus-like Particles (VLP): These are self-assembled particles composed of viral capsid proteins without genetic material, serving as delivery vehicles for macromolecules (proteins, oligonucleotides) and achieving a better balance between immunogenicity and tissue permeability.
Scaling up manufacturing is the final hurdle to commercializing extrahepatic delivery technologies. Unlike the mature manufacturing processes for LNPs, most novel delivery carriers (exosomes, VLPs, and virus-like particles) are currently still in the batch production phase, with batch-to-batch consistency being the greatest CMC challenge. At BIO 2026, if you are interested in the scaled-up manufacturing of CGT, the following metrics are worth focusing on:
① Has production scale been scaled up to bioreactors of 30L or larger?
② Does the number of doses per batch exceed 100?
③ Has the carrier purification process (chromatography vs. ultracentrifugation) been standardized to cGMP?
[Table 2-5] Competitive Landscape and Technology Maturity Assessment of CGT Delivery Platforms in 2026
| Delivery Platform | Representative Companies | Target Tissue | Clinical Stage | Scale-Up Status | CMC Challenges | 2026 BIO Buzz |
| LNP (Standard) | Moderna, BioNTech, Alnylam | Liver (Natural) | Commercialized (COVID vaccine) | Mature (10,000 L scale) | Controllable | ★★★☆☆ (Mature topic) |
| LNP (SORT-modified) | ReCode Therapeutics | Spleen/Liver/Lung (Programmable) | Pre-IND / Phase I | Pilot-scale | SORT Molecular Batch Consistency | ★★★★★ (Hottest Area) |
| LNP (Ligand-Modified) | Genevant, Arcturus | Programmable (Multi-tissue) | Phase I/II | Early-stage expansion | Ligand density control | ★★★★☆ |
| Exosome engineering | Codiak (post-recombinant), Evox | CNS, Oncology (theoretically) | Phase I (pilot-scale) | Extremely difficult to scale up | Batch-to-batch consistency, purification costs | ★★★☆☆ |
| Virus-like particles (VLP) | Recode, Viacyte | Multi-tissue (depending on design) | Preclinical | Preclinical | Assembly efficiency | ★★★★☆ |
| AAV (standard) | Spark (Roche), Luxturna (Novartis) | Eye, CNS (natural) | Approved (for certain indications) | Relatively mature | Empty capsid rate, immunogenicity | ★★★☆☆ (High competition) |
| AAV (engineered capsid) | Dyno, 4DMT, Sparked | Multi-tissue (directed evolution) | Phase I/II | Currently in pilot-scale upscaling | Directed evolution efficiency, immunogenicity | ★★★★★ |
| Non-viral electroporation (ex vivo) | Lonza, Catalent (CDMO) | T cells, NK cells (ex vivo) | Commercially available (CAR-T) | Mature (GMP system) | T-cell depletion, transduction efficiency | ★★★☆☆ (CDMO integration) |
[Interpretation of Table 2-5] Competition in CGT delivery technologies has shifted from “LNP dominance” to a three-way stalemate among “LNP, engineered AAV, and novel vectors.”For BD licensing decision-makers, the most promising directions are SORT-modified LNP (ReCode) and engineered AAV capsids (4DMT, Dyno)—the former is expected to combine the large-scale manufacturing advantages of LNP with extrahepatic delivery capabilities, while the latter demonstrates significant clinical differentiation in CNS and ophthalmic indications.At BIO 2026, we recommend prioritizing discussions with the BD teams of these two types of companies and paying close attention to clinical data readout milestones in 2025–2026.
2.3.2 Practical Applications of AI: Real-World Cases of Patient Stratification, Pipeline Optimization, and Improved Clinical Success Rates
The application of AI in CGT development has moved from the “hype cycle” to the “data validation” phase. However, distinguishing truly valuable AI applications from mere marketing gimmicks is one of the most challenging and critical tasks at BIO 2026. In the CGT field, the practical implementation of AI primarily focuses on three areas: patient stratification, process optimization, and protocol design.
Patient stratification is the scenario where AI’s value in CGT is most clearly defined.Carisma Therapeutics’ CAR-M (macrophage chimeric antigen receptor) platform uses machine learning algorithms to analyze single-cell RNA sequencing (scRNA-seq) data and identify biomarkers in patients most likely to respond to CAR-M therapy—this AI-based patient stratification strategy sets it apart in the solid tumor CAR-T/CAR-M space.Insilico Medicine’s AI-driven drug discovery platform demonstrates another dimension: using generative AI to design novel T-cell receptor (TCR) mimetics, compressing the traditional 2–3-year TCR discovery cycle to 6–9 months.
The application of AI in optimizing CGT manufacturing processes is equally noteworthy. Cellares’ automated cell manufacturing platform (Integrated Cartridge Change, ICC) uses real-time machine learning to monitor critical quality parameters (CQAs) during production, reducing the failure rate of personalized CAR-T cell manufacturing from the industry average of 15–20% to below 5%.For readers considering BD’s licensed CGT platform companies, the ability to optimize manufacturing processes using AI is a critical dimension for evaluating a company’s technological barriers—it directly impacts product affordability and commercialization costs.
[Table 2-6] Practical AI Applications in the CGT Field and Technology Maturity Assessment for 2026
| Application Scenarios | Representative Companies/Institutions | Specific Case | Data Support | Key Points for BD Evaluation |
| AI Patient Stratification | Carisma Therapeutics | scRNA-seq + ML to Identify CAR-M Response Biomarkers | Phase I: 21 patients, ORR 38% | Is the biomarker suitable for commercial testing? |
| AI-Driven TCR Design | Insilico Medicine, Adaptive Biotechnologies | Generative AI → TCR discovery cycle: 2–3 years → 6–9 months | AI-designed TCRs have been validated in clinical trials | IP Ownership (Patentability of AI-Generated Sequences) |
| AI Manufacturing Monitoring | Cellares, Miltenyi Biotec | Real-time ML monitoring of CQA → CAR-T failure rate 15% → <5% | FDA RMAT certification obtained | Is the collaboration model with CDMOs flexible? |
| AI Dose Optimization | Kite Pharma (Gilead), Bristol-Myers Squibb | PK/PD modeling to predict optimal dosing regimens | Model validation based on real-world data | Has the clinical protocol been aligned with regulatory authorities? |
| AI-Driven Indication Expansion | Novartis Gene Therapy Team | AI analysis of existing gene therapy databases → Identification of signals for new indications | Early exploration phase | Regulatory Pathway for Indication Expansion |
| AI Safety Prediction | Multiple academic studies (UCSF, Oxford) | AI prediction of AAV neutralizing antibodies (NAbs) → Reduced risk of clinical failure | Currently undergoing retrospective validation | Has the predictive model undergone prospective validation? |
[Interpretation of Table 2-6] The application of AI in the CGT field has entered a “tipping point”—AI applications backed by clinical data are beginning to command a premium in the business development (BD) market, while purely conceptual AI companies face a funding winter. At BIO 2026, to identify truly valuable AI-driven CGT companies, you need to ask three key questions:
① Have the AI model’s predictions been prospectively validated in clinical trials?
② Is the intellectual property ownership of AI-generated data, sequences, and biomarkers clearly defined?
③ Is the cost of AI-assisted decision-making lower than the incremental clinical and commercial value it delivers?
2.4 Cross-Modal Thinking Framework: How to Apply Lessons Learned from GLP-1 Oral Delivery to Nucleic Acid Drugs and ADCs
Cross-modal thinking is the most critical tool for differentiated insight in this guide. Breakthroughs in oral delivery technology within the GLP-1 space are providing a valuable pathway for transferring experience to delivery innovations in nucleic acid therapeutics and ADCs. Below are three specific examples of cross-modal connections to help you quickly identify convergence opportunities at BIO 2026.
Cross-Modal Connection 1: Modality Expansion of Permeation Enhancers. SNAC (sodium N‑[8-(2-hydroxybenzoyl)amino]caprylate) is the core permeation enhancer in semaglutide oral tablets (Rybelsus). Its mechanism of action involves forming temporary lipophilic channels on the surface of gastric epithelial cells to increase the transmembrane absorption of peptide drugs.This technical approach is currently being explored for the oral delivery of other macromolecular drugs. Exactech Sciences’ (a spin-off from AbbVie) progressive pipeline strategy—”GLP-1 → oral GLP-1 → oral peptides → oral nucleic acids”—demonstrates that the feasibility of extending SNAC-like technologies to the oral delivery of oligonucleotides is being validated.At BIO 2026, if you are following both GLP-1 and nucleic acid therapeutics, look for companies with SNAC or similar permeation enhancer platforms—such platforms hold extremely high cross-modal licensing value.
