- 1.0 bio conference boston: Strategic Introduction — Why 2026 Is a Biopharma "Turning Point"
- 2.0 Technological Depth at bio conference boston: ADCs, Bispecifics & Nucleic Acid Evolution
- 3.0 bio conference boston Pitfall Guide: Reading Between the Lines at PEGS
- 4.0 bio conference boston Attendee Roadmap: Maximizing Your 120 Hours
- 5.0 Regulation & Future at bio conference boston: FDA Trends & Next-Gen Targets
- 6.0 bio conference boston Conclusion: Your Organizational Action Plan
- 7.0 bio conference boston FAQ: PEGS 2026 Essential Questions Answered
1.0 bio conference boston: Strategic Introduction — Why 2026 Is a Biopharma “Turning Point”
1.1 From “Quantity” to “Qualitative Shift”: PEGS Is No Longer Just an Academic Exchange, but a “Preview Stage” for Global Pipeline Transactions
PEGS Boston 2026. bio conference boston. Every May, those of us in the Boston biotech community say the same thing: “During PEGS week, you can’t book a hotel in the Seaport.” This is no exaggeration. When you walk into the lobby of the Omni Boston Hotel at the Seaport, you’re not met with the quiet atmosphere of an academic conference, but rather with BD directors and R&D VPs holding coffee, huddled in small groups in the corners, whispering about early-stage clinical data that hasn’t been publicly disclosed yet.

Boston biotech summit. With an attendance of 3,500 to 4,000 people, the venue is consistently at full capacity, and the hallways are packed—but these numbers alone mean little. What truly sets PEGS Boston apart is this: it is not an ivory tower of academic exchange, but a testing ground for global pipeline deals involving ADCs, BsADCs, and AOCs.
biopharmaceutical conference Boston. Last year, I heard early-stage data on a BsADC here, and it completely overturned my team’s Q3 plans on the spot.The speaker was the CSO of a mid-sized biotech company; he mentioned it to me during a coffee break, his tone as matter-of-fact as if he were discussing the weather. But this kind of “upheaval” happens every day at PEGS. Why? Because when you’re simultaneously facing a dozen teams advancing bispecific ADC projects, and when you can schedule coffee meetings with the heads of five payload technology platforms within two days, the traditional pace of R&D decision-making simply isn’t enough.
protein engineering summit. The paradigm shift of 2026 begins with the end of the homogenized competition in traditional ADCs. Over the past five years, homogenized competition around targets like HER2, TROP2, and CLDN18.2 has completely closed the window of opportunity for first-generation ADCs.But at the same time, the wave of converging technologies is reshaping the rules of the game—BsADCs (bispecific antibody-drug conjugates) are no longer just proof-of-concept, AOCs (antibody-oligonucleotide conjugates) are opening up a whole new battlefield in non-oncology indications, and GenAI-driven protein engineering has moved out of the lab and into the industrial translation phase.
Table 1-1: PEGS Boston 2026 Key Information Overview
| Program | Details |
| Full Conference Name | PEGS Boston 2026 (22nd Protein and Antibody Engineering Summit) |
| Dates | May 11–15, 2026 (5 days) |
| Venue | Omni Boston Hotel at the Seaport, Boston, USA |
| Attendance | Approximately 3,500–4,000 attendees (consistently sold out; the event has a significant influence within Boston’s biotech community) |
| Key Topics | ADC, Antibody Engineering, Immunotherapy, Bispecific Antibodies, Macromolecular Analysis and Manufacturing |
| Official Website | https://www.pegsummit.com/ |
| 2026 Keywords | Convergence, BsADC, AOC, GenAI, Smart Biologics |
1.2 2026 Core Keyword: “Convergence”
Boston life sciences events. “Convergence”—this is the most central keyword for PEGS in 2026. It is not merely a simple layering of technologies, but a complete breaking down of the boundaries between macromolecules, small molecules, and nucleic acid therapeutics.
Novartis’s acquisition of Avidity Biosciences in August 2024 for a total consideration of approximately $1.15 billion was driven precisely by the breakthrough potential of the AOC platform in the field of muscular diseases—combining the precise targeting capabilities of antibodies with the gene regulation capabilities of oligonucleotides. The signal sent by this transaction could not be clearer: the cross-disciplinary convergence of macromolecules and nucleic acid therapeutics has moved from an academic concept to the agenda of industrial mergers and acquisitions.
Those who remain isolated in traditional ADC development will likely see their pipelines fall behind by the end of 2026. Simple ADCs or bispecific antibodies are no longer sufficient; integration with AOC, GenAI, and smart peptides is essential—this is not alarmist rhetoric, but a clear signal emerging from the current technological trajectory. At Omni Boston Seaport in May 2026, this signal will be presented to every attendee with unparalleled clarity.
Table 1-2: Three Major Signals of the 2026 Biopharmaceutical “Tipping Point”
| Signal | Specific Manifestations | Implications for Attendees |
| Accelerated Technology Convergence | Novartis-Avidity AOC deal ($1.15 billion), surge in early-stage clinical data for BsADCs | We can no longer view individual technologies in isolation; we must establish a cross-disciplinary cognitive framework |
| The End of the Traditional ADC Race to the Bottom | Homogeneous competition in HER2/TROP2/CLDN18.2 has reached saturation, making it difficult for me-too projects to secure funding | Focus on differentiated innovation; be wary of technical approaches that are losing their edge |
| AI-driven design has matured | GenAI moves from the lab to the industry; Plenary Fireside Chat focuses on smart biologics | 2026 is a critical window for establishing the concept of AI + Biologics |
1.3 Value of This Article: A Practical Framework Accessible to Non-Experts
This article is unlike any other PEGS coverage you’ll find online. It doesn’t copy-paste official agendas, list speaker bios, or pile on sleep-inducing technical specs. What I’m offering you is a practical framework that allows non-experts to attend, debrief, and implement insights just like “industry veterans.”
Four Unique Value Elements: Trend Analysis + Pitfall Avoidance Checklist + 120-Hour Conference Roadmap + Action Plan for Your Organization—these are the things you should actually take away after spending thousands of dollars on registration fees and flying over ten hours to Boston.
Table 1-3: Overview of the Practical Framework in This Article
| Module | Core Value | Target Audience |
| I. Strategic Introduction | Establish an understanding of the 2026 “turning point” and why this year is critical | All attendees |
| II. Technical Insights | In-depth analysis of four cutting-edge technologies: ADC 3.0, AOC, bispecific antibodies, and GenAI | R&D, Business Development, Investors |
| III. Avoiding Pitfalls: The “Blacklist” | Identify downplayed technology paths and decipher the “subtext” of speakers | Investors, Business Development Professionals, Strategic Decision-Makers |
| IV. The 120-Hour Roadmap | Role-Specific Roadmaps + Practical Networking Skills | All Attendees |
| V. Regulation and the Future | FDA Trends and Outlook on Next-Generation Targets | Regulatory Affairs, Strategic Planning |
| VI. Action List | 3D Reporting Template to Take Back to Your Organization | All Attendees |
Below, I will provide a comprehensive overview of the practical trends for 2026.
2.0 Technological Depth at bio conference boston: ADCs, Bispecifics & Nucleic Acid Evolution
The technical agenda at PEGS Boston expands every year, but the 2026 agenda sends a clear signal: boundaries are dissolving. While traditional tracks—such as “ADC,” “Bispecific Antibodies,” and “Immunotherapy”—still exist, there are increasing points of overlap between them, and cross-disciplinary integration themes are gaining greater prominence. This is no coincidence; it reflects the industry’s technological evolution as manifested in the conference agenda.
For attendees, understanding this “evolutionary” logic is more important than memorizing any specific technical parameters. The following sections will explore the four major technology segments—ADC 3.0, AOC, bispecific antibodies, and GenAI—one by one. Each section will outline the practical insights you can take away after attending the relevant sessions.