Cross-modal connection #2: Mutual learning from nanoparticle engineering experience. Design insights from ionizable lipids in LNPs are profoundly influencing the optimization logic of ADC linker-payload technology.The lipid components used in LNPs to regulate endosomal escape (such as MC3’s lead compound DLin-MC3-DMA) share similar pH gradient-dependent design principles with the pH-sensitive linkers used in ADCs to regulate intracellular payload release.At BIO 2026, asking an ADC company’s linker design team whether they have drawn inspiration from pH-responsive strategies in the LNP field can quickly reveal the depth of that company’s technical vision.
Cross-modal Connection 3: Insights from GLP-1 multi-target synergistic strategies for ADC dual-payload design. The logic behind the synergistic effects of GLP-1/GIPR dual-target agonists—where the two receptors act on different tissues and signaling pathways—is being adopted in the ADC field.Dual-payload ADCs—which carry two payloads with different mechanisms of action simultaneously (e.g., TOP1i + DNA-damaging agent, or TOP1i + immune modulator)—represent a new direction worth watching in 2026. The core logic behind this design is to enhance tumor-killing efficiency through the synergistic action of different payloads while reducing the toxicity risks associated with high doses of a single payload.At BIO 2026, keep an eye on proof-of-concept (PoC) data for dual-payload ADCs—if the data is positive, this will mark the beginning of an entirely new therapeutic class.
[Table 2-7] Cross-modal knowledge transfer pathways among the three major tracks and opportunity identification at BIO 2026
| Transfer Direction | Source Modality Experience | Target Modality Applications | Technical Connection Points | Types of Companies of Interest for BIO 2026 |
| GLP-1 Oral Permeation Enhancers → Oral Nucleic Acid Delivery | SNAC Permeation Enhancer Technology | Oral siRNA/ASO | Increased Transmembrane Absorption, Increased Bioavailability | Companies with GLP-1 oral platforms + nucleic acid pipelines |
| LNP endosome escape design → pH-sensitive ADC linker | pH-responsive lipid design for LNPs | Precise payload release following ADC endocytosis | Common design principles based on pH gradients | CDMOs/biotech companies with LNP platforms and ADC linker assets |
| GLP-1/GIPR Co-agonism → ADC Dual-Payload Synergy | Synergistic signal amplification via dual-receptor activation | Dual-payload ADC (TOP1i + immunostimulant) | Synergistic effects ↑, toxicity risk ↓ | Biotech companies with dual-payload ADC pipelines (PoC stage) |
| GLP-1 formulation expertise → CGT scale-up manufacturing | Scalable processes for GLP-1 biologics | Manufacturing optimization for CGT cell/gene therapies | Experience with automated closed-system manufacturing | CDMO with commercialization experience in GLP-1 biologics |
| AI-Driven Patient Stratification for CGT → GLP-1 Biomarker Discovery | scRNA-seq + ML Patient Stratification | Prediction of GLP-1 Response Biomarkers | Transferable AI-driven biomarker discovery tools | AI + multimodal biomarker platform company |
| AAV capsid engineering → LNP tissue targeting | AAV directed evolution → ↑ tissue specificity | SORT-LNP → Multi-tissue targeting | Common Directed Evolution Methodology (DNA Shuffling) | Technology-driven biotech company with a directed evolution platform |
[Interpretation of Table 2-7] Cross-modal knowledge transfer is the most effective tool for identifying “unmet needs” and “technology convergence points.”At BIO 2026, most attendees were searching for targets within their own therapeutic areas—but your competitive advantage lies in identifying the next technological evolution for ADCs and nucleic acid therapeutics by leveraging your experience in oral delivery from the GLP-1 space. This cross-modal perspective often helps you uncover collaboration opportunities in BD discussions that the other party has not yet recognized.
▶ Action Recommendations (Chapter II):
- ADC Session: Attend with the question, “Does the linker design incorporate LNP’s endosome escape strategy?” You’ll gain unexpected technical insights.
- AOC Forum: Focus on evaluating CMC feasibility and immunogenicity data, as these are currently the most underestimated risk factors.
- Metabolism Track: Prioritize the impact of the full Phase III data for Orforglipron (an oral small-molecule GLP-1 agonist) on China’s GLP-1 market.
- CGT Delivery Session: SORT-LNP is the most promising extrahepatic delivery technology to track in 2026, with ReCode as the top target for attention.
- AI+CGT: Pose three critical questions to all AI companies: prospective validation, IP ownership, and cost-benefit ratio.
3.0. biotech events san diego: 70 Days Before the Event: Biopartnering and the Extreme Optimization of Personal ROI

70 days may sound like a long time, but in a BIO conference hall packed with 21,000 people, it’s enough for an unprepared attendee to get lost in the noise and leave empty-handed. This chapter provides a comprehensive “pre-conference optimization” framework covering Partnering Profile development, target search strategies, agenda customization, and templates for defining four types of targets.The goal is this: to use these 70 days to ensure that, the moment you step into the venue, you are already three months ahead of 80% of the attendees.
3.1 Asset-Centric Profile Building: Use Precise Keywords to Make Potential Partners Find You
The BIO Partnering system is the world’s largest biopharmaceutical business development (BD) matching platform. In 2025, the BIO Partnering system facilitated approximately 38,000 one-on-one meeting bookings and processed connection requests from BD professionals in over 90 countries. Within this system, your Partnering Profile is the only channel through which potential partners can form an impression of you before any face-to-face interaction.However, most Chinese companies’ profiles remain stuck in the “Company Overview + Pipeline List” 1.0 phase—a massive waste of BD resources.
The core concept of an “asset-centric” profile-building approach is to organize your Partnering Profile around “the specific assets you are willing to license out or in,” rather than around “what products your company has.” For Chinese companies expanding overseas, this means you need to treat each specific drug candidate (at the IND stage or beyond) as an independent “asset unit” and write a separate Profile Section for each asset unit, highlighting:
① The asset’s therapeutic differentiation (why does this indication require this molecule?);
② The credibility of key data (number of patients in clinical data, follow-up duration, data sources);
③ The collaboration model you seek for this asset (out-licensing, co-development, joint venture, etc.).
A keyword strategy is a key tool for making your Partnering Profile stand out in BIO system search results. BIO Partnering’s search algorithm is based on text matching, which means the keywords you use in your Profile (therapeutic areas, targets, modalities, and technology platform keywords) directly determine which searchers will discover you. For the four major golden sectors, here are keyword strategy recommendations:
[Table 3-1] BIO Partnering Profile Keyword Matrix for the Four Golden Therapeutic Areas
| Track | High-Value Keywords (Required) | Secondary Keywords (Recommended) | Long-tail Keywords (Supplementary) | Invalid Terms to Avoid |
| ADC/XDC | antibody-drug conjugate, ADC, TROP2, HER2, c-Met, linker-payload, topoisomerase I inhibitor | B7-H3, FRα, tumor-targeting, bystander effect, drug-antibody ratio, site-specific conjugation | next-generation ADC, dual-payload ADC, AOC, XDC | innovative, cutting-edge (no substantive information) |
| Nucleic acid therapeutics/RNA | siRNA, ASO, GalNAc, in vivo delivery, LNP, extrahepatic delivery, oligonucleotides | mRNA vaccine, nucleoside-modified, UPL, exon skipping, antisense | AOC, therapeutic oligonucleotides, nucleic acid therapeutics | advanced technology (no target information) |
| GLP-1/Metabolism | GLP-1 agonist, GLP-1R, tirzepatide, GIP/GLP-1 dual agonist, oral GLP-1, incretin | GLP-1 biosimilar, multi-agonist, amylin, glucagon receptor, NASH/MASH | Small-molecule GLP-1, oral peptide, glucagon-like peptide-1 | metabolic disease (too broad) |
| CGT/Gene Therapy | gene therapy, AAV, in vivo delivery, LNP gene editing, CRISPR, CAR-T | extrahepatic LNP, exosome delivery, manufacturing scale-up, viral vector, cell therapy | iPSC-derived, allogeneic, in vivo gene editing, LNP for rare diseases | cell therapy (no vector/target information) |
[Interpretation of Table 3-1] The core principle for filling in keywords is “specificity”: the more specific the target name, technology platform name, or modality name, the easier it is for BD professionals with genuine needs to accurately find your profile.In the BIO Partnering system, a U.S. BD professional seeking a Chinese partner for a TROP2 ADC would search for “China TROP2 ADC” or “Chinese ADC partner”—your Profile must include all three elements—”China,” “TROP2,” and “ADC”—to be matched with this search query.
3.2 Granular Search and Prioritization Strategies for ADCs, AOCs, Nucleic Acid Therapeutics, and GLP-1+ Therapeutics
The advanced search features of the BIO Partnering system are tools that most attendees fail to utilize to their full potential. The system supports filtering across multiple dimensions, including company type (Biotech/Pharma/CDMO/Investors), region, therapeutic area, modality, and collaboration type. However, true experts refine their search granularity down to the “asset level”—searching not for “ADC companies,” but for “Chinese companies currently conducting a TROP2 ADC Phase II trial and seeking out-licensing opportunities.”Below is a step-by-step guide for conducting such granular searches.
Step 1: Create an “Ideal Partner Profile” (IPP). Before opening the BIO Partnering system, create your IPP offline first. For Chinese biotech companies seeking out-licensing opportunities, the IPP should clearly define:
① The type of target partner (MNC/mid-to-large-sized pharma/biotech);
② The strategic priorities of target partners (Do they actually have a presence in the ADC space? What are their BD preferences?);
③ The target partner’s decision-making chain (who are the key decision-makers for out-licensing?); For Chinese pharma companies seeking in-licensing, the IPP should specify:
① The stage of the asset to be in-licensed (IND/Phase I/Phase II);
② The desired therapeutic indications;
③ Acceptable price range and collaboration structure.
Step 2: Detailed Search and Preliminary Screening. Use the following search combinations in the BIO Partnering system: Modality (ADC/Nucleic Acid Therapeutics/CGT/Metabolics) + Stage (Phase I/II/III) + Region (China/East Asia/USA/Europe) + Collaboration Intent (Out-licensing/In-licensing/Joint Development).Record the number of hits for each search combination and prioritize pipelines with 20–100 hits (too few hits indicate overly narrow search criteria, while too many indicate overly broad criteria).
[Table 3-2] Priority Ranking Framework for BIO Partnering Granularity Searches
| Priority | Search Granularity | Expected Number of Hits | Screening Criteria | Execution Order |
| P1 (Highest) | Exact sector + Phase II + Out-licensing + China | 5–20 companies | Asset differentiation, data reliability, and genuine willingness to collaborate | Executed first (4–6 weeks prior) |
| P2 (High) | Targeted sectors + Phase I/II + Joint Development + No geographic restrictions | 20–50 companies | Clarity of IP ownership, uniqueness of technology platform | Second priority (4–5 weeks prior) |
| P3 (Medium) | Market Segment + Phase I + In-licensing + No Geographic Restrictions | 50–100 companies | Target differentiation, ease of administration, commercialization pathway | Third (3–4 weeks prior) |
| P4 (Supplemental) | Therapeutic Area + IND + All Collaboration Types + Asia-Pacific | 100–200 companies | Used solely to gauge market breadth; no in-depth follow-up | Final (2–3 weeks prior) |
[Interpretation of Table 3-2] The core logic behind the prioritization is a dual assessment of “asset maturity × collaboration potential.” Assets in Phase II have sufficient data depth to support BD discussions and represent the “golden window” for licensing negotiations.Although Phase I assets have limited data, they offer high licensing flexibility (allowing for earlier-stage collaboration structures such as option deals). IND-stage assets are primarily suited for MNCs’ “early pipeline filling” strategies; BD professionals focusing on these assets are typically not the most critical dialogue partners.
3.3 Personalized Agenda Customization: Target High-Value Sessions in Oncology, Next-Gen Biotherapeutics, Business Development, and More
The official BIO agenda tool (Conference Program) serves as the schedule for all sessions and events, but its greatest value lies not in helping you “run from one session to the next,” but in enabling you to make selective trade-offs. With 21,000 attendees, a four-day event, and approximately 80–100 sessions per day, it is impossible for anyone to cover everything. You need a clear framework for prioritization.
Decision-Making Framework: Categorize sessions based on their “relevance to your core objectives.” If your core objective is out-licensing, prioritize attending Business Development & Licensing, Company Presentations, and the Partnering Day Workshop. If your core objective is fundraising, focus on Investor Pitch Sessions, the Financing Trends Forum, and Innovator Company Presentations. If your core objective is supply chain/CMC collaboration, focus on tracks such as Manufacturing & Supply Chain and Quality & Regulatory.
It is worth familiarizing yourself in advance with the personalized schedule feature in the official BIO App (available for download on the App Store and Google Play). You can set preferences within the app (therapeutic area, modality, company type), and the system will recommend the most relevant sessions for you. However, please note: the app’s recommendation algorithm is based on publicly available company information and may not align perfectly with your actual business development goals. We recommend using the app’s recommendations as a preliminary screening tool, and adjusting your final agenda based on your pre-arranged meetings with target companies.
[Table 3-3] BIO 2026 High-Value Sessions Priority Matrix (Categorized by Conference Objectives)
| Your Core Objective | Must Attend (Priority 1) | Recommended (Priority 2) | Optional to Skip | Number of Sessions per Category |
| BD External Authorization | BD&L Workshops, Company Presentations (Partnering), Deal Room Orientation | Oncology Company Presentations, Next-Gen Biotherapeutics, Regulatory Science | Education Sessions (Basic Science), Networking Receptions (Social Only) | P1: 8–12 sessions; P2: 10–15 sessions |
| BD In-licensing | BD&L Sessions, Partnering Sessions, In-licensing Strategy Workshops | Scientific Platform Presentations, CMC & Manufacturing Sessions | Basic Research Sessions, Rare Disease Track (unless otherwise specified) | P1: 6–10 sessions; P2: 10–15 sessions |
| Fundraising/Investor Relations | Investor Pitch Sessions, VC Fireside Chats, Capital Markets Forum | Company Presentations, Dealmaking & Valuation Sessions | Technical Sessions (unless there is a funding story) | P1: 10–15 sessions; P2: 8–12 sessions |
| CMC Supply Chain Matchmaking | Manufacturing & Supply Chain Sessions, CMC Strategy Workshops | CDMO Company Presentations, Regulatory Compliance Sessions | Scientific Frontiers Sessions (unless relevant to your technical focus) | P1: 6–8 sessions; P2: 8–12 sessions |
| Regulatory Insights/Policy | Regulatory Science Sessions, FDA/EMA Policy Forums | Global Regulatory Convergence Sessions | Clinical/Scientific Sessions (unless related to regulatory hot topics) | P1: 4–6 sessions; P2: 6–10 sessions |
[Interpretation of Table 3-3] Regardless of your objectives, BD&L (Business Development & Licensing) sessions are the essential “common ground” you must attend. The official BIO BD&L Workshop is typically led by BD decision-makers from multinational corporations (MNCs), who explain current deal-making logic and valuation methods, providing an excellent window into the mindset of counterparties.It is worth noting that company presentations during BIO typically follow a “15-minute presentation + 5-minute Q&A” format, offering a rare opportunity to establish direct contact with the CFO or BD Head of target companies—be sure to schedule partnering meetings with these companies in advance via the BIO Partnering system.
3.4 Goal Setting and Resource Allocation Framework (Four Main Pillars: BD Authorization, Financing, CMC Supply Chain, and Regulatory Insights)
Goal setting for BIO 2026 should not stop at the level of “I’m going to BIO to attend meetings.” True experts break down their attendance goals into specific KPIs across four main tracks, with clear numerical targets and execution paths for each track. Below are goal-setting templates for the four main tracks; it is recommended to complete them at least three weeks before departure.
[Table 3-4] BIO 2026 Four-Track Goal Setting Templates
| Objective Type | Recommended KPI Targets | Execution Path | Timeline | Resource Allocation |
| Out-licensing | ① Complete initial outreach to 20+ target companies ② Secure follow-up interest from 5–8 MNCs/pharma companies ③ Finalize 1–2 companies for in-depth due diligence (DD) | ①Schedule a Partnering Meeting (6 weeks in advance) ②Profile Optimization (4 weeks in advance) ③One-on-one meetings at the event | Partnering Meeting: Booking must be completed 4–6 weeks before departure | Total time commitment: 40–50% |
| Fundraising/Investor Relations Main Track | ① Engage with 15–25 VCs/Corporate VCs ② Conduct 5–8 in-depth pitch meetings ③ Secure 2–3 firm term sheets | ① Prepare pitch deck (3 weeks in advance) ② Confirm investor list (4 weeks in advance) ③ Confirm VC registry | Finalize Pitch Deck: 3 weeks before departure | Total time commitment: 25–30% |
| CMC Supply Chain Mainstream | ① Complete technical capability assessments of 5–10 CDMOs ② Establish contacts with 2–3 potential CDMO partners ③ Gather the latest CMC quotes and production timelines | ① Prepare CDMO list (4 weeks in advance) ② Schedule manufacturing sessions ③ On-site factory visits (if applicable) | CDMO scheduling: 3–4 weeks in advance | Total time commitment: 15–20% |
| Regulatory Insights Timeline | ① Attend 3–5 Regulatory Sessions ② Informal discussions with FDA/EMA reviewers (if applicable) ③ Bring back at least 3 regulatory insights relevant to pipeline development | ① Pre-register for Regulatory Sessions (2 weeks in advance) ② Prepare a list of key regulatory issues | Regulatory pre-registration: 2 weeks in advance | Total time commitment: 10–15% |
[Interpretation of Tables 3-4] The four main tracks are not allocated in parallel—you need to select one primary track and one secondary track based on your company’s strategic priorities at this stage, and concentrate resources to achieve breakthroughs in these areas.If you are an early-stage biotech company (preclinical/Phase I), fundraising and BD licensing are your two main tracks; if you are a clinical-stage biotech company (Phase II/III), BD licensing and CMC supply chain are your two main tracks; if you are a mature pharmaceutical company, CMC supply chain and regulatory insights are your two main tracks. Choosing the wrong resource allocation priorities is the biggest waste for most Chinese companies attending BIO.