2.1 ADC 3.0: A Technical Pathway to Address Homogenization and Intensifying Competition
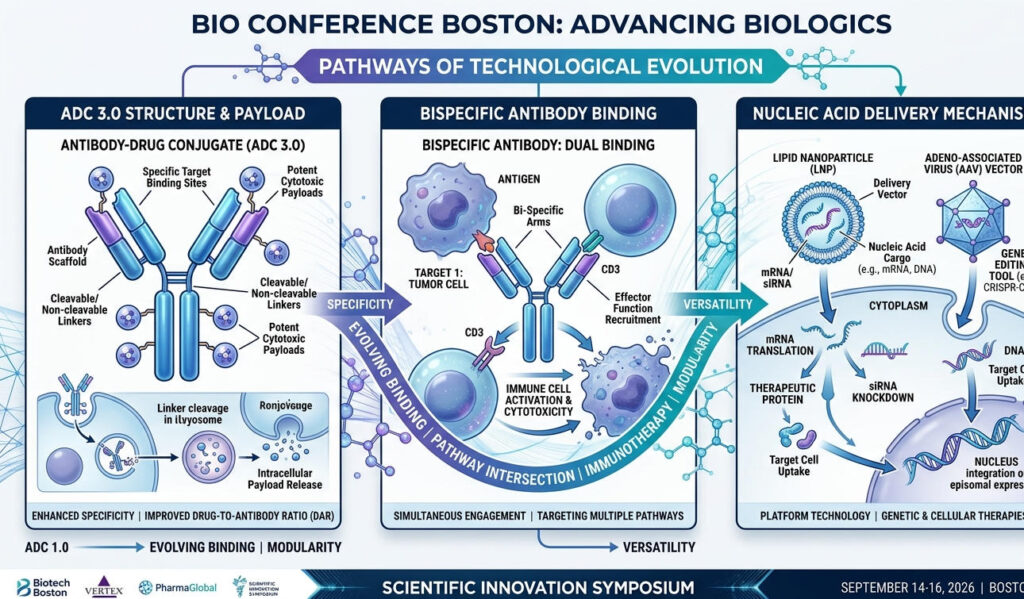
First-generation ADCs (represented by T-DM1) demonstrated proof of concept, while second-generation ADCs (represented by Enhertu) achieved breakthroughs in efficacy through optimizations of the linker and payload. However, the core proposition of third-generation ADCs—ADC 3.0—is how to achieve differentiated competition in an environment of target homogeneity.
The “Driving Clinical Success in ADC” track at PEGS Boston 2026 is centered precisely on this proposition. Based on the published agenda, two technical pathways warrant particular attention this year.
Table 2-1: Comparison of ADC Generational Technology Evolution
| Dimension | ADC 1.0 | ADC 2.0 | ADC 3.0 |
| Representative Drugs | T-DM1 (Kadcyla) | Enhertu (DS-8201) | iADC, BsADC, RDC |
| Key Breakthroughs | Demonstrating the feasibility of the ADC concept | Cleavable linker, high DAR | Immune stimulation, dual-target, cross-boundary payload |
| Payload Types | Primarily microtubule inhibitors | DNA-damaging agents + microtubule inhibitors | Immune agonists, radioisotopes |
| Key Challenges | Limited efficacy, significant toxicity | Homogeneous competition, drug resistance issues | Complex CMC and Target Selection Strategies |
| 2026 Focus | Outdated | Red ocean competition | Blue Ocean Opportunities |
2.1.1 Payload Revolution: The Shift from Traditional Cytotoxins to Immune Agonists (iADCs) and Radioisotopes
The payloads of traditional ADCs are primarily microtubule inhibitors (such as MMAE and MMAF) and DNA-damaging agents (such as DM1 and DXd), with a simple and direct mechanism of action: precisely delivering chemotherapy drugs to tumor cells. However, the limitations of this approach are evident—the immunosuppressive state of the solid tumor microenvironment makes it difficult for simple cell killing to yield lasting benefits.
Immune-activating ADCs (iADCs) are changing this landscape. Their payloads are no longer cytotoxins, but rather immune-activating molecules such as TLR agonists and STING agonists.The design logic is as follows: the antibody handles precise targeting, while the payload transforms “cold tumors” into “hot tumors,” creating conditions for subsequent immunotherapy. AstraZeneca’s AZD8205 (an iADC targeting B7-H4) has already entered clinical trials, with early data showing signs of immune activation.
Radioisotope-conjugated drugs (RDCs) represent another cross-disciplinary direction. Compared to cytotoxic payloads, radioisotopes exhibit a stronger bystander effect and deliver more thorough killing of heterogeneous tumors. The success of Novartis’s Pluvicto (a Lu-177-labeled PSMA-targeted RDC) in the field of prostate cancer has already demonstrated the commercial viability of this approach.At PEGS 2026, more technical discussions are expected regarding alpha-emitters (Ac-225) versus beta-emitters (Lu-177).
Table 2-2: Technical Characteristics of Cross-Border Payload Approaches and 2026 Trend Interest
| Payload Type | Mechanism of Action | Representative Drugs/Platforms | 2026 PEGS Interest Level |
| Immune agonists (iADC) | Activates immune responses in the tumor microenvironment, turning a cold tumor into a hot one | AZD8205 (AstraZeneca) | ★★★★★ |
| Alpha-Nuclide RDC | High-energy, short-range radiolytic killing | Ac-225-labeled drugs (in development) | ★★★★☆ |
| Beta-emitting RDC | Medium-energy, medium-range | Lu-177-PSMA (Pluvicto) | ★★★☆☆ |
| PROTAC-ADC | Targeted protein degradation (intracellular targets) | Early-stage research | ★★☆☆☆ |
[Practical Insights from the Conference] If the ADC projects you’re following still rely on traditional MMAE/DM1 payloads and target crowded fields like HER2 or TROP2, the room for differentiation is already extremely limited. What truly deserves attention are projects that dare to experiment with cross-disciplinary approaches to payload mechanisms—whether immune stimulation or radioisotopes, these represent the evolutionary direction of ADC 3.0.
2.1.2 The BsADC Boom: How Dual-Target Synergy Can Overcome Tumor Escape
Bispecific Antibody-Drug Conjugates (BsADCs) are likely to be one of the hottest technical topics at PEGS 2026. The core logic is this: the greatest challenge facing single-target ADCs is tumor escape (achieved by downregulating target antigen expression), whereas a dual-target design can significantly reduce the probability of such escape.
From an engineering perspective, BsADCs feature several mainstream architectures: CrossMab (resolves light chain mismatch by swapping CH1/CL domains; preferred by Roche), DART (a miniaturized design based on Fv fragments, offering enhanced tumor penetration),, DVD-Ig (which串联s two variable regions and has a relatively simple structure), and TandAb (which串联s two scFv fragments and has a molecular weight between that of a full-length antibody and DART).
In 2025–2026, early-stage clinical data for BsADCs began to be released in a concentrated manner. AstraZeneca’s AZD9592 (an EGFR/c-Met bispecific antibody-drug conjugate) demonstrated remarkable preliminary efficacy in non-small cell lung cancer, while Merck’s MK-1022 (an HER2/HER3 bispecific antibody-drug conjugate) also showed differentiated potential in the field of breast cancer.These data validate a key hypothesis: dual-target designs can indeed overcome resistance issues associated with single-target ADCs to a certain extent.
Table 2-3: Comparison of Mainstream BsADC Technical Architectures
| Architecture Type | Representative Companies | Advantages | Core Challenges | Applicable Scenarios |
| DVD-Ig | AbbVie, J&J | Simple structure, flexible engineering | Stability, immunogenicity | Systemic administration |
| CrossMab | Roche | Resolves light chain mismatch; mature process | High molecular weight, limited tissue penetration | Long-acting applications |
| DART | MacroGenics | Compact size, strong tumor penetration | Short half-life, requiring frequent dosing | Solid tumors |
| TandAb | Affimed | Moderate molecular weight, high flexibility | Significant room for CMC process optimization | Hematologic Cancers |
[Practical Insights from the Conference] A bispecific ADC (BsADC) is not simply “1+1=2”; its value lies in achieving synergistic effects through a rational combination of targets. When evaluating a BsADC project, the key question is: Do these two targets exhibit biological synergy? If target selection is merely a “pile-up of popular targets,” the advantages of the BsADC cannot be fully realized.
2.2 AOC (Antibody-Oligonucleotide Conjugate): The “Pathfinder” for Nucleic Acid Therapeutics
If BsADCs represent a natural evolution within the ADC field, then AOCs (Antibody-Oligonucleotide Conjugates) signify a true cross-disciplinary revolution. By combining the precise targeting capabilities of macromolecular antibodies with the powerful regulatory capabilities of nucleic acid therapeutics, AOCs are opening up therapeutic areas that traditional ADCs struggle to reach.
2.2.1 How Receptors Like TfR1 Solve Delivery Challenges to Muscle and the Nervous System (The Dark Horse Sector to Watch in 2026)
The biggest bottleneck for nucleic acid therapeutics has never been efficacy, but rather delivery. Naked nucleic acid molecules cannot penetrate cell membranes, are easily degraded by nucleases, and—more problematically—struggle to reach tissues beyond the liver.The liver is a “haven” for nucleic acid therapeutics because hepatocytes express abundant ASGPR (asialoglycoprotein receptor), which efficiently internalizes GalNAc-modified nucleic acid molecules. However, tissues such as muscle, the nervous system, and the heart lack such “natural entry points” as ASGPR.