▶ Action Recommendations (Chapter III):
- 【Take Action Now】Optimize your Partnering Profile—start today, with the goal of finalizing it six weeks before departure.
- [Take Action Now] Create an IPP (Ideal Partner Profile) — Beyond the BIO Partnering system, build your own IPP database in Excel.
- 【Take Action Now】Download the BIO App—Familiarize yourself with the agenda customization features in advance and set your preference tags.
- 【Confirm 3 Weeks Before Departure】Ensure the four main KPI categories are filled out and that Partnering Meeting appointments are scheduled (at least 15 targets).
4.0. biotech events san diego: On-Site Execution: Efficiently Identifying High-Value Signals Amid the Noise of 20,000 People

The venue is a battlefield, not a sightseeing tour. Within the 400,000-square-meter space of the San Diego Convention Center, 21,000 people are doing the same thing at the same time: seeking the next collaboration opportunity. This chapter provides a comprehensive “battlefield information extraction system” to help you focus your limited attention and energy on the highest-value signals.
4.1 Breaking Through the Conference: Core Technology Sessions + “Meetings Outside the Conference” (Torrey Pines Satellite Events, Private Dinners)
BIO’s formal sessions are the public battlefield for information, but the conversations that truly reshape the landscape of business deals often take place outside of these sessions.Private dinners, satellite events, and hallway conversations are the primary sources of non-public information—what MNC BD heads say in public is usually what they “can” say in public; at private dinners, they are more likely to reveal “what we are really looking for” and “what we would never admit in public.”
The ecosystem of satellite events in San Diego is a vital component of BIO. Torrey Pines (the renowned coastal area home to the University of California, San Diego) is a hub for the world’s top life sciences research institutions, and every year during BIO, satellite scientific symposia are held around UCSD, Scripps Research, and the Salk Institute.These events are typically not on the official BIO agenda, but the concentration of participants—including actual scientists and early-stage technology decision-makers—is far higher than at the main BIO venue. For business development professionals seeking early-stage technology targets (especially delivery technologies, gene editing tools, and new target discoveries), the Torrey Pines satellite events are an absolute must-attend, representing a true value-for-money opportunity.
Strategies for attending private dinners also require careful consideration. During BIO, there are numerous “invitation-only” dinners hosted by consulting firms, law firms, CDMOs, and investment firms. Admission to these dinners usually requires advance application (recommended 6–8 weeks in advance) or is reserved exclusively for clients/partners of specific organizations.For Chinese companies, we recommend applying for admission through your legal counsel (preferably a U.S. or European law firm familiar with cross-border BD) or your BD advisory network—even if you cannot attend as a formal guest, you can still engage in “corridor follow-up” conversations with participants after the dinner concludes.
[Table 4-1] Types of High-Value On-Site Activities at BIO 2026 and Participation Strategies
| Event Type | Official/Unofficial | Information Value | Difficulty of Participation | Participation Strategy | Time Commitment |
| BIO Official Sessions (Roadshows/Forums) | Official | ★★☆☆☆ (Public Information) | Low (just buy a ticket) | Selectively attend; 1–2 highlights per track | Each session lasts 1–2 hours |
| BIO Partnering 1:1 Meetings | Official (by appointment only) | ★★★★★ (Core BD channel) | Medium to high (must book 4–6 weeks in advance) | Prepare 3 key questions + 1 follow-up action before each meeting | 30 minutes per session |
| Torrey Pines Satellite Workshop | Informal | ★★★★☆ (Cutting-edge scientific information) | Moderate (requires independent research) | Search for “Bio 2026 Torrey Pines satellite” one month in advance and check the Scripps/UCSD official websites | Each event lasts 3–4 hours |
| Private Dinner (by invitation only) | Unofficial | ★★★★★ (Preference for private events) | High (requires an invitation or connections) | Obtain invitations through legal/BD advisor networks; send the agenda in advance and request seat confirmation | Dinner lasts 3–4 hours |
| Exhibition Hall | Official | ★★★☆☆ (Company/Technical Information) | Low | Focus on visiting CDMO and delivery technology platform booths; bring business cards and prepare key questions | 2–3 hours (intensive tour) |
| Spontaneous conversations in hallways/lobbies | Informal | ★★★★☆ (High-value information that cannot be predicted) | Low (depends on initiative) | Carry business cards, practice a 2-minute “elevator pitch,” and proactively ask, “What are you looking for at BIO?” | Anytime (during spare moments) |
| On-site visit to a CDMO facility | Unofficial | ★★★★★ (CMC technical data) | High (advance booking required) | Apply for a facility tour through BIO or by contacting the CDMO directly; book 2 months in advance | Half a day to 1 day |
[Interpretation of Table 4-1] The value of the BIO venue follows the “iceberg model”: official sessions represent only the tip of the iceberg (10%), while the information and relationships that truly determine the success or failure of business development (90%) lie beneath the surface—at the Torrey Pines satellite events, private dinners, hallway conversations, and on-site factory tours.You need to start planning for these “underwater” opportunities at least 8 weeks before departure, rather than waiting until you arrive in San Diego to start looking.
4.2 Cross-Modal Insight Exercise: How to Quickly Identify Convergence Opportunities and Pain Points On-Site
The cross-modal insight exercise is a proactive thinking framework designed to help you not only listen to what your counterpart says during every BIO conversation but also quickly connect the dots to ask, “What does this information mean for my other therapeutic areas?” Below are three specific cross-modal thinking exercises you can perform during Sessions, Partnering Meetings, or even hallway conversations.
Exercise 1: Map discussions on linkers in ADC Sessions to nucleic acid drug delivery. When you hear an ADC company discussing “the payload release efficiency of cleavable linkers in the tumor microenvironment,” immediately ask yourself: Could the same endosome pH-responsive design be used to improve the endosome escape efficiency of LNP-mRNA?Could the specific lipid components used by this ADC company (such as benzene-ring-containing ionizable lipids) be adapted for nucleic acid delivery? This ability to make instant connections allows you to gain insights far beyond ADCs themselves during ADC Sessions.
Exercise 2: Map from GLP-1 CMC discussions to CGT manufacturing. The scaled-up manufacturing processes for GLP-1 biologics (especially the integration of continuous manufacturing and closed-system manufacturing) represent one of the most significant technological advancements in the CMC field over the past five years.When you hear a GLP-1 company discussing “how to increase the production efficiency of a 30,000-liter stainless steel bioreactor to over 80%,” immediately ask yourself: Can this continuous manufacturing process be transferred to the cell manufacturing process for CGT? Can the bottlenecks in CAR-T cell manufacturing (cost, batch consistency, and scaling) be alleviated by leveraging the experience from GLP-1 biomanufacturing?
Exercise 3: Map discussions on CGT AI applications to GLP-1 biomarker discovery. The AI-driven patient stratification technologies currently being developed in the CGT field (machine learning-based biomarker prediction using scRNA-seq data) can be fully transferred to precision medicine applications in the GLP-1 metabolic therapy space. Using AI to identify which T2D/obese patients respond better to GLP-1 agonists is a key pathway to enhancing a drug’s commercial value.If you encounter a promising AI company during the CGT AI Sessions, ask them: “Can your platform also be used for biomarker discovery in metabolic drugs?”—this could open up a whole new avenue for collaboration.
[Table 4-2] BIO 2026 Cross-Modal Insight Rapid Identification Framework (Printable for On-the-Go Reference)
| If you hear or see | Immediately map to | Core Issue (Ask on the Spot) | Potential BD Opportunities |
| ADC linker innovation (pH-responsive/enzyme-cleavable) | LNP endosome escape efficiency | “How does your endosome escape design differ from the pH-responsive linker design in ADCs?” | Cross-licensing of delivery technology platforms |
| Data on oral permeation enhancers for GLP-1 | Oral Delivery of Nucleic Acid Drugs | “Can SNAC-type technologies be used for the oral absorption of oligonucleotides?” | Multimodal Applications of the Permeation Enhancer Platform |
| CGT AI Patient Stratification Tool | GLP-1 Biomarker Prediction | “Does your AI biomarker platform have applications in the field of metabolic diseases?” | Commercial Licensing of AI/Biomarker Technologies in the Metabolic Disease Sector |
| LNP Manufacturing Process Optimization (Continuous Manufacturing) | Scaling up CGT cell manufacturing | “Has anyone validated continuous manufacturing processes for CGT cell manufacturing?” | Cross-modal expansion of CMC/CDMO partnerships |
| GLP-1 dual- or multi-target agonists | ADC Dual-Payload Design | “Can the design logic of multi-target synergy be applied to dual-payload combinations in ADCs?” | Early-stage technology collaboration or joint development |
| Directed Evolution of AAV Capsids | LNP Tissue Targeting | “Can the methodology of directed evolution be applied to LNP-based SORT molecular screening?” | Horizontal integration of delivery platform technologies |
| CGT Gene Editing Safety Data | Immunogenicity Risks in Gene Therapy | “Have safety risks associated with gene editing altered your rationale for selecting delivery platforms?” | Cross-sector comparison of regulatory strategies and clinical development pathways |
[Interpretation of Table 4-2] Cross-modal thinking is not the exclusive domain of geniuses; it is a skill that can be trained. We recommend printing this framework on A4 paper, carrying it with you, and spending 5 minutes filling out the “Mapping” column after each BIO conversation. After four consecutive days of practice, you will find yourself “hearing more” at the event—this is the most direct manifestation of cognitive differentiation.
4.3 Practical Information Collection Template: A Structured Recording Method Centered on “Pain Points—Pipeline—Collaboration Interest—Next Steps”
The most common mistake during BIO is “piling up business cards after meetings, only to find you can’t recall anything once you return home.” The key to solving this problem is a structured information-gathering template that can be quickly filled out on-site. We recommend using the “4P Method”: Pain Point, Pipeline, Partnership Interest, and Next Action. Below are the specific design and usage instructions for the template.
[Table 4-3] BIO 2026 On-Site Information Collection Template (Printable; fill out after each conversation)
| Field | Example of Content to Fill In | Date | Key Tips |
| Company Name | Example: Denali Therapeutics | Before the Call (Preparation) | Record both the company code and headquarters location |
| Name of the contact | Example: John Smith, VP of Business Development | Confirm Before the Call | Record the LinkedIn QR code or add contact information |
| Pain Point (the other party’s core pain point) | Example: LNP’s suboptimal extrahepatic delivery efficiency; seeking a differentiated delivery technology platform | Notes during conversation (keywords only) | Focus on recording the “challenges” or “gaps” explicitly mentioned by the other party |
| Pipeline (Pipelines/technologies of interest to the other party) | Example: GLP-1 dual-target agonist; oncology ADC (Phase I) | Notes from the conversation | Distinguish between projects the other party is actively advancing and potential collaboration targets they are seeking |
| Partnership Interest (Specific expressions of interest in collaboration) | Example: “We are looking for in-licensing opportunities in Asia for our Phase II oncology assets” | Notes from the conversation (quote verbatim) | Record the exact words as much as possible, rather than your interpretation; this is key material for follow-up emails |
| Next Action | Example: The other party has committed to sending a company presentation within one week; we need to send a draft Letter of Intent (LOI) | Before ending the conversation (must be clarified) | A conversation without a clear Next Action = an ineffective conversation |
| Your Potential Value (1–5 points) | Rate on a scale of 1–5 | Within 1 minute after the conversation ends | ≥4 points: Send a follow-up email within 24 hours of returning |
| Notes/Impressions | Example: The other party has high price expectations; decision-making cycle is 6 months or longer | After the conversation | Record “soft information” that cannot be conveyed via email |
[Interpretation of Table 4-3] The key to using this template is “immediacy”—fill it out immediately after each conversation, rather than waiting until you return to the hotel to recall the details (research shows that people forget about 50% of conversation details within two hours).We recommend using the template feature in your phone’s Notes app to create a record for each Partnering Meeting, using the template fields as headings and filling them out in real time. This way, once you return to your hotel, you’ll only need to spend 15 minutes organizing all the day’s records, rather than sifting through a pile of disorganized handwritten notes.
4.4 Informal Networking Tips and Energy Management (Avoiding Information Overload)
Informal networking during BIO is often more valuable than formal sessions, but it is also the factor most likely to drain your energy and lead to a “second-half burnout.” The San Diego Convention Center spans approximately 400,000 square meters, and completing all networking opportunities over four days requires exceptional energy management skills. Below are energy management strategies validated by numerous BIO attendees.
The “Marathon” Principle of Energy Allocation. BIO is not a 100-meter sprint, but a four-day marathon. Daily energy peaks typically occur between 9:00–11:00 AM and 3:00–5:00 PM; you should schedule your most critical business development conversations (partnering meetings, high-value sessions) during these two time slots.Lunchtime (12–1 p.m.) is usually a low-energy period, ideal for visiting the Exhibition Hall or engaging in brief “information exchange” conversations with lower-priority contacts. Evening events (receptions/dinners) mark another low-energy period—but these events often have the highest information density, so you need to reserve some energy during the day.
The 3-sentence version of your “elevator pitch.” In the BIO hallways, you’ll frequently encounter opportunities for 30-second to 2-minute “stand-up conversations.” You need to prepare a 3-sentence version you can launch at any moment:
① “Who we are” (one-sentence company positioning);
② “What we do” (one-sentence core pipeline/technology);
③ “What we’re looking for” (one-sentence collaboration needs). This version doesn’t need to be perfect—what’s needed is fluency, naturalness, and the ability to leave the other person enough room for the next step. Practice this three-sentence version until it sounds as natural as your normal speech.
[Table 4-4] BIO 2026 Daily Energy Management Schedule (Reference)
| Time Slot | Recommended Activity | Energy Expenditure | Key Points of the Strategy |
| 7:00–8:30 AM | Breakfast + Review of the Day’s Agenda | ★★☆☆☆ | Wake up 30 minutes early to review the day’s Priority Meeting list; do not skip breakfast |
| 08:30–11:00 | Priority 1 Partnering Meetings / High-Value Sessions | ★★★★★ | Peak energy period—focus only on the most important BD conversations; turn off phone notifications |
| 11:00–12:00 | Networking Hall / Low-Priority Information Exchange | ★★★☆☆ | A great time for hallway conversations; brief exchanges with CDMO booths in the Exhibition Hall |
| 12:00–1:30 PM | Lunch + Break | ★★☆☆☆ | Do not attend business lunches—this is time to recharge; you can check WeChat/emails |
| 1:30 PM–3:30 PM | Priority 1 Partnering Meetings / Q&A Sessions | ★★★★★ | Afternoon energy peak; this is the best time to ask speakers questions and build direct connections |
| 3:30 PM–5:00 PM | Torrey Pines Satellite Events / Exhibition Hall In-Depth Visits | ★★★★☆ | The satellite sessions are packed with information; be sure to check the route and schedule in advance |
| 5:00 PM–6:00 PM | Organize notes from the day + Confirm priorities | ★★☆☆☆ | Fill out the 4P log immediately upon returning to the hotel; confirm tomorrow’s appointments |
| 6:00 PM–8:00 PM | Receptions / Private Dinners | ★★★★☆ | The busiest time of the day; but also when energy levels are at their lowest—bring a bottle of water and stay hydrated |
| 8:00 PM–10:00 PM | Review the day / Prepare for the next day | ★★★☆☆ | Share the day’s biggest takeaways with team members; adjust the next day’s agenda |
| After 10:00 PM | Rest | ★☆☆☆☆ | Don’t stay up late—tomorrow’s meetings require you to be at your best |
[Interpretation of Table 4-4] Energy management is the competitive dimension that BIO attendees most often overlook. Most people begin to experience “decision fatigue” on the second or third day of BIO, causing them to perform at their worst precisely when high-quality BD conversations are most needed. By managing your energy according to the schedule above, you can maintain over 90% cognitive performance throughout the four-day conference.Remember: BIO isn’t about who runs the fastest, but who maintains the most consistent performance.
▶ Action Recommendations (Chapter IV):
- [8 Weeks Before Departure] Start planning your approach for securing invitations to the Torrey Pines satellite events and private dinners.
- 【4 Weeks Before Departure】Print 20 copies of the 4P information collection template and carry them with you.
- 【2 Weeks Before Departure】Practice your 3-sentence elevator pitch, record your version, and ask colleagues for feedback.
- 【Every Night Before Bed】Review the next day’s Priority Meeting list, rank items by priority, and strictly follow through.
5.0. biotech events san diego: Post-Event Follow-Up: Quickly Turn Business Cards into Tangible Business Progress

The real battle for BIO isn’t on the conference floor—it’s after you return home. Most of a BD’s value is realized through post-meeting follow-up emails, calls, and proposal adjustments—the four days at the conference merely establish a “first contact.” True partnerships must be solidified during the critical 48–72-hour follow-up window after the event. This chapter provides a 48-hour follow-up template, a framework for building internal influence, long-term relationship maintenance strategies, and special risk warnings in the geopolitical context.
5.1 The 48-Hour Follow-Up Rule: Personalized Email Templates for the U.S. Market (Emphasize Specific Meeting Details, Avoid AI-Generated Tone)
The 48-hour window is the most critical time for BD follow-ups. Psychological research shows that people’s memories and emotional connections to “new contacts” significantly fade after 48–72 hours; if no follow-up occurs within a week, the relationship’s momentum drops by approximately 60%. For BD follow-up emails targeting the U.S. market, the following three principles must be strictly adhered to:
Principle 1: Cite specific meeting details. U.S. BD professionals receive 50–100 BD emails daily and can instantly spot “mass-sent template emails.” Your follow-up email must include specific information that would only appear in an in-person conversation:
① Specific pain points the other party mentioned during the conversation (quote their exact words);
② Specific technical issues you discussed;
③ Specific next steps you agreed upon. The combination of these three elements is the strongest proof that your email is “personalized.”