This is where AOCs come into play. By conjugating nucleic acid therapeutics to antibodies that target specific receptors, tissue-specific delivery beyond the liver can be achieved. The most advanced example to date is TfR1 (transferrin receptor 1)-targeted AOCs: TfR1 is highly expressed in muscle tissue, making it an ideal target for AOCs in muscle-related diseases.Avidity Biosciences’ AOC 1001 (which delivers DMPK siRNA targeting TfR1 for the treatment of type 1 myotonic dystrophy) has entered clinical trials, with positive early data. Novartis’ acquisition of Avidity was driven precisely by the platform’s potential for expansion in the field of muscle diseases.
Table 2-4: Comparison of AOC vs. Traditional Nucleic Acid Drug Delivery Technologies
| Delivery Technology | Target Tissue | Representative Drug/Platform | Advantages | Limitations |
| GalNAc-siRNA | Liver | Onpattro, Givosiran (Alnylam) | Highly efficient maturation | Liver-specific |
| LNP-mRNA | Liver (default) | COVID-19 Vaccines (Pfizer/Moderna) | Capable of encapsulating large-molecule mRNA | Immunogenicity risk |
| TfR1-AOC | Muscle | AOC 1001 (Avidity/Novartis) | Breakthrough in extrahepatic delivery | Long-term safety to be validated |
| CD71-AOC | Multi-tissue | Early-stage research | Widespread receptor distribution | Less tissue-specific than TfR1 |
2.2.2 AOC Discussion in the PEGS ADC Track: The Convergence of Large and Small Molecules Is an Inevitable Trend
The agenda for PEGS indicates that the organizers are placing increasing emphasis on AOCs year by year. In 2025, AOCs were still merely a “marginal topic” within the ADC track, but by 2026, they are expected to become one of the core topics. Although the official website has not established a separate AOC track, the “Driving Clinical Success in ADC” track will certainly delve deeply into this technical direction of small-molecule-macromolecule fusion—this shift itself demonstrates a shift in the industry’s focus.
From a biological perspective, complex diseases—especially those regulated by multiple genes—are difficult to address with drugs based on a single mechanism. Antibodies excel at targeting cell surfaces, small molecules excel at penetrating cell membranes, and nucleic acid therapeutics excel at regulating gene expression—only by combining them can we effectively tackle truly complex diseases.From a commercial perspective, homogenized competition in traditional ADCs has already closed the early-stage window; companies must seek differentiated pathways, and AOCs offer a relatively untapped competitive landscape.
[Practical Insights from the Conference] AOC is not merely an “extension of ADCs,” but a “game-changer” in the nucleic acid therapeutics field. If your target indications include muscular disorders, neurological diseases, or any tissues other than the liver, AOC may represent a more effective technological pathway than traditional nucleic acid therapeutics.
2.3 The Truth About the “Survival Rate” of Bispecific Antibodies
Bispecific antibodies (BsAbs) are not a new concept, but at PEGS 2026, this topic will be approached with a more pragmatic perspective. After years of clinical validation, the industry has gained a clearer understanding of the “survival rate” of BsAbs: not all bispecific antibody designs can hold their ground in clinical trials, and engineering details determine success or failure.
2.3.1 Real-World Clinical Feedback on Frameworks Like CrossMab and DART
The PEGS track titled “Engineering Bispecific and Multispecific Antibodies” is expected to focus on real-world clinical feedback regarding different technical architectures. CrossMab (Roche’s core technology) is currently the most clinically validated bispecific antibody architecture.The success of emicizumab (Hemlibra, used to treat hemophilia A) demonstrates the effectiveness of CrossMab in resolving light-chain mismatch issues. However, the limitations of CrossMab are also evident: with a molecular weight approaching 150 kDa, its tumor penetration is inferior to that of miniaturized designs.
DART (MacroGenics’ technology platform) follows a miniaturization approach. Through Fv-based design, DART’s molecular weight can be controlled at around 50–60 kDa, offering significantly better tumor penetration than full-length bispecific antibodies. However, this comes at the cost of a substantially shorter half-life, requiring more frequent dosing. BiTE (Amgen’s technology platform, with Blinatumomab already approved) faces the same half-life issue and requires continuous infusion.
Table 2-5: Assessment of “Survival Rate” for Bispecific Antibody Technology Architectures (from Clinical Enrollment to Approval)
| Architecture | Number of Clinical-Stage Projects (Est.) | Number of Approved Drugs | Primary “Causes of Failure” | 2026 Interest in PEGS |
| CrossMab | 20+ | 1 (Emicizumab) | Immunogenicity, CMC Challenges | ★★★★☆ |
| BiTE | 15+ | 1 (Blinatumomab) | Short half-life, inconvenient administration | ★★★☆☆ |
| DART | 10+ | 0 (Pending approval) | Half-life, efficacy signal | ★★★☆☆ |
| DVD-Ig | 10+ | 0 | Stability, Immunogenicity | ★★☆☆☆ |
Note: Data is estimated based on publicly available information; “success rate” refers to the proportion of candidates that progress from the clinical stage to approval
2.3.2 Integration with AI-Driven Protein Engineering and Emerging Peptide Therapies
A new trend at PEGS 2026 is that bispecific antibodies are no longer discussed in isolation but are examined within the same framework as AI protein engineering and peptide therapeutics. “Emerging Peptide Therapeutics” is a new track added in 2026, which in itself indicates that peptide therapeutics are regaining attention.
Through rational engineering modifications (such as cyclization, introduction of non-natural amino acids, and fatty acid modifications), the disadvantages of peptides in terms of stability and oral bioavailability are gradually being overcome.Even more noteworthy is the way AI-driven protein engineering is empowering bispecific antibody design: predicting the stability and expression levels of different domain combinations, optimizing CDR sequences to enhance target affinity, and simulating the three-dimensional interactions between bispecific antibodies and their targets—tasks that previously required extensive experimental validation are now seeing their timelines significantly shortened by AI.
Table 2-6: Overview of Trends in the Convergence of Bispecific Antibodies and Peptide Therapies
| Convergence Direction | Technical Features | Representative Cases | 2026 PEGS-Related Agenda |
| Bispecific Peptides | Two targeting sequences fused onto a single peptide backbone | Early-Stage Research | Emerging Peptide Therapeutics |
| Peptide-Antibody Fusions | Peptides as the “warhead” of antibodies | Proof-of-concept stage | Engineering Bispecific Antibodies |
| AI-Optimized Peptide Design | Machine Learning Prediction of Peptide-Target Interactions | Multi-disciplinary Academic/Industrial Collaboration Projects | Machine Learning for Protein Engineering |
2.4 GenAI and Smart Biologics: The “Smart Design” Insights You Shouldn’t Miss in the Plenary Fireside Chat
If ADCs, AOCs, and bispecific antibodies represent innovations in molecular form, then GenAI (Generative AI) is fundamentally transforming how these molecules are designed. The Plenary Fireside Chat at PEGS 2026 (May 14, 11:35 AM, titled “How to Think About Designing Smart Biologics in the Age of GenAI”) offers the best window into this trend.
The application of GenAI in biologics design occurs at three levels: the first level is sequence optimization (predicting the impact of mutations on protein stability and optimizing CDR sequences using ESM and AlphaFold-derived models); the second level is de novo design (generating entirely new protein structures that meet specific functional requirements without relying on natural protein scaffolds; the feasibility of this approach was demonstrated by David Baker’s team in 2024 with RFdiffusion);The third tier is smart biologics (which automatically regulate activity based on environmental signals such as disease-specific biomarkers, local pH, and enzyme activity).
[Practical Insights from the Conference] GenAI is not merely a “nice-to-have” tool; it is reshaping the core infrastructure of biopharmaceutical development. 2026 is a critical window of opportunity for establishing a framework for AI+biopharmaceuticals—if you still do not understand what tools like ESM, AlphaFold, and RFdiffusion can do, you may be left behind in the next three years.
3.0 bio conference boston Pitfall Guide: Reading Between the Lines at PEGS

The PEGS Boston agenda expands every year, but not all technical topics deserve equal attention. As an attendee, you need to learn to identify which technical paths are being downplayed—this often signals that industry giants have quietly cut their losses—and which topics are merely included in the agenda as a formality, with little substantive progress actually made.
In this chapter, I’m providing you with a blacklist and a guide to avoiding pitfalls. This isn’t alarmist rhetoric, but rather the judgment accumulated by industry veterans over years of attending the conference. Learning to read between the lines of what speakers say will help you stay clear-headed amid the flood of information and avoid wasting time and resources on directions that have already lost their competitive edge.
3.1 Identifying Polite Abandonment: Which Technology Pathways Are Being Downplayed at the Summit
What is a “polite withdrawal”? When a technology faces major clinical or commercial setbacks, but the relevant companies or research teams do not want to publicly admit failure, they choose to downplay the situation—they still attend the conference, but their presentations skirt around the critical issues, omit key data, and no longer outline future plans.
At PEGS 2026, several technical approaches may exhibit signs of this “polite abandonment”:
Sign #1: Certain first-generation BsADC projects. BsADCs are a hot area, but not all BsADCs are worth following.If you notice a presentation on a BsADC project that only discusses in vitro data without mentioning in vivo efficacy, focuses solely on mechanisms of action without addressing clinical progress, or highlights the technology platform without detailing specific pipeline candidates—this could be a sign of a polite withdrawal. First-generation BsADCs (where target selection lacks biological rationale and linker-payload design simply replicates the experience of single-target ADCs) are currently undergoing the rigorous test of clinical validation, and some projects may have already been quietly discontinued.
Signal 2: Certain “me-too” ADC projects. Homogeneous competition around targets such as HER2, TROP2, and CLDN18.2 has already closed the window of opportunity. If you see an ADC project still pursuing minor innovations (such as changing the linker or fine-tuning the DAR value) in these crowded target areas without true differentiation in payload mechanism or indication selection—this is likely a prelude to a “polite withdrawal.”
Table 3-1: Warning Signs of a “Courtesy Abandonment” to Watch for at PEGS 2026
| Signal Type | Specific Manifestations | Interpretation | Countermeasures |
| First-Generation BsADCs | Focuses solely on in vitro data while avoiding discussion of clinical progress | Clinical data is underwhelming; development may have been halted | Ask for specifics on clinical stage and enrollment status |
| Me-too drugs targeting crowded markets | HER2/TROP2 minor innovations with no breakthroughs in mechanism | Homogeneous competition, limited room for differentiation | Focus on payload or indication differentiation |
| Proof-of-concept stage technology | Extensive preclinical data, but no IND timeline | Challenges in technology translation | Inquire about progress on CMC and regulatory communications |
| Cross-disciplinary hype | Frequent changes in technical labels (AI+, Nucleic Acid+) | Core technology is not sufficiently robust | Inquire about core technology barriers and patent strategy |
3.2 The “Valley of Death” in Mid-Stage Clinical Development: Root Causes of Most ADC/BsADC Projects Stalling in Phase II
There is an open secret in the biopharmaceutical industry: Phase II clinical trials are the “valley of death” for most projects. For ADCs and BsADCs, this valley is particularly treacherous. The PEGS track “Driving Clinical Success in ADC” is expected to address this topic, but speakers rarely say outright, “Our project died in Phase II”; instead, they tend to express it in more circumspect terms.
There are typically three core engineering reasons for ADC/BsADC failures in Phase II:
First, linker stability issues. If the linker breaks down prematurely in the bloodstream, the payload is released before reaching the tumor, leading to systemic toxicity. This is one of the most classic failure patterns in the ADC field. When evaluating an ADC project, it is essential to scrutinize its plasma stability data for the linker—if a presenter avoids this topic or only presents in vitro stability data, it is a red flag.
Second, the balance between payload potency and toxicity. A payload that is too potent may result in unacceptable toxicity (particularly ocular and neurotoxicity), while a payload that is too weak fails to achieve the therapeutic window. BsADCs face even greater challenges in this regard—the dual-target design implies two distinct endocytosis efficiencies, and balancing payload release across both targets presents a complex engineering challenge.
Third, off-target toxicity. Even with a well-designed linker, the antibody itself may cross-react with normal tissues. The off-target risk for BsADCs is on the order of the square of that for single-target ADCs—both targets carry the potential for off-target effects, and the dual-target binding may lead to unexpected cross-reactions.
Table 3-2: Three Major Engineering Causes of Phase II Failures in ADCs/BsADCs
| Failure Type | Specific Manifestations | Engineering Root Cause | Early Warning Signs |
| Insufficient linker stability | Higher-than-expected systemic toxicity, narrow therapeutic window | Premature degradation in the bloodstream | Presentation of in vitro stability data only |
| Imbalance in payload efficacy | Insufficient efficacy or excessive toxicity | Failure to balance efficacy and toxicity | Lack of dose-exposure-response relationship data |
| Off-target toxicity | Adverse reactions in non-target organs | Antibody cross-reactivity | Lack of comprehensive tissue cross-reactivity studies |
3.3 CMC First-Mover Advantage: Why Is Process Equivalent to Drug?
For complex molecules such as ADCs, BsADCs, and AOCs, CMC (Chemistry, Manufacturing, and Control) is not an issue to be considered only in the late stages of development; rather, it must be incorporated as a core element of the design from the laboratory phase onward. PEGS’s dedicated tracks on “Maximizing Protein Production Workflows” and “Characterization for Novel Biotherapeutics” are expected to delve deeply into this topic.
The phrase “process is drug” is particularly applicable in the field of complex biopharmaceuticals. No matter how well a molecule performs in the laboratory, if it cannot be produced stably, reproducibly, and at scale, it will forever remain a research tool rather than a drug. For ADCs, CMC challenges include: consistency in antibody-payload conjugation efficiency, batch-to-batch control of the DAR (drug-to-antibody ratio), removal of aggregates, and stability testing.
The CMC challenges for BsADCs are on the order of magnitude of those for standard ADCs. Two distinct antibody fragments must be precisely paired; light-chain mismatch issues must be resolved; and the expression balance between the two targets requires fine-tuning. This is why, despite its limitations, the CrossMab architecture still holds an advantage in terms of process maturity—Roche’s years of accumulated process optimization experience cannot be matched by new architectures in the short term.
For AOCs, the CMC challenges lie in the chemical synthesis and purification of the nucleic acid payload. Solid-phase synthesis of oligonucleotides exhibits batch-to-batch variability, and controlling coupling efficiency with antibodies is more complex than with traditional payloads. A key factor in Novartis’ acquisition of Avidity was the CMC process maturity of its AOC platform.
Table 3-3: Key CMC Considerations for Complex Biologics
| Molecular Type | Core CMC Challenges | Process Maturity Benchmarks | 2026 PEGS-Related Agenda |
| ADC | Coupling Efficiency, DAR Control, Aggregate Removal | Enhertu (Daiichi Sankyo/AZ) | Maximizing Protein Production Workflows |
| BsADC | Light-chain pairing, expression balance, stability | Emicizumab (Roche) | Characterization of Novel Biotherapeutics |
| AOC | Nucleic acid synthesis purity, conjugation efficiency, stability | AOC 1001 (Avidity/Novartis) | ML and Digital Integration |
[Practical Insights from the Conference] When evaluating an ADC/BsADC/AOC project, don’t just ask, “Does it work?”—also ask, “Can it be manufactured?” If a presenter avoids discussing CMC issues or only presents lab-scale data, this is a red flag. Truly mature projects will demonstrate at least pilot-scale CMC data early on.
3.4 Pitfalls of Target Selection: Why Are Popular Targets Often Pitfalls?
In the biopharmaceutical field, there is a counterintuitive phenomenon: the hottest targets are often the most dangerous pitfalls. Targets like HER2, TROP2, and CLDN18.2 are popular because they have been validated—but this also means they have been over-exploited.
Three pitfalls of target selection:
Pitfall 1: The Follower’s Trap. When multiple drugs for a target have already been approved or are in development, it is difficult for latecomers to differentiate themselves. Unless you possess breakthrough technology (such as a novel payload mechanism or a dual-target design), a follow-on strategy is almost guaranteed to fail in a crowded market.
Pitfall 2: The Biomarker Trap. Some targets perform well in preclinical models, but their expression profiles in humans differ significantly from those in models. If a project lacks sufficient human tissue expression data to support it, the error in target selection may not be discovered until Phase II.
Pitfall 3: The Indication Trap. The value of the same target can vary significantly across different indications. For example, HER2 is a high-value target in breast cancer but may have limited value in certain other tumor types. Blind cross-indication development is a common pattern of failure.
Table 3-4: Red Ocean vs. Blue Ocean Assessment of Target Selection
| Target Type | Representative Targets | Competitive Landscape | Difficulty of Differentiation | Recommended Strategy for 2026 |
| Red Ocean Targets | HER2, TROP2, CLDN18.2 | 10+ Projects in Development | Extremely High | Avoid unless breakthrough technology is available |
| Orange Ocean Targets | B7-H4, Nectin-4, EGFRvIII | 3–5 projects | Moderate | Feasible, but requires rapid advancement |
| Blue Ocean Targets | Newly discovered tumor antigens, tissue-specific receptors | 0–2 projects | Low | Focus on, but biological validation is required |
[Practical Insights from the Conference] When hearing about a target at PEGS, your first reaction shouldn’t be, “This target is trending; it’s worth following up on,” but rather, “Why hasn’t this target been successfully developed yet?” If a target has been attempted by multiple companies but has failed every time, there may be an inherent issue with the target itself (e.g., unsuitable expression profile, low endocytosis efficiency).If a target is very new but the presenter cannot clearly explain the biological rationale, it may be conceptual hype.
3.5 “Data Makeup”: How to See Through Embellished Clinical Data
When presenting data, speakers often employ various “tricks” to make the results look better. As an attendee, you need to learn to recognize these techniques and see through the data to the truth behind it.