Principle 2: Provide concrete value, not requests. It is a cultural norm in the U.S. market (especially in the BD field) not to make any direct requests in the first follow-up email. First, provide value—such as a paper they might find interesting, additional data about your technology, or an infographic that helps them understand the value of your platform—and then naturally introduce a proposal for the next steps in your collaboration.
Principle 3: The email subject line must be personalized. Avoid generic subject lines like “BIO 2026 Follow-up” or “Following up from BIO.”Recommended format: “[The specific pain point we discussed] — [Your asset name] follow-up from BIO” (e.g., “LNP extrahepatic delivery challenges we discussed — AOC platform follow-up from BIO”). Subject lines like this have a 3–4 times higher open rate than generic ones.
[Table 5-1] 48-Hour Follow-Up Email Template (U.S. BD Market, Ready-to-Use Version)
| Email Type | Email Subject Line Format | Core Content Structure | Word Count Requirement |
| First Follow-Up Email (High-Priority Conversations) | [Specific pain points mentioned by the recipient] — [Name of your core asset/technology] Follow-up from BIO 2026 | ① Quote the recipient’s exact words (1–2 sentences) ② Provide concrete value (data/references/additional information) ③ Reiterate next steps ④ Clear Call-to-Action | 150–250 words |
| First Follow-Up Email (Medium-Priority Conversation) | [The specific technical issue you discussed] — follow-up from BIO 2026 | ① Thank them and mention the key points of your conversation ② Provide relevant supplementary materials ③ Extend an invitation to connect again | 80–150 words |
| Resource-sharing follow-up (low priority/first contact) | [Title of relevant literature/data] — Thought you might find this useful from BIO 2026 | ① Brief explanation for sharing ② Attach the material ③ Casually mention a desire to stay in touch | 50–100 words |
| LinkedIn connection request | Great to have met you at BIO 2026 — [Your area of expertise] | ① Mention a specific conversation (1 sentence) ② Explain your reason for wanting to stay in touch | 300 characters or fewer (LinkedIn limit) |
[Original Template Example: First Follow-Up Email (High-Priority Conversation)]
Subject: Your concern regarding [specific challenge they mentioned] — Follow-up on [Your Asset Name] from BIO 2026
Hi [Name],
It was a pleasure speaking with you at BIO 2026. I particularly appreciated your point about [quote the specific pain point mentioned during the conversation] — this is exactly the challenge we set out to solve with [your core asset name/technology platform].
[Provide 1 specific value proposition: Share specific data, technical parameters, or clinical information that piqued their interest during the conversation. Format: “Based on our conversation, I wanted to share some additional data on [specific information] — [provide the data or indicate where it can be found].”]
As discussed, the next step would be for our [specific role, e.g., Chief Medical Officer] to arrange a follow-up call with your team to discuss [specific next steps]. Would [specific date, e.g., next Wednesday or Thursday] work for a 30-minute call?
I’ve attached our [latest version of the Executive Summary / Partnering Deck] for your reference.
Looking forward to continuing our conversation.
Best regards,
[Your Name]
[Your Title]
[Company Name]
[Phone] | [LinkedIn Profile URL]
[Interpretation of Table 5-1] The value of a template isn’t to have you copy it mechanically, but to provide structure and logic—the specific content must be filled in based on the actual context of each conversation. “Quoting the other person’s exact words” is the most critical signal of personalization; without it, any email will become just another mass-sent template. If you’re unsure what the other person said during the conversation, it’s better not to send an email at all than to send a hollow template—an empty email does more harm than no email at all.
5.2 Building Internal Influence: A Ready-to-Use “BIO 2026 Insights Report” Framework to Help Your Team and Boss Quickly See the Value
The value of attending BIO lies not only in the business development progress you achieve but also in the insights and knowledge you can share with your internal team. A high-quality “BIO 2026 Insights Report” not only helps non-attendees understand industry trends but also reinforces your position as an “industry expert” within the company, securing more resources for next year’s attendance. Below are design recommendations for the report framework.
Core design principles for the report framework: Actionable > Comprehensive > Detailed. Most executives at Chinese companies do not have time to read a 50-page BIO conference report. Your report should ensure that non-attendees can understand the top 3 industry trends, the top 2 risks, the top 3 opportunities, and the most critical action recommendation within 10 minutes.
[Table 5-2] BIO 2026 Insights Report Framework (Ready-to-Use Version)
| Report Sections | Content Requirements | Page Count (Recommended) | Target Audience |
| Executive Summary | 3 Key Trends + 2 Major Risks + 3 Major Opportunities + 1 Key Action Recommendation | 1 page | CEO/CFO/Head of Business Development |
| Chapter 1: Market Segment Heat Assessment | “Thermometer” Scores (1–10) for the Four Major Sectors: ADC, CGT, GLP-1, and Nucleic Acid Drugs + Key Data Support | 2–3 pages | BD Team + R&D Team |
| Chapter 2: Changes in the Competitive Landscape | Specific case studies (3–5) on new entrants, technological breakthroughs, and shifts in valuation logic | 3–4 pages | BD Team + Strategic Planning |
| Chapter 3: Cross-Industry Convergence Opportunities | Cross-modal opportunities we have identified + Implications for our existing pipeline + Recommended areas for exploration | 2–3 pages | BD Team + R&D Team + Executive Leadership |
| Chapter 4: Detailed BD Intelligence | Detailed evaluation of each target company (ranked by priority), our outreach efforts, their feedback, and follow-up plans | 5–8 pages | BD Head + Legal + Executive Management |
| Chapter 5: My Personal Observations | The 3 most valuable cognitive biases I’ve learned, 2 underestimated opportunities, and 1 critical reminder | 1–2 pages | CEO (Personal Observations) |
[Interpretation of Table 5-2] The Executive Summary is the “face” of the entire report—it must be written by you personally, not compiled by an assistant.The CEO typically reads only this page. The BD Intelligence section in Chapter 4 represents the report’s core value, but it is also the most sensitive—it is recommended to mark it “Confidential” and restrict its distribution. Chapter 5 (Personal Observations) is the best chapter to showcase your independent thinking; it is recommended to include some “contrarian” insights—opportunities or risks you identified on-site during the BIO that most people have not recognized.
5.3 Long-Term Relationship Management: Leverage BIO as a Tool in Your Annual Execution Cycle, Not a One-Time Event
The most valuable aspect of BIO is not a one-time interaction in a given year, but the long-term industry network built through consistent attendance. A BD professional with over 10 years of BIO attendance typically possesses a stable network of relationships spanning the decision-making levels of major global biotech and pharmaceutical companies—a strategic asset that cannot be measured in monetary terms. Below are specific strategies for building this long-term network.
Strategy 1: BIO is the starting point of the annual engagement cycle, not the endpoint. After establishing contacts at BIO each year, maintain communication at the following time points in the following year:
① One month after BIO concludes (send follow-up materials);
② Mid-Q3 (send industry news commentary relevant to the contact);
③ Two months before BIO (invite them to your company’s events or ask them to recommend suitable contacts). This “four-times-a-year” engagement rhythm ensures that by the time the next BIO rolls around, the other party has already built up a significant level of familiarity with your company.
Strategy 2: Create your “BIO Relationship Calendar.” After BIO concludes each year, use Excel or a CRM tool (such as Salesforce or HubSpot) to create a comprehensive “BIO Relationship Calendar”: each row records a contact, including name, company, title, year of first contact, date of last contact, summary of interaction, next contact plan, and next contact date.This system serves as your “battle map” for long-term relationship management—without it, you’ll find that most BIO contacts have gone “off the grid” within 2–3 years.
[Table 5-3] Annual Relationship Maintenance Calendar (BIO Relationship Management Reference Template)
| Timeline | Action | Details | Estimated Time |
| During BIO (June) | Create contact profiles | Create a profile for each new contact: Name + Company + Position + Summary of interaction + Plan for next contact | 3 minutes per contact |
| 1 month after BIO (July) | Send follow-up materials | Send supplementary materials/literature/invitations based on commitments made during the conversation | 5 minutes per contact |
| Mid-Q3 (August–September) | Send an industry insights email | Send 1–2 industry articles or commentary pieces that you think the recipient might find interesting | 3 minutes per contact |
| Early Q4 (October–November) | Prepare to launch BIO | Send your BIO goals for the coming year to give them advance notice (to increase the chances of scheduling a Partnering Meeting) | 3 minutes per contact |
| Late Q4 (December) | Holiday/Year-End Greetings | Send personalized year-end greetings (keep them brief and sincere; avoid mass emails) | 2 minutes per contact |
| Q1 of the following year (January–March) | Ongoing follow-up | Notify high-priority contacts immediately if there is progress on the pipeline | As needed |
| Two months prior to the following year’s BIO (April–May) | BIO scheduling | Schedule BIO Partnering Meetings with high-priority contacts in advance | 2 minutes per contact |
[Interpretation of Table 5-3] The essence of relationship maintenance is “continuously providing value”—it doesn’t mean every interaction must have a commercial purpose, but rather that you should periodically ensure high-priority contacts “see you.”A useful rule of thumb is: with every contact, provide at least one piece of “non-commercial value” that the other party might find interesting (such as industry insights, technical commentary, or third-party referrals), and then naturally transition into a business conversation. This “give before you take” approach to relationship management is a core principle of U.S. business development culture.