Common data manipulation techniques:
Tactic 1: Selective Presentation. Showing only the best subgroup data while omitting the overall data. For example, “In patients positive for XX biomarker, the ORR reached 60%”—but if biomarker-positive patients account for only 20% of the total, the overall ORR might be as low as 20%.
Tactic 2: Timing Selection. Presenting data from the time point with the best results. For example, showing data at the peak of tumor shrinkage but omitting subsequent progression due to drug resistance.
Tactic 3: Manipulation of the control group. Select the control group that shows the greatest disparity with the study drug. For example, using an outdated standard of care as the control rather than the current best available treatment.
Tactic 4: Downplaying Toxicity Data. Describing serious adverse events as “manageable” or “clinically controllable” without presenting specific incidence rates.
Table 3-5: Guide to Identifying Clinical Data “Tricks”
| Type of Manipulation | Common Phrases | Identification Methods | Follow-Up Strategies |
| Selective Reporting | “In the XX subgroup…” | Check whether data for the overall population is presented | “What is the ORR for the overall population?” |
| Selecting time points | “The best response rate is…” | Check if data on duration of response is available | “What is the median duration of response?” |
| Control group manipulation | “Superior to standard treatment” | Check what the control group specifically is | “Is the control group the current standard of care (SOC)?” |
| Downplaying toxicity | “Clinically manageable” | Check the specific incidence and grade of AEs | “What is the incidence of Grade 3 or higher AEs?” |
[Practical Insights from the Conference] During the Q&A session at PEGS, asking a probing technical question is more valuable than simply saying “Great talk.” Good follow-up questions include: “Have you observed any off-target toxicity signals?” “How is the batch-to-batch consistency of the CMC?” “What are your differentiating advantages compared to the current standard of care?” These questions help you determine whether the presenter is showcasing genuine progress or engaging in PR.
3.6 Partner Due Diligence: How to Quickly Evaluate Potential Partners at PEGS
PEGS is not only a place for learning but also for finding partners. But how can you assess the reliability of a potential partner in a short amount of time? Here are the key points for a quick due diligence:
Evaluation Dimension 1: Scalability of the Technology Platform. Can their claimed technology platform truly be scaled to multiple targets/indications? Or is it merely the success of a specific molecule being packaged as a platform?
Evaluation Dimension 2: The team’s track record. Does the core team have a history of successfully advancing projects from early-stage to late-stage clinical trials? Or do they have only academic backgrounds and lack industrial experience?
Evaluation Dimension 3: Sustainability of Funding. Can their current funding sustain them until the next inflection point? Or will they soon need to raise capital, potentially forcing them to accept unfavorable terms?
Evaluation Dimension 4: Clarity of Intellectual Property. Is the ownership of the core technology’s IP clear? Are there any potential infringement risks or licensing disputes?
Table 3-6: PEGS On-Site Rapid Due Diligence Checklist
| Evaluation Dimension | Key Questions | Red Flags | Green Flags |
| Technical Platform | Has the platform been validated for multiple targets? | Only one molecular data point, yet it is claimed to be a platform | There is proof-of-concept data for 2+ different targets |
| Team Background | Does the team have IND/BLA experience? | Purely academic background, no industrial experience | Do core members have a track record of successfully advancing projects? |
| Funding Status | Will funding last until the next milestone? | Just completed Series A funding and claims to be moving forward quickly | Adequate funding with a clear roadmap |
| IP Status | Is the ownership of the core IP clear? | IP comes from third-party licensing, with unclear terms | Owned IP or clear licensing terms |
[Practical Insights from the Event] Any cooperation intentions established at PEGS must be followed up within 48 hours. Once the conference ends and everyone returns to their daily work, the momentum will quickly fade. Follow-up emails should be concise and clear: thank the recipient for the discussion, reaffirm your interest in collaboration, and propose specific next steps (such as scheduling a technical due diligence meeting).
4.0 bio conference boston Attendee Roadmap: Maximizing Your 120 Hours
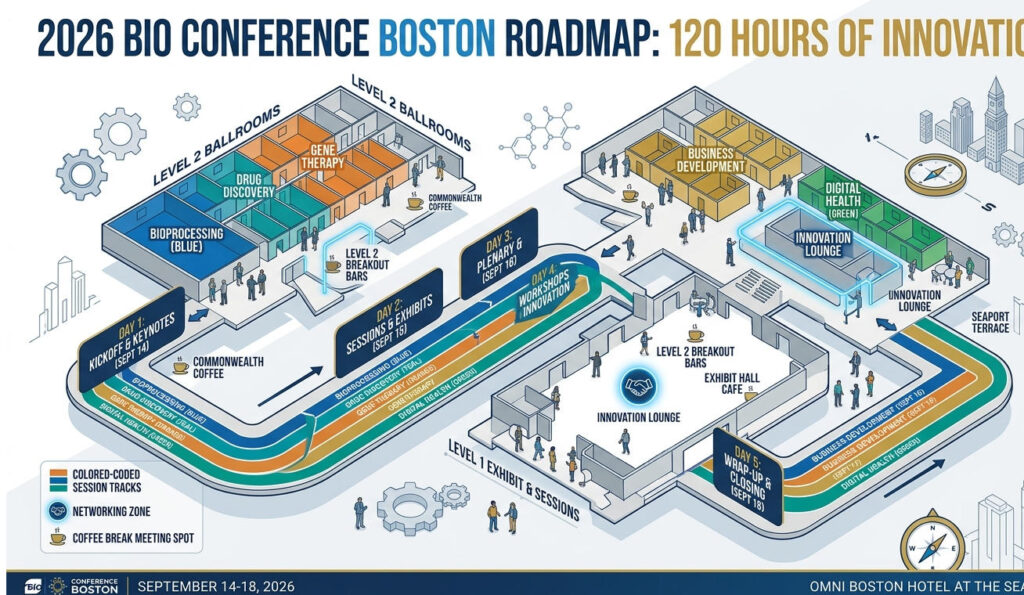
PEGS Boston is a 5-day conference. Assuming 8 hours of productive participation per day, you have approximately 120 hours at your disposal. How can you maximize value within these 120 hours? This chapter provides a practical roadmap to help you navigate the event like a seasoned veteran.
Core Principle: Over 50% of the value at PEGS Boston comes from Networking Breaks and Post-session Conversations, not the presentations themselves.
4.1 Role-Specific Itineraries
Attendees in different roles should focus on different program tracks. Below are customized route recommendations for two typical roles: R&D and BD/Investors.
4.1.1 R&D Roles: Must-Attend Tracks on Antibody Engineering & Multispecificity + Emerging Peptides
For R&D scientists, the core value of PEGS lies in understanding the latest technological advancements and engineering practices. Must-attend tracks for 2026 include:
First, “Engineering Bispecific and Multispecific Antibodies.” This is the most authoritative track in the bispecific antibody field, covering the latest engineering optimization experiences for mainstream architectures such as CrossMab, DART, and BiTE.
Second, Emerging Peptide Therapeutics. This is a new track for 2026, reflecting the resurgence of peptide therapeutics. It is particularly important for teams working on small molecule/peptide fusion projects.
Third, Driving Clinical Success in ADC. If you are involved in any ADC-related work, this track is a must-attend. The focus for 2026 is expected to be on early-stage clinical data for BsADCs and AOCs.
Table 4-1: Recommended Must-Attend Sessions for R&D Professionals at PEGS 2026
| Track Name | Recommended Date | Key Takeaways | Target Audience |
| Engineering Bispecific | May 12–13 | Optimization of Bispecific Antibody Architecture | Antibody Engineers, Protein Scientists |
| Emerging Peptide Therapeutics | May 14–15 | New Trends in Peptide Therapeutics | Peptide/Small Molecule Teams |
| Driving Clinical Success in ADC | May 13–14 | ADC/BsADC Clinical Data | ADC Project Team |
| Machine Learning for Protein Engineering | May 14 | AI-Driven Protein Design | Computational Biologist |
[Practical Insights from Attendees] R&D professionals attending the conference should not try to cover every session. It is more valuable to select 2–3 of the most relevant tracks and engage deeply with them than to listen superficially to all tracks. For each track, read relevant background literature in advance and prepare technical questions so that you can ask in-depth questions during the Q&A session and build connections with the speakers.
4.1.2 BD/Investors: Focus on the Fireside Chat + Young Scientist Alumni Panel
For BD professionals and investors, the value of PEGS lies not in understanding every technical detail, but in building professional networks and identifying early-stage investment opportunities. Key events to watch in 2026 include:
First, the Plenary Fireside Chat (May 14, 11:35 AM, topic: “How to Think About Designing Smart Biologics in the Age of GenAI”). This is the highest-profile dialogue session of the conference, typically featuring industry leaders sharing strategic insights.
Second, the Young Scientist Keynote Alumni Panel. This session often features presentations of the most innovative early-stage projects and serves as an excellent window for discovering unpriced innovation opportunities.
Third, satellite events in the Exhibition Hall. Many biotech companies host small receptions in the exhibition hall, providing an excellent opportunity to connect with founders and CSOs.
Table 4-2: Key Events for BD/Investors at PEGS 2026
| Event Type | Time/Location | Key Value | Action Recommendations |
| Plenary Fireside Chat | May 14, 11:35 AM, Main Hall | Strategic Insights from Industry Leaders | Arrive early to secure a seat and prepare your questions |
| Young Scientist Panel | See the agenda for details | Discovery of Early-Stage Innovation Projects | Note down projects and contacts of interest |
| Exhibition Hall Side Events | Exhibits in the exhibition hall | Direct interaction with the founders | Review the list of exhibiting companies in advance |
| Networking Breaks | Breaks between tracks | Informal networking | Prepare business cards and an elevator pitch |
[Practical Insights for Attendees] The key for BDs and investors attending the event is to “be present.” Don’t hide in your hotel room replying to emails, and don’t just sit in the back row listening to presentations. Take the initiative—approach speakers during coffee breaks, actively engage with booth staff in the exhibition hall, and proactively switch tables at receptions to meet new people. The value you gain from PEGS is directly proportional to the time you invest on-site.
4.2 The Unwritten Rules of Networking
Over 50% of the value at PEGS Boston comes from Networking Breaks and Post-session Conversations, not the presentations themselves. Mastering the unwritten rules of networking is the key to a successful conference experience.
4.2.1 Why Networking Breaks Are More Valuable Than Presentations
Formal presentations are often meticulously prepared, with information “polished” to perfection. However, during informal conversations at coffee breaks, you’ll hear more “unvarnished” and authentic insights: the actual progress of a project, internal developments at a company, or the real challenges of a particular technical approach.
Practical Tips: During coffee breaks, proactively approach speakers whose topics interest you. Instead of simply saying “Great talk,” ask a specific question. For example: “The linker stability data you mentioned in your presentation was very interesting. I’d like to know more about batch-to-batch consistency at pilot scale.” Such specific questions are more likely to spark in-depth discussions.
Another tip is to wait by the podium. After the presentation, wait by the podium for an opportunity to speak with the speaker one-on-one. At this time, there is the least competition, and you can have the most in-depth conversation.
4.2.2 Targeted Engagement Strategies for the Exhibition Hall, Coffee Breaks, and Satellite Events Around Seaport
The exhibition hall is a key battleground for networking. Recommended strategies:
First, research the list of exhibiting companies in advance and mark the 5–10 companies you most want to speak with. Don’t wander aimlessly through the hall; have a clear objective.
Second, don’t just pick up materials at the booth; proactively ask, “Is your technical lead here today? Could I schedule a brief 15-minute meeting?” Many booth staff are merely sales representatives, while the actual technical decision-makers may be in a meeting room.
Third, there are many satellite events (Satellite Symposia) around Seaport, typically dinners or breakfast meetings sponsored by biotech companies or CRO/CMO firms. These events often have low entry barriers but offer the chance to sit at the same table and network with key decision-makers from your target companies. We recommend contacting the organizers in advance via their company website or LinkedIn to request an invitation.
Table 4-3: Checklist of Practical Networking Tips
| Scenario | Specific Tips | Points to Note | Expected Outcome |
| Networking During Coffee Breaks | Ask specific technical questions | Avoid generalities | Gain access to non-public information |
| Visiting the Exhibition Hall | Schedule an appointment with the technical lead in advance | Prepare a self-introduction | Establish Initial Contact |
| Side Events | Take the initiative to move between tables and network | Bring plenty of business cards | Expand your professional network |
| Post-session | Wait by the podium | Prepare 1–2 in-depth questions | Connect with the speaker |
[Practical Insights from Attendees] The true value of PEGS Boston often lies beyond the official program. A seasoned attendee’s strategy is to selectively attend sessions (focusing only on the 2–3 most relevant tracks) and spend the majority of their time networking. Remember, you’ve flown over ten hours to Boston not to listen to PowerPoint presentations, but to build the kind of connections that can’t be made through email.
4.3 The Art of Time Management: How to Plan Your 5-Day Schedule
Five days may seem like a long time, but without proper planning, it’s easy to fall into a state of “busy but unproductive.” Here are some time management tips:
Day 1 (Monday): Focus on observation—familiarize yourself with the venue layout, attend the Opening Session to understand the overall agenda direction, and spend most of your time exploring the exhibition hall and doing light networking.
Day 2–3 (Tuesday–Wednesday): Deeply engage in core tracks (based on your role)—this is the primary time for technical learning. Use coffee breaks and lunch to network.
Day 4 (Thursday): Attend major events such as the Plenary Fireside Chat, and begin scheduling one-on-one meetings (pre-arranged). This is a critical day for business development (BD) professionals and investors.
Day 5 (Friday): Attend events like the Young Scientist Panel to discover early-stage projects, organize your meeting notes, and send follow-up emails. In the afternoon, you can schedule meetings with local partners.
Table 4-4: Recommended 5-Day Conference Schedule
| Date | Main Activities | Recommended Time Allocation | Key Outputs |
| Day 1 | Opening Ceremony, Initial Tour of the Exhibition Hall | 40% Lectures / 60% Networking | Build initial professional connections |
| Day 2–3 | In-depth Participation in Core Tracks | 60% Lectures / 40% Networking | Technical Learning and In-Depth Exchange |
| Day 4 | Plenary Sessions and One-on-One Meetings | 30% Lectures / 70% Networking | High-level connections, partnership opportunities |
| Day 5 | Early-stage projects, follow-up and organization | 20% Presentations / 80% Follow-up | Follow-up emails, action plans |
4.4 Pre-Conference Checklist: Maximizing the Value of 120 Hours
A successful PEGS experience starts with preparation. Here is a recommended checklist:
2 weeks before the event: Download the official app, review the full agenda, and mark must-attend sessions and people you want to meet. Also, update your LinkedIn profile to ensure potential contacts see your latest information.
1 week before the event: Schedule one-on-one meetings with key individuals via the app or email. Don’t wait until you’re at the venue to make appointments—by then, everyone’s schedule will be fully booked.
3 days before the event: Stock up on business cards or set up your digital business card (e.g., HiHello). Also, prepare your company’s elevator pitch—a 30-second explanation of what you do and why you’re unique.
1 day before the event: Organize your company presentation and project materials (such as a one-pager), and prepare follow-up email templates to facilitate quick follow-ups after the event.
Table 4-5: PEGS Boston 2026 Pre-Event Checklist
| Preparation Items | Recommended Time | Details |
| Review the agenda | 2 Weeks Before the Event | Download the app, mark must-attend talks and people you want to meet |
| Conference Registration | 1 week before the conference | Schedule one-on-one meetings with key speakers via the app or email |
| Prepare business cards | 3 days before the event | Prepare enough business cards or set up your digital business card |
| Organize materials | 1 day before the meeting | Organize company profiles and project materials, and prepare your elevator pitch |
| Follow-up plan | 1 day before the meeting | Prepare follow-up email templates to facilitate quick follow-ups after the meeting |
[Practical Insights from Attendees] The quality of your pre-meeting preparation directly determines the outcome of the event. Those attendees who appear to be “in their element” have all invested significant time in preparation beforehand. Another key point is that the speed of your follow-up matters more than the content itself. Sending a follow-up email within 48 hours of the meeting yields a success rate several times higher than sending one a week later.
5.0 Regulation & Future at bio conference boston: FDA Trends & Next-Gen Targets
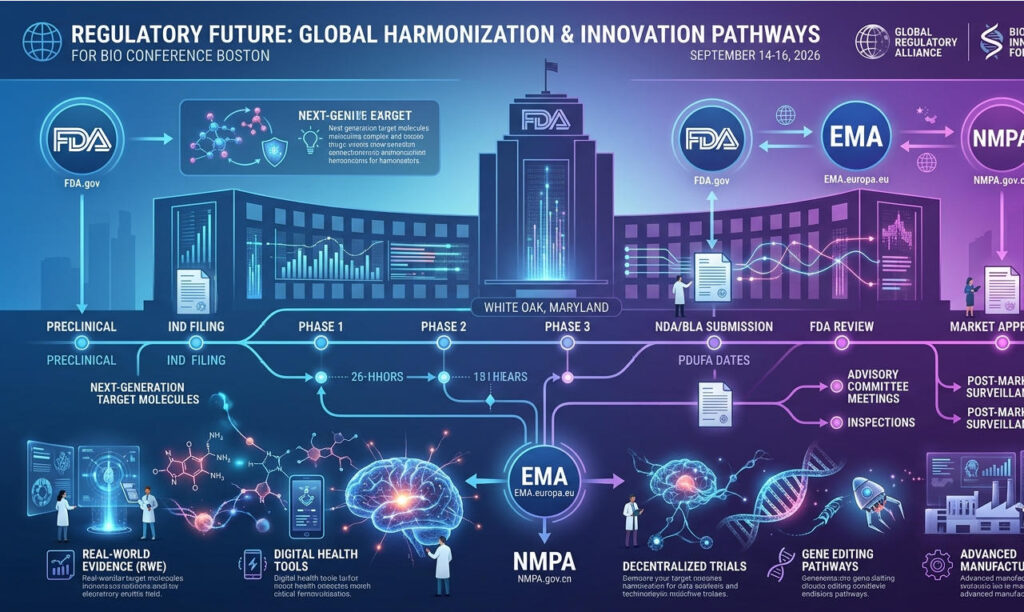
PEGS Boston serves not only as a platform for technical exchange but also as a vital window into regulatory trends. Officials from the FDA and EMA, industry regulatory consultants, and representatives from companies that have recently achieved key regulatory milestones all gather here to share their latest insights on the trends in the review of novel biologics.