5.4 Risk Alert: Key Practical Considerations for IP Protection, Supply Chain Security, and Compliance in Cross-Border Licensing Amid the Current Geopolitical Landscape
The geopolitical environment is an unavoidable issue for Chinese companies attending BIO in 2024–2026. Tighter scrutiny by CFIUS (Committee on Foreign Investment in the United States) in the biopharmaceutical sector, concerns over data security in U.S. clinical trials conducted by Chinese biotech firms, and a “trust deficit” toward Chinese companies held by some U.S. institutions are all influencing the pace and structure of U.S.-China BD transactions. Below are the key risk points and response strategies you must understand during BIO 2026.
Core Principle of IP Protection: Prioritize Patent Portfolios Over BD Discussions. Before engaging in any business development (BD) discussions, ensure that your core assets have completed patent applications in the U.S., Europe, and Japan (or have at least entered the National Phase). The patent application process at the U.S. Patent and Trademark Office (USPTO) takes approximately 2–3 years. Disclosing technical details during discussions without having filed a patent application will expose you to irreversible IP risks.It is recommended to adopt a “data room” approach during BIO discussions: do not discuss specific parameters of core technologies in public settings; only present information within the scope of what is “publicly disclosable.”
Practical recommendations for supply chain security. If your BD transaction involves commercialization in the U.S. market, you must consider the source disclosure requirements for the “Chinese supply chain.”The FDA’s Drug Supply Chain Security Act (DSCSA) requires traceability across all nodes of the pharmaceutical supply chain—if your active pharmaceutical ingredients (APIs) or critical excipients come from Chinese suppliers, you must prepare supply chain transparency documentation in advance. For CMC collaborations, you must also assess the feasibility of “nearshore alternatives” in advance—proactively mentioning your supply chain “de-risking” plan during BD negotiations can significantly reduce uncertainty in MNC decision-making.
[Table 5-4] Key Risk Matrix and Mitigation Strategies for U.S.-China BD Transactions in 2026
| Risk Type | Specific Risk Description | 2026 Risk Level | Countermeasures | Response Scripts for BIO Dialogue |
| CFIUS Review Risk | Transactions involving U.S. Protected Health Information (PHI) or biosecurity-related technologies | Medium to high (depending on asset type) | Conduct CFIUS pre-screening in advance; design transaction structures to avoid review (e.g., technology licensing vs. equity transactions) | “We have structured our licensing deal as a pure technology license without an equity component, which typically does not trigger CFIUS review.” |
| Data Security/Privacy Risks | BD transactions involving U.S. patient data or genetic data | High (for CGT/gene therapy) | Prepare a data de-identification plan and a Data Security Management Plan (DSMP); consider data transfer restrictions | “Our clinical data is stored in [specific location] and we have a GDPR/China PIPL-compliant data management protocol.” |
| Concerns about IP theft | Systemic distrust among U.S. MNCs/VCs regarding Chinese companies’ ability to protect IP | Moderate (varies by company reputation) | Provide comprehensive evidence of IP protection (patent portfolio, technical confidentiality systems, confidentiality clauses in partnership agreements) | “We hold US-registered patents for [specific IP], and our technical team operates under strict NDA protocols.” |
| Supply Chain Source Disclosure | FDA DSCSA Supply Chain Traceability Requirements | Medium (depends on whether U.S. commercialization is involved) | Prepare a supply chain map in advance; proactively develop “de-China” alternatives | “Our API is manufactured in [non-China location] with full cGMP certification for the U.S. market.” |
| Biosafety-Related Restrictions | Transactions involving viral vectors, biological samples, and specific biotechnologies may be restricted | Moderate (depending on the specific technology) | Familiarize yourself in advance with the U.S. Commerce Control List (CCL) and the BIS Entity List | “Our technology falls under [specific category], and we have confirmed it does not fall under [specific export control category].” |
[Interpretation of Table 5-4] None of the above risks imply that “China-U.S. business development (BD) transactions cannot be conducted”—but each requires you to be prepared before engaging in BD discussions. At BIO events, when faced with sensitive questions from U.S. partners, having prepared, candid, transparent, and verifiable answers in advance is the fastest way to build trust.Remember: The concerns of U.S. business development professionals are often not due to bias against your company, but rather their own compliance responsibilities to their internal management and board of directors—helping them address this issue is the best way to secure a partnership opportunity for yourself.
▶ Action Recommendations (Chapter V):
- 【Within 24 hours of returning】Send follow-up emails for all high-priority conversations (the 48-hour golden window).
- 【Within 1 week of returning】Complete the first draft of the “BIO 2026 Insights Report” and present it to the BD Head and senior management.
- 【Within 2 weeks of returning】Establish a comprehensive BIO relationship database (Excel or CRM) and populate it with initial contact information.
- 【Throughout the Year】 Continuously follow up with high-priority contacts according to the milestones in the relationship maintenance calendar.
6.0. biotech events san diego: Conclusion: BIO 2026 is not the end goal, but a purpose-driven long-term lever
6.1 To Chinese and Asian Players: How to Win Over Partners on the Global Stage with a “Purpose-Driven” Narrative
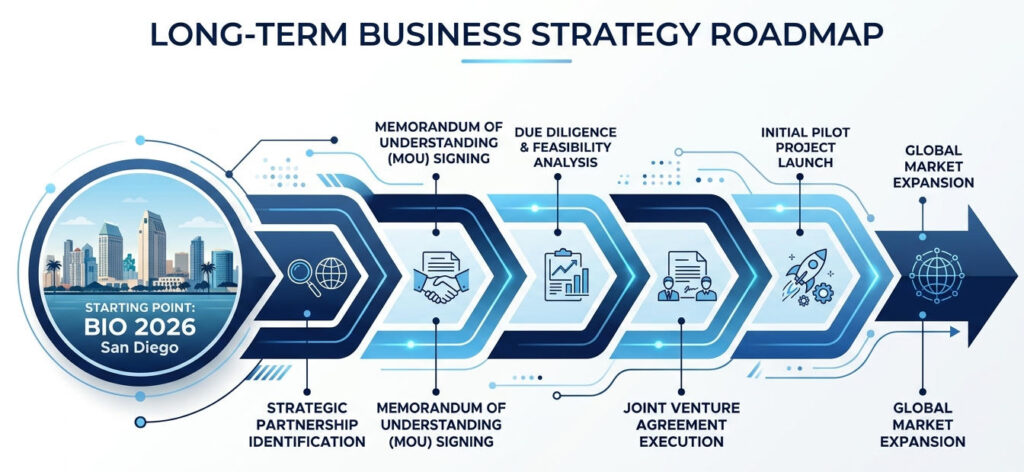
For attendees from China and Asia, the greatest challenge at BIO conferences is often not a technological gap, but a “narrative gap.”When pitching, founders of Western biotech companies typically follow a well-established narrative logic: starting with unmet medical needs, demonstrating the differentiation of their technology platform, presenting clinical data, and finally outlining a commercialization roadmap. Behind this narrative structure lies a deep alignment with the Western business culture’s emphasis on being “purpose-driven”—it is not merely a pitching technique, but a core business value.
In contrast, BD pitches by Chinese companies often overemphasize technical parameters (targets, molecular activity, IC50) and regulatory milestones (IND approval, clinical enrollment), while neglecting the fundamental question: “Why is this pipeline important?”This is not because Chinese companies do not value patient outcomes—rather, it stems from a structural difference between the “pragmatic” narrative tradition in Chinese business culture and the “meaning-first” narrative style prevalent in U.S. BD culture. During BD discussions at BIO 2026, learning to articulate “which patients your drug can help and to what extent” in a way that resonates with Western audiences can boost the effectiveness of your pitch by at least 30%.
Specifically, we recommend incorporating the following “narrative enhancement” elements into your pitch deck:
① Patient Story: Use 1–2 slides to describe the real-life circumstances of the target patient population for the indication (e.g., quality of life scores, symptom burden, financial strain), and quantify the magnitude of unmet need;
② Clinical Differentiation Narrative: Instead of simply stating “Our ORR is 50%,” explain that “following failure of current standard treatments, our drug candidate shrinks tumors and maintains remission in these patients for an average of X months longer than the current best therapy—and what those X months mean for them”;
③ Global Access Vision: Proactively mention your pricing strategy and market access plans to demonstrate that you are not merely “selling a drug,” but rather building a treatment regimen that benefits patients worldwide.