For developers, understanding regulatory trends is just as important as understanding technological trends—a project that is technically flawless but not feasible from a regulatory standpoint is equally worthless.
5.1 Proactive Planning for Regulatory Compliance: Subtle Shifts in the FDA’s Review of AOCs, BsADCs, and Novel Conjugates
The review framework for novel conjugates (AOCs, BsADCs, RDCs, etc.) is rapidly evolving. Compared to monoclonal antibodies, the complexity of these molecules presents new challenges for regulatory authorities: How should the off-target risk of bispecific targets be assessed? How should CMC quality control standards for AOCs be established? How should dose-exposure-effect relationship studies for radiopharmaceuticals be designed?
Based on the FDA’s review practices in recent years, several trends are worth noting:
First, CMC upfront. For complex conjugates, the FDA increasingly emphasizes the establishment of a robust CMC control strategy starting from the IND stage. This means developers must resolve many process issues before entering clinical trials, rather than gradually optimizing processes during the clinical phase as with traditional antibodies.
Second, the growing importance of biomarkers. For molecules with complex mechanisms such as BsADCs and AOCs, the FDA encourages developers to establish a biomarker system capable of predicting efficacy and safety. This is not only a scientific issue but also a regulatory strategy—reliable biomarkers can accelerate clinical development and reduce the risk of failure.
Third, the expansion of non-clinical safety assessments. Traditional ADC safety evaluations primarily focus on payload toxicity, but BsADCs and AOCs require more comprehensive assessments—including cross-reactivity between dual targets, off-target effects of nucleic acid payloads, and the safety of impurities associated with the conjugation process.
Table 5-1: Evolution of FDA Review Focus Areas for Novel Conjugates
| Molecular Type | CMC Focus | Nonclinical Requirements | Clinical Strategy Recommendations |
| Traditional ADCs | DAR Control, Aggregate Removal | Standard Toxicology Package | Dose escalation + expansion cohort |
| BsADC | Light chain pairing validation, dual-target DAR | Dual-Target Cross-Reactivity Assessment | Biomarker-Driven Enrollment |
| AOC | Nucleic acid purity, conjugation efficiency | Assessment of nucleic acid off-target effects | Tissue-specific PK/PD |
| RDC | Radioactive labeling efficiency and stability | Radiation dosimetry assessment | Image-Guided Dose Optimization |
[Practical Insights from Attendees] When listening to regulatory-related content at PEGS, pay close attention to “experiences shared by companies that have just completed FDA communications.” This firsthand information often holds greater practical value than official guidelines. In particular, presentations where speakers are willing to share “what questions the FDA asked” can help you understand the regulator’s current focus areas.
5.2 Opportunities in Non-Traditional Targets: From the Cell Surface to the Cytoplasm—How Protein Engineering Can Break Through the “Undruggable” Impasse
Traditional antibody drugs primarily target cell surface proteins, but the majority of potential therapeutic targets in the human genome are located intracellularly. How to overcome this “undruggable” barrier is a key topic at PEGS 2026.
Several noteworthy technological directions:
First, intracellular delivery technologies. Through engineering modifications, antibodies or antibody fragments can be engineered to penetrate the cell membrane and enter the cell. This includes cell-penetrating peptide (CPP) fusion, protein transduction domain (PTD) modification, and designs based on endocytosis-escape mechanisms.
Second, Targeted Protein Degradation (TPD). Technologies such as PROTACs and molecular glues can directly target intracellular proteins and degrade them by hijacking the cell’s ubiquitin-proteasome system. The “Emerging Targets for Oncology & Beyond” track at PEGS 2026 is expected to discuss this area.
Third, RNA-targeted therapies. In addition to AOCs, there are small molecules that directly target mRNA (such as splicing modulators) and CRISPR-based gene editing technologies. These technologies can regulate the expression of any gene, unrestricted by the location of the target.
Table 5-2: Technological Pathways to Overcome the “Undruggable” Barrier
| Technological Pathways | Mechanism of Action | Representative Technologies/Companies | 2026 PEGS-Related Agenda |
| Intracellular Delivery | Antibody/Fragment Penetration of Cell Membranes | CPP Fusion, PTD Modification | Emerging Peptide Therapeutics |
| Targeted Protein Degradation | Hijacking the UPS system to degrade target proteins | PROTAC, molecular glue | Emerging Targets for Oncology |
| RNA Targeting | Regulation of mRNA splicing or stability | Splicing modulators, ASOs | AOC-Related Discussions |
| Gene editing | Permanent correction of genetic defects | CRISPR-based therapies | Machine Learning for Protein Engineering |
5.3 Next-Generation Target Selection Strategies: From “Hot” to “Rational”
The logic behind target selection is evolving. In the past, selecting a “hot” target (such as HER2 or PD-1) was a relatively safe strategy, as there were already validated pathways to follow. However, in the current landscape, “hot” often implies a “red ocean,” where differentiation is more important than following the crowd.
Next-generation target selection should consider the following dimensions:
Dimension 1: Biological Rationale. Does the target play a key role in disease onset and progression? Is there sufficient genetic evidence to support it (e.g., GWAS data, rare disease mutations)?
Dimension 2: Drugability. Does the target have a suitable binding site? For intracellular targets, are there viable delivery technologies available?
Dimension 3: Competitive Landscape. Has the target been over-developed? If multiple drugs have already been approved, where is the differentiation opportunity for latecomers?
Dimension 4: Clinical Translation Pathway. Is the pathway from target validation to clinical proof of concept (POC) clear? Are there suitable patient populations and biomarkers?
Table 5-3: Strategy for Selecting Next-Generation Targets
| Target Type | Traditional Challenges | Emerging Solutions | Therapeutic Area |
| Intracellular signaling proteins | Antibodies cannot enter cells | PROTAC, intracellular delivery technologies | Cancer, Autoimmune Diseases |
| Transcription factors | Lack of enzymatic active sites | Molecular glue, protein degradation | Cancer, Rare Diseases |
| RNA targets | Poor stability, difficult delivery | AOC, LNP encapsulation | Neuromuscular diseases |
| Epigenetic targets | Poor selectivity | Targeted degradation, AI-assisted design | Cancer, metabolic diseases |
[Practical Insights from the Conference] Regulatory strategy is not an afterthought but an element that must be incorporated into the overall development plan from the early stages of a project. For novel molecules such as AOCs and BsADCs, it is recommended to hold pre-IND meetings with the FDA prior to filing an IND to clarify CMC and non-clinical development requirements. At PEGS 2026, attendees are advised to pay close attention to the experiences shared by companies that have recently completed FDA communications—this firsthand information often holds greater practical value than official guidelines.
5.4 Trends in Global Regulatory Harmonization: Review Differences Among the FDA, EMA, and PMDA
With the globalization of biopharmaceutical development, understanding the review differences among various regulatory agencies has become increasingly important. The FDA, EMA (Europe), and PMDA (Japan) exhibit both convergence and divergence in their review of novel biologics.
Convergence Trends: Regarding complex conjugates, all three regulatory agencies are strengthening CMC requirements, encouraging early engagement with regulators, and emphasizing the use of biomarkers.
Differences: The FDA places greater emphasis on innovation and is more proactive in granting Breakthrough Therapy Designation, but its CMC requirements are stringent; the EMA focuses more on a comprehensive assessment of the risk-benefit ratio and has higher requirements for long-term safety data; the PMDA places greater emphasis on comparisons with existing treatments and has unique requirements for clinical endpoint design.
Table 5-4: Comparison of Review Characteristics Among Major Global Regulatory Authorities
| Regulatory Authority | Review Characteristics | Attitude Toward Novel Conjugates | Communication Recommendations |
| FDA | Encourages innovation, but has strict CMC requirements | Proactive, but requires comprehensive data | Early communication; emphasis on CMC |
| EMA | Comprehensive evaluation, with a focus on long-term safety | Caution; Requires a Complete Data Package | Comprehensive safety data |
| PMDA | Emphasis on comparison with existing treatments | Conservative, requiring Japanese data | Consider conducting early-stage clinical trials in Japan |
[Practical Insights from the Conference] If your project has global development plans, we recommend networking with professionals who have cross-border regulatory experience at PEGS. Many CROs and regulatory consulting firms exhibit at PEGS, making it an excellent opportunity to understand differences in regulatory practices across countries. Additionally, pay close attention to presentations sharing case studies on “How We Obtained Approval in Country X”; these practical insights are invaluable.
6.0 bio conference boston Conclusion: Your Organizational Action Plan
The five-day PEGS Boston trip is coming to an end, but the real value translation has only just begun. When you return to the office, facing a backlog of emails and projects awaiting decisions, you’ll need a clear action plan to translate the insights gained in Boston into actionable strategic steps.
6.1 Three-Dimensional Reporting Template: One Key Trend, Two Potential Partners, One Pipeline Risk Warning
When reporting your conference takeaways to management, avoid a mere chronological recap of the agenda. We recommend using this three-dimensional reporting template:
Dimension 1: One Key Trend. From all the information gathered at PEGS 2026, distill the single trend assessment most critical to your organization. For example: “BsADCs are transitioning from proof-of-concept to a clinical boom phase; we recommend initiating an evaluation of relevant technology platforms in Q3.” Or: “A breakthrough in AOC for muscular diseases means we need to reassess our strategic positioning for nucleic acid therapeutics.”
Dimension 2: Two Potential Partners. During networking sessions, identify two potential partners most worthy of further collaboration. These could be technology platform companies, CRO/CMO service providers, or biotech companies with complementary products. Prepare a one-page summary for each partner: who they are, what their core capabilities are, what the collaboration entry points are, and what the next steps are.
Dimension 3: A pipeline risk alert. Based on information gathered at PEGS, identify one pipeline risk that requires attention. For example: “Our HER2-ADC project in the pipeline faces competitive threats from BsADCs; we recommend accelerating clinical development or considering combination therapy strategies.”
Table 6-1: Example of a Three-Dimensional Reporting Template
| Dimension | Key Content | Recommended Reporting Format |
| Key Trends | BsADC clinical breakthrough, AOC muscle disease breakthrough, GenAI design maturity | 1-page slide: Core conclusions + supporting data |
| Partners | Technology Platform Providers, CROs/CMOs, Complementary Biotech Companies | 1 page per partner: Capabilities + Entry Points + Next Steps |
| Risk Warnings | Competitive Threats, Technological Substitutes, Regulatory Changes | 1-page slide: Risk description + mitigation recommendations |
6.2 From Information Nodes to Value Nodes
The true value of PEGS Boston lies not only in the information you bring back, but also in the position you establish within this network. Every time you attend, you become a node in the network of influence within the biopharmaceutical industry.
Post-conference follow-up strategy: First, send a follow-up email within 48 hours. For key contacts made at the conference, send a follow-up email within 48 hours. Second, internal knowledge sharing. Organize a PEGS takeaways session within your team. Third, continuously track key projects. Establish a continuous tracking mechanism for technologies or companies identified at PEGS that warrant attention.
Table 6-2: Post-Conference Follow-Up Action Plan
| Action Item | Timeframe | Details | Expected Output |
| Follow-up Email | Within 48 hours | Send a follow-up email to key contacts | Strengthen relationships and advance collaboration |
| Internal Sharing | Within 1 week | Organize an internal team sharing session | Spread knowledge and establish an expert image |
| Ongoing Follow-Up | Ongoing | Set up a watchlist and review progress regularly | Capture collaboration and investment opportunities in a timely manner |
| Management Reporting | Within 1 week | Prepare a three-dimensional presentation | Strategic Impact, Resource Support |
Take this guide with you to Boston. May 2026, Omni Boston Hotel at the Seaport—we’ll see you there.
7.0 bio conference boston FAQ: PEGS 2026 Essential Questions Answered
Q1: What is the difference between PEGS Boston and BIO International?
PEGS Boston focuses more on the technical depth of macromolecular engineering. Attendees are primarily R&D scientists, protein engineers, and CMC experts, and the program is more technically oriented. BIO International is a broader biopharmaceutical industry event covering all fields—including small molecules, macromolecules, cell and gene therapy, and diagnostics—with a more diverse attendee base (investors, government relations, business development, etc.).
If your primary focus is on the technical details of ADCs, bispecific antibodies, and antibody engineering, PEGS Boston is the better choice. If you need a broader industry perspective and more business development opportunities, BIO International may be a better fit.
Table FAQ-1: Comparison of PEGS Boston vs. BIO International
| Dimension | PEGS Boston | BIO International |
| Core Focus | Macromolecular Engineering Technologies | Across the Entire Biopharmaceutical Spectrum |
| Primary Attendees | R&D Scientists, Engineers | Investors, Business Development Professionals, Executives |
| Program Highlights | Technical Depth | Broad Coverage |
| Ideal for | Technical learning, early-stage project discovery | Extensive Networking, Policy Insights |
| 2026 | May 11–15 | Usually in June |
Q2: How should I plan my schedule for my first time attending PEGS?
We recommend a “2+1” strategy: select two tracks most relevant to your work and engage deeply (attend all sessions, take notes, and participate in Q&A), while setting aside one day for networking and visiting the exhibition hall. Don’t try to cover the entire program—that will only lead to information overload.
Download the official app in advance, mark the must-attend sessions and people you want to meet, and create a daily action plan. For first-time attendees, we recommend spending the first day primarily observing to get a feel for the venue atmosphere, and beginning in-depth participation starting from the second day.
Q3: Which technology direction—BsADC or AOC—is more worth focusing on in the long term?
This depends on your therapeutic area. If you’re in oncology, BsADC is the more mature, commercially viable direction, with clinical validation data accumulating rapidly. If you’re in non-oncology fields (particularly muscular or neurological disorders), AOC represents a truly breakthrough opportunity, as it addresses the biggest bottleneck in nucleic acid therapeutics—extrahepatic delivery.
In terms of risk-reward ratio, BsADC is suitable for investors seeking mid-term returns, while AOC is suitable for pioneers willing to take on higher risks in exchange for greater breakthroughs.
Table FAQ-2: Comparison of Investment/Focus Strategies for BsADC vs. AOC
| Dimension | BsADC | AOC |
| Technology Maturity | Relatively mature, with clinical data currently being accumulated | Early stage, proof-of-concept phase |
| Primary Indications | Oncology | Muscle disorders, neurological disorders |
| Competitive landscape | Gradually intensifying | Relatively untapped market |
| Risk level | Medium | Higher |
| Potential return | Above average | High (if successful) |
| Suitable for investors | Seeking medium-term returns | Early adopters willing to take on high risk |
Q4: How can you determine if a PEGS presentation is worth attending?
To assess whether a presentation is worth attending, consider the following factors:
First, the speaker’s background. Are they from industry or academia? Speakers from industry typically share more practical experience, while those from academia may focus more on basic research.
Second, content type. Is it early-stage research or clinical data? Is it proof of concept or commercialization experience? Choose based on your needs.
Third, company stage. Presentations from large pharmaceutical companies are often more “polished,” while those from small biotech firms may be more direct and authentic.
Fourth, session placement. Plenary sessions are usually the most important content of the entire event and should be prioritized.
Table FAQ-3: Quick Assessment of PEGS Presentation Value
| Evaluation Dimensions | High-Value Indicators | Low-Value Indicators |
| Speaker Background | Experience in successfully advancing projects | Purely academic background, no industry experience |
| Content Type | Clinical data, lessons learned from failures | Purely conceptual, overly hyped |
| Company stage | Projects in the clinical phase | Stuck at the proof-of-concept stage |
| Agenda Slot | Plenary, Core Tracks | Breakout Sessions, Breakout Tracks |
Q5: PEGS Boston 2026 Attendee Checklist
Table FAQ-4: PEGS Boston 2026 Attendee Checklist
| Preparation Items | Recommended Time | Details |
| Review the agenda | 2 Weeks Before the Event | Download the app, mark must-attend talks and people you want to meet |
| Conference Scheduling | 1 week before the conference | Schedule one-on-one meetings with key speakers via the app or email |
| Prepare business cards | 3 days before the event | Prepare enough business cards or set up your digital business card |
| Organize materials | 1 day before the meeting | Organize company profiles and project materials, and prepare your elevator pitch |
| Follow-up plan | 1 day before the meeting | Prepare follow-up email templates to facilitate quick follow-ups after the meeting |
| Dress Code | 1 day before the meeting | Opt for business casual attire and bring comfortable shoes (the venue is quite large) |