[Table 6-1] Comparison Checklist for “Narrative Enhancement” in Chinese Company Pitch Decks
| Traditional Narrative (Missing Elements) | Narrative-Enhanced Version (Added Elements) | Example Content | Impact on BD Outcomes |
| Lists only targets and molecular activity | → Add description of unmet patient needs | “Currently, only 35% of T2D patients achieve HbA1c control, and the median annual cost of existing GLP-1 therapies is $12,000, which is beyond the financial means of most patients” | Help the audience establish a cognitive foundation for “why this problem is worth solving” |
| Listing only clinical data (ORR/PFS) | → Add the practical significance of the data for patients | “Our candidate drug extends PFS from 6 months to 11 months. For patients, these additional 5 months mean being able to attend 3 family gatherings and complete 2 trips.” | Make the data relatable and emotionally resonant |
| Listing only market size (TAM) | → Add patient flow and accessibility analysis | “We plan to use differentiated pricing to increase treatment accessibility from the current market’s 15% to 40%, covering low- and middle-income patient groups.” | Demonstrate your deep understanding of market access to alleviate commercialization concerns |
| Listing only the competitive landscape | → Add your unique value proposition (UVP) | “Unlike all existing competitors, our core differentiation lies in [specific mechanism], which enables us to achieve efficacy in [specific patient subgroup] that competitors cannot match” | Shift from “We are in this therapeutic area” to “Our unique position within this therapeutic area” |
| No explanation of IP sources | → Add a statement on IP independence and risk disclosure | “Our core technology platform is protected by [country] patents, with an expiration date of [year], and we have verified the technology’s originality and freedom to operate (FTO).” | Proactively disclose IP information to address the primary concerns of U.S. partners |
[Interpretation of Table 6-1] Narrative enhancement is not about “telling a story,” but about effectively conveying the maximum value of your technology within the context of Western business culture. The greatest BD opportunities for Chinese companies often come from niche markets that are unfamiliar to Western MNCs but possess genuine clinical value—in such markets, “narrative value” (enabling the other party to quickly understand and recognize the market’s potential) is more important than the “technical value” itself.A strong narrative can enable a Chinese company with a technical value of 9 but a narrative capability of 3 to secure business development opportunities comparable to those of a Western company with a technical value of 8 and a narrative capability of 8.
6.2 Final Call to Action: Registration Is Just the Beginning; Preparing Your Value Proposition and Acting Immediately Is the Key to Success
The battle for BIO 2026 begins the moment you finish reading this article—not when you set foot in San Diego. Among the 21,000 attendees, how many have prepared a high-quality Partnering Profile? How many started scheduling Partnering Meetings 70 days in advance? How many arrive at the venue with clear, four-category KPI goals?
This article isn’t just for reading—it’s an action plan. We’ve distilled all our recommendations into the following “Top 10 Action Checklist.” Please complete each item over the next 70 days:
[Table 6-2] BIO 2026 Top 10 Action Checklist for Conference Preparation (Check off each item before departure)
| No. | Action Item | Execution Standard | Deadline | Status |
| 1 | Complete Partnering Profile Optimization (Asset-Centric Version) | Each asset entry must be ≥200 words, including a unique description, key data, and willingness to partner | 6 weeks prior to departure | ☐ |
| 2 | Establish an IPP (Ideal Partner Profile) database | Complete information on at least 20 target companies (contacts, decision-makers, preferences) | 5 weeks before departure | ☐ |
| 3 | Schedule at least 15 Partnering Meetings | Use the BIO Partnering system to search and prioritize by granularity | 4–6 weeks before departure | ☐ |
| 4 | Finalize the Pitch Deck (including narrative enhancement elements) | Includes Patient Story, Clinical Differentiation Narrative, and Global Access Vision | 4 weeks before departure | ☐ |
| 5 | Arrange channels for participating in Torrey Pines satellite events | Identify at least 2 worthwhile satellite workshops and complete registration | 4 weeks before departure | ☐ |
| 6 | Complete IP self-assessment (patent portfolio + FTO analysis) | Confirm the patent status, FTO risks, and CFIUS pre-screening results for core assets | 4 weeks before departure | ☐ |
| 7 | Prepare 4P information collection templates (20 or more) | Print or create a mobile Notes template; fill it out after each conversation | 3 weeks before departure | ☐ |
| 8 | Complete Bio 2026 app installation and agenda customization | Set treatment areas and modality preferences; bookmark Priority Sessions | 2 weeks before departure | ☐ |
| 9 | Practice your 3-sentence elevator pitch | Record a video and ask two colleagues for feedback to ensure it sounds fluent and natural | 2 weeks before departure | ☐ |
| 10 | Prepare a 48-hour follow-up email template | Using the template in Table 5-1, pre-fill draft follow-up emails for high-priority companies | 1 week before departure | ☐ |
2026 marks the “Year of Execution” for the global biopharmaceutical industry—a shift from storytelling to delivering results. BIO 2026 is the most important asset on your report card. Prepare your value proposition and start executing immediately.
See you in San Diego.
▶ Take action now: Start with the first item on the Top 10 Checklist in this article and complete it today!
7.0. biotech events san diego: Appendix: Frequently Asked Questions (FAQ)—A Practical Guide to Attending BIO 2026

The following FAQs are compiled from the most common questions asked by BIO 2026 attendees for your quick reference. If you have additional questions, please feel free to contact us via the Bio Conference Central platform.
Q1: When does the BIO Partnering system open for scheduling?
A1: The BIO Partnering system typically opens for scheduling 6–8 weeks before the conference begins. We recommend starting to book meetings with popular companies 4–6 weeks in advance, as the schedules for MNC BD teams and large biotech companies are usually fully booked within 48 hours of opening.
Q2: What is the biggest misconception Chinese companies have about attending BIO?
A2: The biggest misconception is the “wait-and-see” approach—failing to prepare a Partnering Profile, a list of target companies, and a Pitch Deck in advance, and only starting to look for partners once at the venue. This is akin to going into battle unarmed. We recommend starting systematic preparations at least 70 days in advance.
Q3: How can you determine during BIO whether an AI company is truly valuable?
A3: Three key questions:
① Have your AI predictions been validated through prospective clinical trials? (Relying solely on retrospective data analysis is insufficient.)
② Is the ownership of intellectual property for AI-generated sequences/biomarkers clearly defined? (To avoid IP disputes)
③ Is the cost of AI-assisted decision-making lower than the incremental clinical/commercial value it delivers? (Calculate actual ROI)
Q4: Which companies are currently the most advanced in AOC (antibody-oligonucleotide conjugate) development?
A4: The most noteworthy companies are Avidity Biosciences (most advanced, with Phase II clinical data available), Denali Therapeutics (in collaboration with Secarna), and Tallac Therapeutics (possessing a unique TLR agonist-AOC platform). There are currently no approved AOC products; the field is in a critical phase of clinical proof-of-concept.
Q5: What are the most noteworthy developments to watch in the GLP-1 space in 2026?
A5: The three most noteworthy developments are:
① Orforglipron (oral small-molecule GLP-1, Eli Lilly Phase III) — If successful, it will disrupt the entire GLP-1 market landscape;
② Retatrutide (triple-target agonist, Eli Lilly Phase III) — boasts the most aggressive weight loss data;
③ Phase III data for GLP-1 in NASH/MASH indications — the market size is 3–4 times that of obesity.
Q6: What are the most frequently asked IP questions Chinese companies face during BD negotiations in the U.S.? How should they be addressed?
A6: The most common questions are: “Are your patents registered in the U.S.? Is your technology free-to-operate (FTO)?” Standard response template:
① Clearly state the patent registration locations (U.S., Europe, Japan, etc.) and expiration dates;
② Explain the results of the FTO analysis;
③ If there are issues (e.g., partial reliance on third-party patents), proactively disclose them and provide solutions (licensing or design-around).
Q7: How can one quickly identify “convergence opportunities” at BIO?
A7: Use the “Cross-Modal Insight Exercise” framework provided in Chapter IV of this document—during each session, immediately ask “What does this technology mean for nucleic acid therapeutics?” when ADC discussions arise; immediately ask “Can this process be transferred to CGT manufacturing?” when GLP-1 CMC discussions occur. Print Table 4-2 in advance, keep it with you, and fill in the “Mapping” column after each conversation.
Q8: What is the most important thing in a 48-hour follow-up email?
A8: Quote the exact words the other party used during the conversation (Pain Point). This is the strongest evidence that you “actually spoke with them,” and the most direct signal that sets you apart from “mass-sent template emails.” Follow-up emails without direct quotes have an open rate of less than 5%.
End of article. BIO 2026, see you in San Diego.







